Leighton Howard is a patent information expert with extensive generics experience. He has worked within the professional patent information industry and major generic pharmaceutical firms and now provides consultancy services to the generics industry and government organisations via his company XIP Pty Ltd. He is also the founder of GenericsWeb and remains instrumental in maintaining the high quality and accuracy of data contained in Pipeline Patent Intelligence. Please email any comments or queries. |
|
Drug In Focus: Risperidone
Towards the end of 2007, the expiry of patents covering the antipsychotic active ingredient Risperidone (Risperdal) in some major markets, will likely result in a significant amount of generic activity. Based on information contained in the GenericsWeb Pipeline Selector report for Risperidone, this months Drug In Focus analyses the patent landscape surrounding this product with a view to launching generic equivalents.
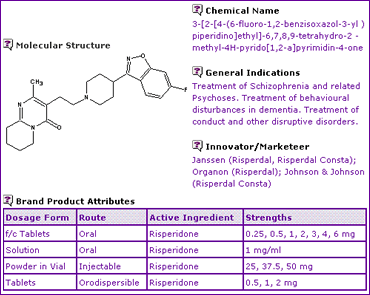
The Risperidone General Information (Table 1) indicates that this active ingredient is available in several dosage forms, including three administered via the oral route and one sustained-release injectable formulation.
Table 1: Risperidone General Information
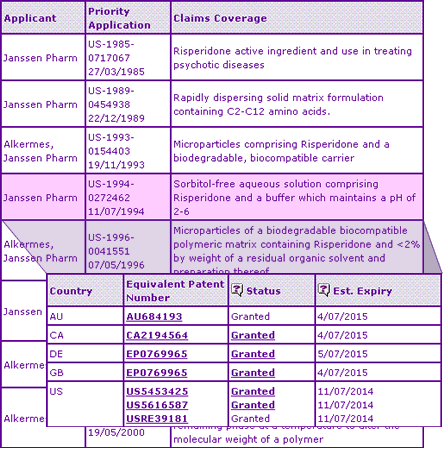
Data exclusivity periods have expired for this product in major markets, and the Key Patent Indicator (Table 2) confirms that the extensions on the Risperidone active ingredient patent expire in December 2007 in the UK, USA and Germany, whilst the Australian extension expires almost one year later due to a later TGA approval date, upon which the extension is calculated. The litigation alert now available in new Pipeline Selector reports shows that this US active ingredient patent was been upheld by a US district court.
Table 2: Risperidone Key Patent Indicator
The corresponding patent in Canada expired in July 2006, the response being shown by the new MA holders section in the Pipeline Selector report, in that 62 new authorisations were granted by the end of August. Of the ten generic competitors to launch, only two of them have launched a solution formulation, the remainder opting to launch only the tablet formulation (including Ranbaxy who has filed patents on both Orodispersible and solution formulations). There is a notable absence of any generic orodispersible or injectable dosage forms.
With the DMF feature identifying 17 submissions filed with the FDA, indications are that the US generic response is likely to be similarly ferocious in respect of the conventional tablet formulation, however further clues in regard to the additional dosage forms can be found by analysing the additional constraining patent families identified by the Key Patent Indicator.
The lack of additional protection of the conventional tablet formulation, a single family protecting each of the oral solution and orodispersible tablet formulations, and several patent families protecting the injectable dosage form explain why most generic competitors have maintained a narrow product focus in Canada.
Table 2 shows that a key patent on the oral solution expires in 2015 in most major markets, making research & development to circumvent this patent relatively unattractive, whereas broad protection of the sustained-release injectable dosage form is likely to constrain generics until at least 2014, regardless of R & D efforts. According to a Barr Laboratories press release, they have filed a Paragraph IV certification challenging the Orange Book-listed patents that protect the orodispersible formulation in the US. Depending on the outcome of the litigation, this suggests that generic versions of this particular dosage form may be available in the US earlier than the patent expiry suggests, after any applicable exclusivity to Barr has expired.
The Patent Category Distribution (Figure 1) shows the response to the innovators patent position in terms of generic competitor patent filings.
Figure 1: Risperidone Patent Category Distribution

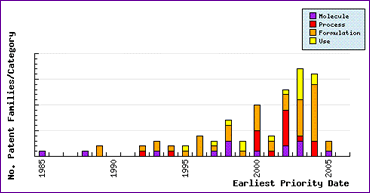
Furthermore, the Patent Filing Trends graph (Figure 2) shows that Generic competitors have experimented with new molecular forms (later filings) to attempt to alleviate problems experienced with patent restrictions on the formulations. It could also be argued that these patent filings relate to broadly claimed molecular forms which have been filed by more aggressive players to deter smaller competitor entry by creating confusion and uncertainty in the patent landscape.
Figure 2: Patent Filing Trends
Although formulation patents may represent a barrier to launch of generic Risperidone products in major markets, those companies who have skills in formulation technology and are proactive in developing alternative formulations are likely to be rewarded for their efforts in extending their product line, whilst others are likely to suffer the effects of considerable price erosion in offering only a tablet formulation.
In summary, the market for Risperidone has been well protected by the innovators beyond the life of the active ingredient patents. Later patenting of some formulations provide limited protection from generic entry, but any efforts by the innovator in switching patients to the sustained release injectable dosage form before expiry of the active ingredient patent may assist in protecting revenues post-expiry.
Comprehensive data for patent families relating to Risperidone, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
GenericsWeb Pipeline Patent Intelligence is available for any pharmaceutical product on a per-drug basis. Please visit www.GenericsWeb.com or contact us by email at info@genericsweb.com for further information.
|