As a provider of comprehensive patent intelligence to generic pharmaceutical companies in over 25 countries, GenericsWeb frequently fields questions about how our patent information products can offer advantages in the development of generic pharmaceuticals.
This article is the second part of a series, and follows on from last months article Getting The Information You Need. It is intended to offer some insight into the best ways of managing patent information within an organisation with maximum efficiency once you have obtained it.
I will firstly address the important aspect of data currency and then move on to dissemination, both of which fall under the category of Knowledge Management.
Stay In-The-Know
Once you have completed a patent search and are certain that you have all of the information you require, the very next stage should be the development and implementation of a program to maintain the accuracy of the information, often referred to as Monitoring.
There are two main reasons underpinning the importance of monitoring, the dynamic nature of patents and the Priority Blind Spot.
"This may result in offering poor advice to R&D teams or senior management, in wasting time and effort circumventing patents that have lapsed, or in missing vital opportunities to oppose weak patents to avoid the high costs of court action"
Dont Read Yesterdays News
Within 24 hours of completing a patent search, the information is likely to be out of date due to the dynamic nature of patent publication information. On a daily basis new inventions are published for the first time, additional patent applications are published for known inventions, procedural statuses change and litigation may affect the scope and/or enforceability of granted patents.
You wouldnt read out of date news, so it makes sense that you wouldnt want to refer to out of date patent information. This may result in offering poor advice to R&D teams or senior management, in wasting time and effort circumventing patents that have lapsed, or in missing vital opportunities to oppose weak patents to avoid the high costs of court action.
Table 1: Updating system for generic pharmaceutical patents
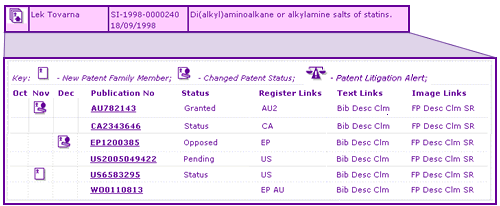
Check your Blind Spot
If I filed a new priority patent application today, it is highly unlikely that you would be able to know anything about that application until at least 18 months time. This is due to the fact that I would have one year from today to file national applications and these would be published as a matter of procedure 18 months from the priority filing date. I refer to this phenomenon as the Priority Blind Spot.
Whilst there are some possible variations to this scenario, this is predominantly the case with patents filed over the past decade or so. As an inventor with a new idea, it is unlikely that I would publicly disclose the contents of this application until I am required. Why would I do so when I could keep it a secret and spring a belated surprise on a generic competitor, thus wasting months of valuable development time and funds, setting them back to square one?
The Priority Blind Spot creates havoc for some companies who believe they are the first to innovate and later find out that they have filed patents concurrently with another applicant and do not have the rights that they originally believed. There is no way around this other than to mitigate the damage by ensuring that you discover such concurrent filings as soon as possible. The only way to do this is by monitoring your patent information.
What and How Often?
Monitoring should use a search strategy that is identical to that used for the original patent search (if not broader), but limited to identify new patent publications in the territory of interest, status changes and litigation. Ideally, new patent families should be distinguished from new publications within known families to avoid duplication of effort in determining relevance.
It is also extremely important to ensure that the monitoring strategy is updated to reflect changes in nomenclature and patent classification systems used on a regular basis (monitoring of the monitoring strategy!).
"The Priority Blind Spot creates havoc for some companies who believe they are the first to innovate"
Whilst it would seem both unreasonable and unmanageable to ensure that information is updated on a daily basis, the point I make is that regular monitoring of patent information is critical to maximising internal efficiency and ensuring ultimate project success. Anything longer than a monthly monitoring cycle is likely to significantly disadvantage a company who is developing in such an active industry as pharmaceuticals.
Reading from the Same Page
So, assuming that you now have comprehensive, relevant data that is updated regularly, it is time to ensure that you get the maximum value. This involves efficient dissemination of relevant information such that every department has a similar understanding of the issues faced and is not working against the other.
A patent information audit is possibly a good place to start. This is a series of investigative interviews with key stakeholders and process reviews to determine how patents might affect various departmental activities within a company. The result should be a definitive list of who needs to review patent information, which parts of, or changes to the information they require and how often they need to see it.
My experience suggests that providing access to patent information to too many people can result in lack of consensus on the issues faced. The number is obviously dependant on the individual company, but for most sizes of company a maximum of five key users of the information is sufficient.
"Electronic dissemination of information across geographically dispersed companies and customisation of display and report formats is obviously critical"
Once these users have been established, systems should be set up to ensure that they are all able to quickly and easily access the patent information that they are particularly interested in on a regular basis. Electronic dissemination of information across geographically dispersed companies and customisation of display and report formats is obviously critical in achieving this. The ability for users to quickly and easily drill down to the appropriate level of information to suit their requirements and understanding is a further significant benefit.
For example, you wouldnt want your API development manager having to trawl through hundreds of pages of abstracts relating to formulation patents whilst trying to remember what was contained in last months report so he could identify any new API process patents. When you consider such molecules as Atorvastatin and Pantoprazole, this scenario is entirely likely without the design and implementation of a suitable patent information management system.
Solutions for Efficient Patent Management
I have demonstrated above that obtaining the right patent information is only the first stage in understanding and responding to the issue of generic pharmaceutical patents. To really benefit from the hard work involved in generating this information, it needs to be kept up to date and disseminated in a meaningful fashion.
An ad-hoc approach to patent management in todays generic pharmaceutical industry is likely to produce ad-hoc results.
Because of our expertise working within the generic pharmaceutical industry we at GenericsWeb understands exactly what is important to our customers in meeting their patent information needs.
We have spent over 7 years developing a database system that allows effective monitoring via monthly update flags and efficiency in organisational understanding by categorisation of patent filings, fast flexible reporting formats and intuitive investigative tools.
Again, we can afford to do all of this and keep it within your budget by economies of scale. We know how much it costs to build such a complex database, and we know how difficult and time-consuming it is to ensure that the information contained in it is comprehensive and up to date. We therefore know that our Pipeline Patent Intelligence is the ultimate tool for managing patent information for generic pharmaceutical development, as it has been designed with that one goal in mind.
GenericsWeb Pipeline Patent Intelligence is available for any pharmaceutical product on a per-drug basis. Please visit www.GenericsWeb.com or contact us by email at info@genericsweb.com for further information.