Leighton Howard is a patent information expert with extensive generics experience. He has worked within the professional patent information industry and major generic pharmaceutical firms and now provides consultancy services to the generics industry and government organisations via his company XIP Pty Ltd. He is also the founder of GenericsWeb and remains instrumental in maintaining the high quality and accuracy of data contained in Pipeline Patent Intelligence. Please email any comments or queries. |
|
Drug In Focus: Valaciclovir
Valaciclovir (Valtrex) is a popular treatment of Herpes virus-related disorders with reported global sales of £700 million in 2005 and growing at 22% per annum according to the 2005 GSK Annual Report. The report also states that patent protection of the active ingredient, including applicable extensions, will not expire until 2009 in Europe and the USA.
Based on information contained in the GenericsWeb Pipeline Selector report for Valaciclovir, this months Drug In Focus analyses the patent landscape surrounding this product with a view to launching generic equivalents.
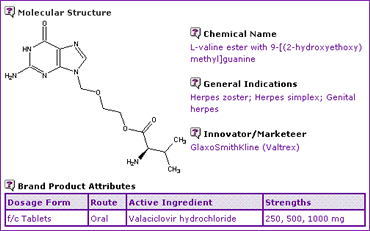
The Valaciclovir General Information (Table 1) indicates that the product is available only as oral tablets, unlike its parent compound Acyclovir, which is also available as injectable and topical preparations.
Table 1: Valaciclovir General Information
Valaciclovir Regulatory Data shows that data exclusivity provisions should not delay launch beyond molecule patent expiry in key territories, the latest key market data protection expiring in 2004 in Europe. A focus on the patent protection should therefore offer some insight into the timing and strength of Generic competition to the Valtrex product.
The Key Patent Indicator (Table 2) shows that the enamine tautomer of the Valaciclovir molecule was originally patented in 1981 for use as an immunomodulator. Patents in this family have been extended in the UK and Australia resulting in ongoing protection of the API (via its tautomerism) until 2007. However, the main protection of the Valtrex product appears to result from a later patent filing protecting the molecule specifically, and its use as an antiviral. Whilst litigation is underway in both the USA (resulting from Ranbaxys Paragraph IV filing) and Canada that may enable launch prior to this later molecule patent expiry, no such legal challenges have been identified in other key territories.
Table 2: Valaciclovir Key Patent Indicator
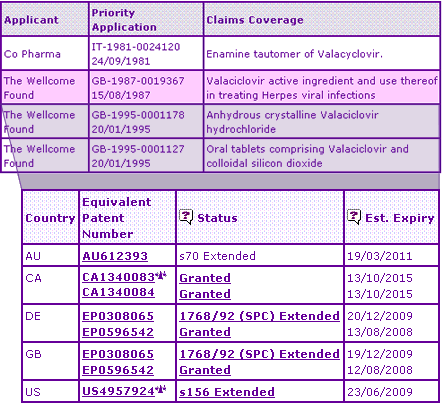
In addition to these patent families, further constraining patent families have been identified by the Key Patent Indicator, relating to the anhydrous crystalline salt of Valaciclovir, and the use of colloidal silicon dioxide in oral formulations. Although these later patents may represent a barrier to launch of generic products in major markets, it is not likely that they will prevent generic competition completely as they may be circumvented by switching salts, and by reformulating the oral tablet.
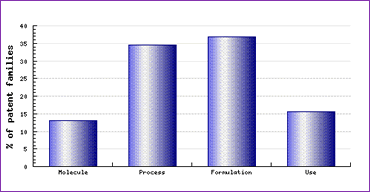
Study of the Patent Risk Analysis section, based on comprehensive patent data (details of which are accessible in the corresponding Pipeline Developer subscription) indicates that patenting activity relating to this drug is heavy in all four development areas, but particularly with respect to process and formulation patents (Figure 1). This indicates that caution is necessary with regard to employing any variation from the prior published synthetic routes and manufacturing conditions in preparing the API for generic products, as well as the substitution of excipients in developing formulations. Furthermore, lesser-prepared generic developers may also discover that the particular molecular form they intend to use in circumventing the anhydrate patent may already be protected by more proactive developers.
Figure 1: Valacivlovir Patent Category Distribution
The Patent Filing Trends graph provides greater insight into the strength of GSKs patent position resulting from its lifecycle management strategy for Valtrex. The number of patent filings prior to product launch in 1995 can be seen in the areas of molecule, process and formulation patents indicating significant attempts to protect any innovation in bringing Valtrex to the market. Greater interrogation of these data will assist in building a strategy for development of a generic Valaciclovir product.
Figure 2: Valaciclovir Patent Filing Trends
In summary, the market for Valaciclovir has been reasonably well protected by the innovators beyond the life of the active ingredient patents. Later patenting of the commercialised active ingredient and formulations will provide only limited protection as generic companies are likely to circumvent the patents. However patenting by more proactive generics companies may limit the number of products that are launched upon active ingredient patent expiry. Comprehensive data for patent families relating to Valaciclovir, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector and Developer reports are available for any active ingredient. Please visit www.GenericsWeb.com or contact us by email at info@genericsweb.com for further information. |