|
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
Drug In Focus: Quetiapine
The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic equivalents of Quetiapine, a top-selling thiazepine molecule indicated for the treatment of bipolar disorder, depression, manic episodes and schizophrenia.
General information
An analysis of the currently marketed products containing Quetiapine shows that Quetiapine is currently available as film-coated immediate-release (IR) and extended-release (XR/XL) oral tablets.
Figure 1: General Information table for Quetiapine
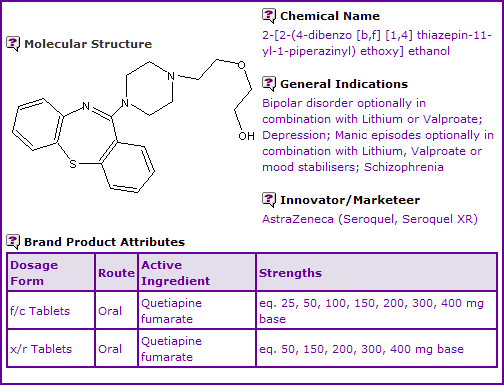
INN Constraint Comment
At the time of publication, patents within the original API family are still valid in certain countries and hence are potentially preventing the launch of generics. In several European states, the European patent in this family has had Supplementary Protection Certificates (SPCs) granted with expiries of March 2012, however it should be noted that in certain European countries, the patents claim processes for the preparation of Quetiapine and as such may be circumvented.
In Australia and the US, patent extensions have been granted for the Seroquel IR formulations extending the patent protection until March 2012 (the US patent includes a 6 month paediatric extension); however generics for the IR formulations have been identified in Australia. Whilst no generics have currently been identified in the US, the US patent mentioned above is the subject of litigation regarding several Paragraph IV filings (See Figure 4). The Canadian equivalent of the API family has expired and there has been considerable generic activity for the IR formulations in Canada. The second patent family relates to processes for the production of Quetiapine and as such is not considered to be constraining.
AstraZeneca reported a growth of 66% in global sales for the Seroquel XR formulations in the period 2009-2010, whereas sales of the IR formulations decreased by 1% over the same period. This data is significant and highlights the importance of the extended-release formulations within the marketplace, both for the innovator and any potential generic manufacturers. In terms of these Seroquel XR formulations, the third and fourth patent families are worthy of detailed consideration as they appear to protect the extended-release formulations of Quetiapine.
In the majority of territories these patents are due to expire in May 2017. In the US, equivalent patent protection is extended to November 2017 via a 6 month paediatric extension. This FDA Orange Book listed patent (US 5,948,437) is also the subject of litigation, with Paragraph IV filings and subsequent infringement proceedings having been identified concerning the Seroquel XR formulations between AstraZeneca and several generic companies (See Figure 4).
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Quetiapine
Figure 3: Patent Family View priority application US87928886A
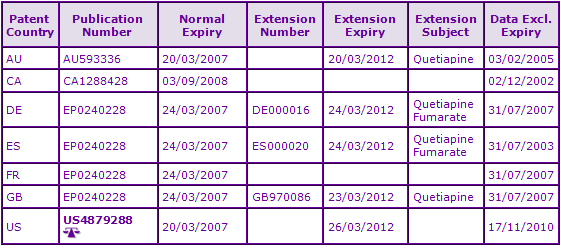
Figure 4: US Paragraph IV filings for Quetiapine Fumarate
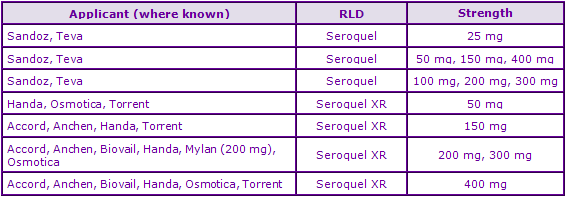
Figure 5: Marketing Authorisations for products containing Quetiapine in Key Countries
In the UK, data exclusivity expired in July 2007 and since late 2008 a great number of generic marketing authorisations have been approved, suggesting that generic competition will be fierce upon expiry of the API patent in the UK. The Canadian data highlights the presence of a large number of generics currently available. There is currently no evidence of any marketing authorisations obtained by generic companies relating to the extended-release formulations..
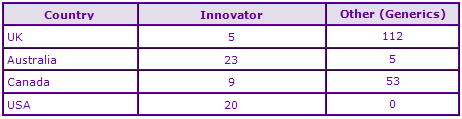
Figure 6: Top Patent Applicants
A representation of patentees who have filed the most patent families for this INN.
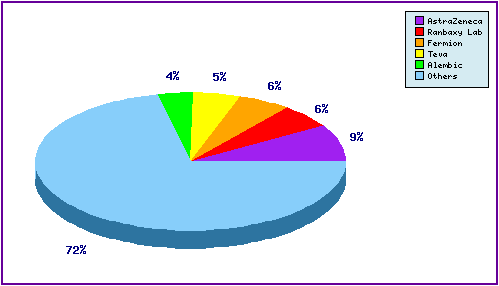
Figure 7: Patent Filing Trend
Represents the timing of the earliest priority filing date for each patent family identified for this molecule as well as the type of claims found in the applications. Note the consistent patenting by generic companies post-approval (1997 for Seroquel IR in the US) in the area of process, use and formulation, and the relative increase in formulation patent filings following approval of Seroquel XR (2007-2008).
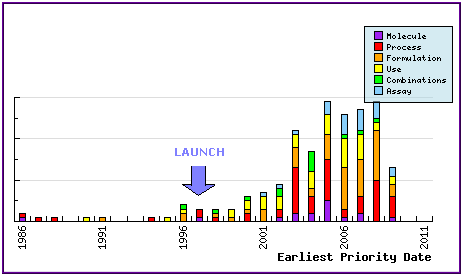
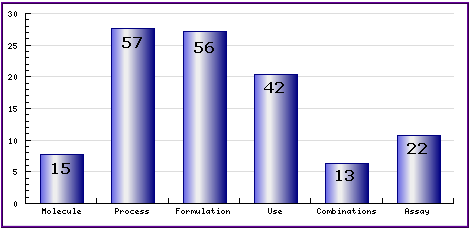
Figure 8: Patent Category Distribution
The types, number and relative distribution of patents that have been filed for Quetiapine obtained via a comprehensive patent search (Pipeline Developer report). Note that there exists a roughly equal number of process and formulation patents. Of the process patents identified, the majority relate to the complete synthesis of Quetiapine.
Such formulations may be protected in the near future however this is by no means certain, as generic extended-release formulations have been given tentative FDA approval.
In summary, where Quetiapine has not already gone generic it is likely to become available to generic companies in Europe upon expiry of any SPCs. The large number of marketing authorisation holders identified in the UK and the number of Paragraph IV filings is indicative of the critical interest shown by generic companies for Seroquel generics, not surprising considering that Seroquel and Seroquel XR generated sales of US $4.15bn and $1.15bn in 2010. Most generics or potential generics relate to the Seroquel IR formulations, however it is likely that generic parties are closely monitoring the patent landscape situation surrounding the Seroquel XR formulations. Such formulations may be protected in the near future however this is by no means certain as for example; generic extended-release formulations have been given tentative FDA approval.
Please visit www.GenericsWeb.com to find our more about Pipeline Patent Intelligence and how it can assist your generic drug development. Register to receive our monthly newsletter INNsight, and a GenericsWeb staff member will call you to discuss your needs.
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
|