|
Welcome to the February edition of INNsight; after his passing this month, INNsight focuses on the life and the impact of John Woolfe on the generics industry. In addition, the Drug In Focus is Orlistat, and Peter Wittner discusses the rise of Asian generics.
If you were forwarded this email and would like to receive future editions of INNsight, become a member by clicking here.
| 'Drug In Focus' is written by Leighton Howard, a patent information expert with extensive generics experience. He has worked within the professional patent information industry and major generic pharmaceutical firms and now provides consultancy services to the generics industry and government organisations via his company XIP Pty Ltd. He is also the founder of GenericsWeb and remains instrumental in maintaining the high quality and accuracy of data contained in Pipeline Patent Intelligence. Please email any comments or queries. |
 |
Orlistat
This months Drug in Focus will relate to a drug that expires some time into the future, to assist in the development of generic products and identification of potential pitfalls whilst there is still time to address any arising issues. Based on information contained in the GenericsWeb Pipeline Selector report for Orlistat, we will analyse the patent landscape surrounding this product with a view to launching generic equivalents.
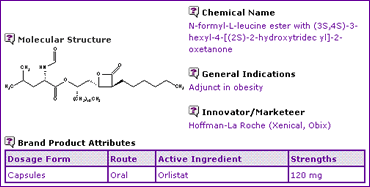
The Orlistat General Information (Table 1) indicates that the active ingredient is available only as an oral dosage form in a strength of 120mg, for use as an adjunct in the treatment of obesity.
Table 1: Orlistat General Information
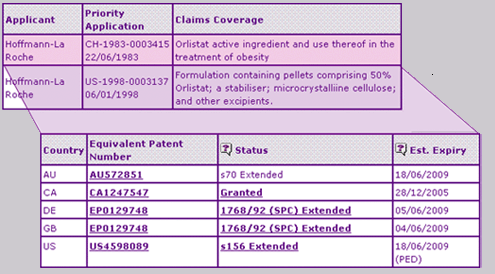
The Key Patent Indicator (Table 2) for Orlistat confirms that the extensions on the Orlistat active ingredient patent expire in Australia, Germany, the UK and the USA in June 2009, whereas the Canadian equivalent patent expired at the end of 2005 due to a 17-year patent term calculated from issue date combined with the lack of provisions for pharmaceutical patent extensions seen in the other countries. It is important to note that the US patent benefits from a paediatric extension that will further constrain generic competition until December 2009.
Table 2: Orlistat Key Patent Indicator
As is the norm these days with generic developments, there is more to Orlistat than meets the eye in terms of opportunities for generic launch, which may explain why a generic was not launched in Canada upon patent expiry.
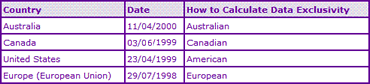
Data exclusivity occasionally offers the innovator protection beyond patent expiry and should always be considered when projecting development timings. The Data Exclusivity information (Table 3) for this product shows that it took more than 15 years from filing the first Orlistat API patent to attain marketing approval in a major market. Due to the varying manner in which data exclusivity provisions are applied in different countries (see our associated help sheets for further information), this affects potential generic launch in many ways. For example, in major European markets the regulatory authorities will not accept a generic application for Orlistat that relies on innovator data until July 2008, and applicants will likely not gain approval until some time after the active ingredient patent has expired. However some smaller European countries, Australia and the US may accept applications earlier, due to shorter data exclusivity provisions and will probably experience some generic competition immediately upon active patent expiry. In Canada, however, NOC's were not issued to generic competitors in time for patent expiry, and appear to have not been issued. This may be due to effect of the five-year data exclusivity provisions, which expired in June 2004, or it could indicate insurmountable patent problems due to the late-expiring formulation patent.
Table 3: Orlistat Data Exclusivity
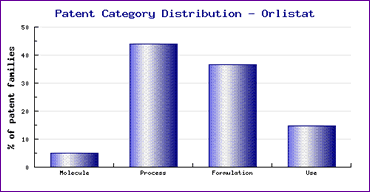
In addition to the molecule patent family, the KPI also indicates that another patent family protects the marketed oral formulation until 2018. This would account for the large proportion of formulation patents filed for Orlistat shown in the Patent Risk Analysis section (Figures 1 and 2). Such late patent filings often reflect the interest of generic competitors in circumventing key patents and preventing other generics from following suit however, in this unusual case, the filings are by the innovator, Hoffman-La Roche and were made prior to launch. This shows the effort that the innovator is prepared to go to in order to protects its market and thus has been rewarded by considerable strength in its patent position.
Figure 1: Orlistat Patent Category Distribution
Also noteworthy from the Patent Filing Trends graph (Figure 2) is the proportion of process patents filed prior to launch of the product, which can be safely assumed to be innovator filings. The response again by generic competitors is to file many more process patents later in the lifecycle, indicating that synthesis of the API is also a significant hurdle in bringing a non-infringing generic to the market.
These graphics indicate that caution is necessary in considering all relevant patents with a view to avoiding infringement of both the innovators key process and formulation patents and the generic competitors later patents.
Figure 2: Orlistat Patent Filing Trends
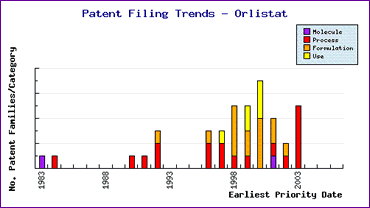
In summary, Orlistat is well protected by the innovators beyond the life of the active ingredient patent. Later patenting of processes and formulations by Hoffman-La Roche will cause development problems for many, lesser-informed generic competitors. Should these patents be circumvented by the handful of generic competitors who have already filed patents, data exclusivity provisions would appear to offer an additional period of monopoly in some major markets.
GenericsWeb Pipeline Selector reports are available for any active ingredient upon request at AU$400 each (US$300). Comprehensive data for patent families relating to Orlistat, based on professional patent searching, may be accessed by upgrading to the corresponding Pipeline Developer report, which includes twelve monthly updates to keep you abreast of recently published patents and applications. Until 15th March 2006, GenericsWeb are offering a 10% discount on the standard price of a Pipeline Developer subscription to Orlistat, reducing the cost to AU$3870 (US$2880).
Click here for more info on GenericsWeb's Pipeline Selector and Pipeline Developer patent intelligence.
Leighton Howard
Managing Director
GenericsWeb
February 2006
l.howard@genericsweb.com
Back to top
Generics legend John Woolfe passes away
Many people were shocked and saddened last week by the tragic news that John Woolfe, a well-known and highly respected member of the generics industry, had passed away.
I have since been in touch with some of Johns former colleagues who have been kind enough to assist me in preparing a few words to show respect for the great contributions John made to the generics industry, to his company and to the many people with whom he worked around the world.
John worked for many years at IVAX (formerly Harris Pharmaceuticals and then Norton Healthcare), and was a technical/research director for much of that time. He had an incredible technical knowledge across all the functions necessary to bring a generic product from concept through to launch, and would often be involved in every stage. John best applied this knowledge to the numerous patent actions in which he became involved, and thereby earned the friendship and respect of UK barristers and Intellectual Property QC's. Some of the most legally significant patent cases affecting the generics industry today were won on the back of Johns extreme intellect combined with a profound knowledge of chemistry and how this applied to patents and pharmaceuticals. Aside from this John, often without much assistance, succeeded in developing many more challenging generic products that circumvented key patents, providing significant opportunities for his company whilst leaving competitors in awe.
John had a unique way of filing the reams of information that he required to fulfil such an important role, and many of his colleagues will fondly remember the layers of documents strewn across his desk on a permanent basis. Despite this, John was able to locate any specific document in an instant; a task, which experience shows, would have taken him a considerable while longer if the document were stored on a computer. Many notable attempts were made to change Johns working habits to fit in with others expectations, but to succeed in doing so would have been to remove the brilliance that allowed him to achieve such amazing things.
John Woolfe is the reason I became involved in the generics industry, and am still involved in it today. I realise that without the kindness and generosity expressed by John in offering to teach me what he knew, I would not be so fortunate to have learned a small part of Johns seemingly infinite knowledge. Thinking that I was alone in such fortune, the remarks made by his other colleagues made me realise that this is by no means true. John, it seems, modestly and thanklessly assisted so many other of his colleagues in a variety of ways to help them learn and carry out their respective roles.
John was a talented bridge player and an avid stamp collector, having been a long-standing member of the Eastern Region of the Royal Philatelic Society London and last year winning the Large Vermeil medal for his collection of Early New Zealand Overseas Mail during the 1874-1908 Chalon era. John also collected a great deal of another kind of stamp - in his passport. Hardly a week went by when John wouldnt get on a plane to meet with his many colleagues and business contacts all around the world whilst leaving a lasting impression with new acquaintances.
John, through his dedication to his company, was therefore known and admired by a vast amount of people in the generics industry today. He also worked hard to enjoy a busy social life and win the friendship of many other like-minded people. I believe I speak for everyone who knew John when I say that he will be sadly missed both in work and social circles, and may we all remember the contribution John made to our lives in one way or another.
John Woolfe's funeral will be held on 22nd February 2006 at 2.00pm, at the Randalls Park Crematorium in Leatherhead, Surrey, United Kingdom. His family have requested that rather than flowers, they would prefer donations made to Macmillan Cancer Relief, which can be sent directly to the funeral directors at the following address:
WA True Love & Sons Ltd, 14-18 Church Road, Epsom KT17 4AB United Kingdom.
Leighton Howard
February 2006
Back to top
|
DEVELOPMENTS AT GENERICSWEB
|
|
Canadian and US data exclusivity
In addition to the existing European and Australian data, all new Pipeline Selector and Pipeline Developer reports now contain Canadian and US data exclusivity information.
New Pipeline Developer reports
Recent Pipeline Developer additions to our database include Bisoprolol, Cisatracurium, Exemestane, Gadopentetate, Indapamide, Mometasone, Nicorandil, Octreotide, Rabeprazole, Riluzole and Zolmitriptan. For assistance with the development of non-infringing generics for these or any other drugs, please contact us.
| Any questions? Click here to see our FAQs.
Back to top
If you are new to GenericsWeb you may not know that there is daily generics news archived at our site: Click here for this month's major generics news headlines
| Contributor Anna McKay specialises in IP Law and Strategy for Pharma companies. As an English solicitor, she won many leading patent cases before the Patents Court, Court of Appeal, EPO, and House of Lords for generic companies. She now works independently, advising companies worldwide on IP exploitation and strategy. She is also a qualified mediator. |
|
If you would like to contact Anna to give feedback or enquire about her services, please email anna@annamckay.com
John Woolfes impact on crucial outcomes for the generics industry
I thought I would add to Leightons recollections of John Woolfe and tell you a little of the cases on which John worked, and their importance for the Generic industry.
John was unique. I worked with him very closely in many patent and other actions I acted for Norton Healthcare and he briefed me.
Litigation can be a very intense experience, especially when one is acting for a generic company which is attacked by a weighty originator with seemingly unlimited resources. One often has to act very fast, under great pressure: it is stressful and a kind of environment in which one really comes to know ones colleagues. John could be curmudgeonly and kind at the same time. He rarely gave compliments. His glasses were not rose tinted, but some shade of grey, and he expressed his regard and affection through curious terms of endearment (he called me Chuck, which seemed to me quite improbable) and criticisms of everybody else. Like all of us, he had his flaws. I had a very soft spot for him, not least for his intelligence and decisiveness. He tended to be a little apologetic about both of those qualities.
Johns biggest contribution to the generics industry was probably in relation to licences of right and early litigation which preceeded my involvement with him.
Before the 1977 Patents Act, UK patents lasted for sixteen years. With the 1977 Act, they were extended to 20 years from the date of application, and there was a transitional period during which the final four years of the patent were endorsed licenses of right. In other words, it was possible for a generic company to obtain a licence of right to use an invention during the final four years of the patent. The legislation provided for reasonable royalties to be paid, but gave no guidance as to what these should be, and debate and opinion varied widely as to whether these should be the very low royalty figures which applied to licenses of right in, for example, Canada (around 4%) or extremely high percentages. In fact, relatively high royalties were eventually determined to be appropriate. It was the ability to obtain a licence of right which gave momentum to the generics industry, and Johns ability to pick molecules that could be manufactured easily enough, yet were difficult enough to deter competitors, and for which there was sufficient commercial opportunity, and possibly other patents which needed to be removed or avoided before marketing was possible, helped make Harris Pharmaceuticals (later called Norton, and then part of IVAX) the success it was at that time. Harris/Norton had an enviable reputation. It was quick to manufacture and quick to defend its position, and often first on the market.
When I first met John, I had had a non-contentious background, and I expected to do commercial work for pharma companies. Instead, I began to work with John on the Terfenadine case (Marion Merrell Dow v- Norton). John was good to work with, on what was my first patent case and it was an extraordinary case, and legally, very important. The molecular patent for Terfenadine had expired, and Norton was selling Terfenadine preparations. A later patent, which claimed a metabolite of Terfenadine, was still in force. That metabolite was made in the patients body after he had swallowed a tablet containing Terfenadine itself. Merrell Dow claimed that Norton were providing the means to patients to infringe the patent (s 60 (2) Patents Act 1977). In other words, they were providing a product for patients which inevitably produced a product which infringed the metabolite patent. Before the date of application for the metabolite patent, the fact that chemical activity resulted from the metabolite was not known. However, patients had already been generating metabolite unknowingly: clinical trials had already been carried out. It had always been the case that once something had gone into the public domain, others were free to do it. That principal was established many hundreds of years ago. We claimed that the metabolite patent was invalid for lack of novelty. Merrell Dow claimed that with the 1977 Patent Act the law had changed and that later patents could be used, in certain circumstances, to prohibit activities which had occurred in the past. To show anticipation and lack of novelty, it was necessary, argued Merrell Dow to show that the product had been made available and that did not invariably occur with use.
The case went to the House of Lords. It was the first patent case involving issues of substantive law to go to the Lords for around ten years. The House of Lords did decide that the law had changed, Lord Hoffmann saying;
"An invention is a piece of information. Making matter available to the public within the meaning of section 2(2) therefore requires the communication of information. The use of a product makes the invention part of the state of the art only so far as that use makes available the necessary information."
However, the direction that Terfenadine could treat hayfever was sufficient disclosure to knock out the metabolite patent. Lord Hoffmann again :
"[K]nowledge of the acid metabolite was in my view made available to the public by the terfenadine specification under the description a part of the chemical reaction in the human body produced by the ingestion of terfenadine and having an anti-histamine effect. Was this description sufficient to make the product part of the state of the art? For many purposes, obviously not. It would not enable anyone to work the invention in the form of isolating or synthesising the acid metabolite. But for the purpose of working the invention by making the acid metabolite in the body by ingesting terfenadine, I think it plainly was. It enabled the public to work the invention by making the acid metabolite in their livers. The fact that they would not have been able to describe the chemical reaction in these terms does not mean that they were not working the invention. Whether or not a person is working a product invention is an objective fact independent of what he knows or thinks about what he is doing."
Had this case not been won, life for generic companies might have been a lot more difficult: it is often not apparent what metabolite, enantiomer or polymorph creates activity, and there would have been scope for later patents covering use of all of these elements (not just their manufacture) even if prior products had already used them, and absent any special advantage.
After the Terfenadine case, John and I worked on a series of cases relating to aerosols. These covered all sorts of applications. The first was on the use of non-CFC gases for medicinal aerosols and involved us reviewing a vast body of literature relating to the effect of CFCs on the ozone layer, and the extent to which that had been known, as well as the potential effect on medicinal aerosols, at the time of the patent. We also dealt with patents on various aerosol formulations, and aerosol technology. Johns perceptiveness was remarkable. Amongst a stack of documents he would quickly identify the most relevant matter and if I, or one of the barristers handling the matter failed to take this as seriously as he thought it merited, he would continue doggedly, until we had seen his point. He was invariably correct.
Had we not managed to revoke the patent relating to use of non-CFC gases for aerosols, generic companies would effectively have been unable to market any aerosol products at all, since by that time use of CFC gases had been prohibited.
I also worked with John on the Paclitaxel case (Bristol-Myers Squibb Co v Baker Norton Pharmaceuticals Inc and Napro). That too was both legally and commercially, an important case. It established that the selection of a dosage range was not patentable. It was not really a swiss claim for a new use, although it was worded as such, rather, it was a disguised method of treatment. Of course, in some circumstances, where as a matter of fact, a dosage range is inventive or not obvious, it might possibly be patentable, but such circumstances would be rare. In this case, BMS claimed that the dosage range chosen was counter-intuitive, in that it was a range which would have been regarded as having an unwelcome side effect profile. We were able to show that with such a drug, a natural way of working was to seek to minimize side effects in other ways, and that it was obvious to try different dosage ranges.
I shall certainly miss John and it seems to me that the UK Generics industry could be quite different had it not been for his contribution to it.
Anna is an independent adviser on Intellectual Property matters to the pharmaceutical industry. She has studied in England and France and has conducted many cases for generic companies. She is a qualified mediator.
Anna McKay
January 2006
anna@annamckay.com
Tel: +44 (0)208 347 8734
www.annamckay.com
Back to top
| Contributor Peter Wittner has been in the pharmaceutical industry for 30 years. He has worked for the former Evans Medical and then Norton Pharmaceuticals (now part of IVAX) where he was responsible for European Sales & Marketing. After leaving Norton Peter set up his own consultancy in 1993 and operated independently until 1996 when he joined the Indian company Ranbaxy to set up the infrastructure of their new subsidiary. For the last 7 years he has been a consultant in the field of generics. |
|
If you would like to contact Peter to give him feedback or enquire about Interpharm Consultancy, please email him at peter@interpharm-consultancy.co.uk
Asia the next big thing in generics but at what price?
Most people in the generics industry are aware of what a force Asian companies by which I mean Indian, Chinese and lately Korean companies have become as sources of APIs and low cost formulations. Indeed, I pointed out that the scale of their increasing presence can be measured indirectly by the CPhI yardstick.
In a previous article, I noted that the number of Chinese exhibitors at CPhI had grown from 39 in 1997 to 578 in 2005. Over the same period, the Indian representation increased from 59 to 163 and the number of Koreans from 1 to 35. Altogether a significant increase in possible Asian sources of APIs and generic formulations, all offered at prices that seem to Western eyes extremely attractive, impossibly cheap and difficult to refuse.
There are signs that other Asian countries are very interested in establishing strong generic industries in their own markets. A very distinct indication this came just over a year ago in Kuala Lumpur. An Asian regional workshop on TRIPS and Access to Medicines was held on 28-30 November 2004 and was attended by policy makers and NGOs from 21 countries
In early December, the participants distributed a press release from which I have extracted a number of significant statements.
5
Data on prices of various products within and across Asian countries were presented by resource persons showing that prices of branded products are significantly (and often greatly) higher than similar generic products, and also that the presence of generics brings down the prices of branded products in the same country
. It is therefore essential that patented drugs do not enjoy monopoly and that competition from generics should be enabled, so that the patients have more choice and prices can be brought down. Many participants also called for price controls to be placed by governments on medicines since these are essential items
.
6.
.. The Doha Declaration has clarified that there are some flexibilities and safeguards including the ability of governments to implement measures such as compulsory licensing, government use/rights and parallel importing, to offset the monopoly of patents
7. Many participants asked that governments undertake a serious review process of TRIPS so
that countries are enabled to exclude patents on medicines and food
...
8. In the immediate term, governments are urged to urgently review their patent laws and amending them to
.. enable the country to provide compulsory licenses, government use orders and parallel importing in simple and effective ways.
Statements made by the Novartis CEO Thomas Ebeling when announcing the companys results for 2004 reinforce this impression. The parent of Sandoz commented that two planned areas for expansion are the rapidly expanding markets of China and India, where the company is targeting an increased presence, particularly in the generics market. He also said that Asia was the biggest growth market in the world for generics.
The question therefore is whether Western companies would be able to compete successfully in these markets given that, judging by the comments made in Kuala Lumpur, pricing will be the most critical issue. In view of the difficulties that many Western companies have competing even in their own domestic markets, the probability is that it will be local companies and manufacturers that take the major market share.
However, such a rush by local Asian countries to generate a cheap generics market in a hurry brings with it the possible risk that product quality might suffer in the process.
Counterfeiting is not a topic that pharmaceutical companies like to talk about and some prefer not to even admit that it exists. Nonetheless, it does exist and is quite prevalent in certain African countries with British Pharmaceutical Conference 2003 having held a session on the topic in 2003 on the subject.
The Sunday Times newspaper in South Africa suggested in an article that possibly as much as 20% of the medicines sold in South Africa are fakes or stolen and are almost impossible to distinguish from the real thing. According to WHO, in a press release in May 2005, counterfeits account for about $35 billion a year globally. Shigeru Omi, WHO Western Pacific director, said that the problem is very serious in Africa, where poverty and poor enforcement have created a breeding ground for bogus pharmaceuticals and also in Asia, where many fake drugs are manufactured.
Additionally, he said the problem is most serious in developing countries, including the Mekong region of Southeast Asia where studies have found that most drugs on sale to treat malaria are fakes.
What the Asian countries will need to remember is that there is no such thing as a free lunch and that the public does not just need cheap generics but also good quality generics. A failure to set up effective regulatory mechanisms at the same time as they promote use of generics could prove a very expensive saving in the long term.
If you have any comments or questions on this article, or would like to suggest a topic for an article, please feel free to contact me on peter@interpharm-consultancy.co.uk.
Peter Wittner
February 2006
www.interpharm-consultancy.co.uk
Back to top
This week's feature reports are:
Injectable Generic Drugs: Prospects and Opportunities to 2010
This 142 page information-packed report covers injectable cancer, cardiovascular, gastrointestinal, anti-infectives and biopharmaceuticals.
Generic Competition to 2009: the impact of patent expiries on sales of major drugs
Use this report to:
# Forecast the market share that will be lost to generic competition
# Understand which global companies are under pressure from generics
# Consider the effect of expiry on the patent holders competitors
# See whether there are any outstanding litigation issues
# Learn which generic companies are active in filing
Global BioGenerics Market in 2010: A Consensus Market Models report
Use this report to:
# Understand key market conditions that underpin the future of BioGeneric products
# Assess the current market research and analysis from a broad range of sources
# Learn how regulatory authorities are shaping the market
# Gain insight into which companies are working in this area
View our entire collection of generics specific research reports by clicking here.
Back to top
|
GENERICS EVENT WATCH FEBRUARY 06 - APRIL '06 |
CPhI/ICSE Japan
24-26 April, Tokyo: Business meeting place for the leading Japanese and international pharmaceutical ingredient manufacturers and traders. Expecting 15,000+ attendees and exhibitors from 30+ countries.
FCE Pharma 2006
18-20 April, Brazil: Latin America´s largest trade show for technology for the pharmaceutical industry. In 2004 more than 300 suppliers of raw material, packaging, equipment, machinery and services displayed their products.
4th Annual Generic Drug Forum
5-7 April, Washington: Topics include; Navigating the 505 (B) (2) Process for Increased Generic Drug Approval, Embracing Medicare Modernization Act and Its Implications for the Generic Drug Industry, Maximizing Product Output Through Partnerships, Co-Promotion, M&A, Laying the Groundwork for the Approval of Generic Biological Products, and Observing the Explosion of Indian Pharmaceuticals in the North American Generic Market.
Manupharma Europe
27-29 March, Montreux: The third annual ManuPharma European Summit is an exclusive gathering that brings together leading Pharma Manufacturing executives with a select and diversified group of leading Solution Providers to address vital industry issues.
Interphex 2006 USA
21-23 March, New York: INTERPHEX is the place where the industry meets annually for sourcing products and services, networking, education, and insights into the dynamic issues facing the industry today.
5th Annual Global Generic Strategy Summit
20-23 March, Barcelona: Strategy presentations from leading Generics companies.
DUPHAT
13-15 March, Dubai: One of the largest annual pharmaceutical exhibitions in the Middle East. Duphat 2005 attracted over 3000 pharmaceutical professionals from over 25 countries.
EuroPLX 27
8-9 March, Athens: Focussing on: Collaborative Agreements in Generics (incl. Bio-generics), OTC Drugs, and Nutraceuticals - Development, Licensing, Marketing and Distribution.
Global Generic Pharmaceutical Conference 2006
7-9 March, Mumbai: "The Changing Landscape" investor event. Two days of speaker presentations and simultaneous one-on-one meetings with institutional investors and other pharmaceutical companies.
PHARMtech Congress CEE 2006
22-23 February, Warsaw: The opportunity to meet senior representatives from leading vendor companies at home in Central and Eastern Europe.
If you would like us to include details of any additional events that are relevant to the generics industry in our future newsletter please email us.
|
The GenericsWeb Patent Intelligence Newsletter is available in both HTML-format (text & images) and text-format. If you have any problems with the format, please let us know by emailing: info@genericsweb.com . If this information is not relevant or of interest, please accept our apologies.
Disclaimer: Please note that all information provided in this newsletter is considered accurate at the time of writing but is not guaranteed and may change over time. Opinions expressed in this newsletter are those of the authors and are not intended to represent legal or investment advice. Information should be verified for accuracy and advice should be sought from a suitable professional before acting upon any information contained herein. |
|
GenericsWeb
|
|
GenericsWeb is a generics industry community website featuring Pipeline Patent Intelligence, careers, resources, industry research, product development and news. If you are interested in contributing to the GenericsWeb newsletter or would like to discuss promotional or partnership opportunities please feel free to contact us.
email: info@genericsweb.com
web: www.genericsweb.com
tel no.: +61 2 9818 6111
fax no.: +61 2 9818 7786
Address: PO Box 202
Balmain NSW 2041 Australia |
To unsubscribe from this newsletter list, click here: ##Unsubscribe Link##
|
CONTENT
Patents
Drug In Focus: Orlistat
Resources
Events Watch
Industry Reports
|