|
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
Drug In Focus: Olanzapine
Welcome to the new format Drug In Focus; please give us your feedback.
The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic equivalents of Olanzapine, an Eli Lilly antipsychotic having global sales of over $5 billion in 2010.
General information
An outline of the currently marketed products containing Olanzapine shows that the Olanzapine products are available in numerous dosage forms, the injectable forms using salts of Olanzapine compared with the base compound used in the oral dosage form. An oral fixed-dose combination with Fluoxetine is also available.
Figure 1: General Information table for Olanzapine
INN Constraint Comment
The most constraining patent family preventing the launch of generics relates generally to the API - GB9009229A (25/04/1990). Patents in this family are subject to extensions which expire in Europe mostly in 2011, with some exceptions such as in Greece and Portugal where SPCs citing a later first EU MA date (for the orodispersible formulation) have been granted. In the US, the most significant patents in the family appear to expire in October 2011 after accounting for paediatric extensions to the patent term. There are many key patent families which protect physical forms used in the preparation of Olanzapine that will unlikely be a major constraint to generic competition. Patents protecting the commercial form of Olanzapine (form II) are also unlikely to stop generic competitors who seem to be using prior-disclosed form I according to US DMF and patent filing data (Figure 4). Generic injectables will seemingly need to avoid the pamoate salt until 2018/19, but the tartaric acid salt does not appear to be protected so may be a viable alternative. Generic versions of the combination product in Australia, Canada and the US may need to be cautious with indications protected by family US8644498P (22/05/1998) until 2017 (2019 in US). Data exclusivity is not likely to constrain entry of generic products for this INN.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Olanzapine

Figure 3: Patent Family View priority application GB909229A
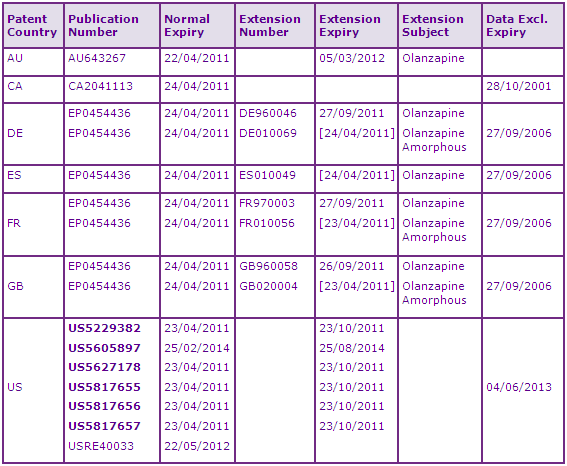
Figure 4: Companies who have established activity in developing Form I Olanzapine API:

*Other companies may also have developed form I API but this cannot be confirmed via patent or DMF data.
Marketing Authorisations pending and approved suggest that generic competition will be strong upon expiry of the API patent across all dosage forms and all major regions (Figure 5).
Figure 5: Marketing Authorisations for products containing Olanzapine in Key Countries

Figure 6: Top Patent Applicants
A representation of patentees who have filed the most patent families for this INN. Note the strong filing activity by the innovator, Eli Lilly.
Figure 7: Patent Filing Trend
Represents the timing of the earliest priority filing date for each patent family identified for this molecule as well as the type of claims found in the applications. Note the strong responsive patenting by generic companies post-launch, particularly in the area of molecular forms.
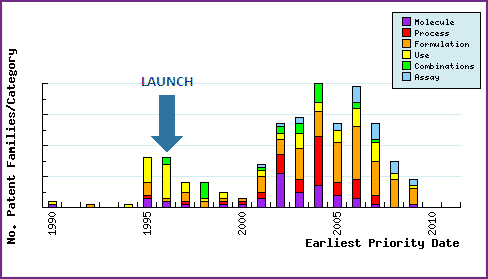
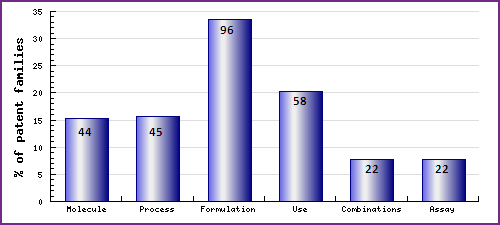
Figure 8: Patent Category Distribution
The types, number and relative distribution of patents that have been filed for Olanzapine obtained via a comprehensive patent search (Pipeline Developer report). Note that 44 patent families - or 15% of all patent families - relate to molecular forms, an unusually high proportion.
In summary, Olanzapine is likely to go generic very quickly in most major territories after key API patent expiry, despite the extensive patent-related life-cycle management activity post discovery of the API. The only areas of extended protection appear to be additional SPC terms gained from the use of later first EU MA dates as the basis of SPC applications in some countries in Europe.
Please visit www.GenericsWeb.com to find our more about Pipeline Patent Intelligence and how it can assist your generic drug development. Register to receive our monthly newsletter INNsight, and a GenericsWeb staff member will call you to discuss your needs.
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
|