| 'Drug In Focus' is written by Leighton Howard, a patent information expert with extensive generics experience. He has worked within the professional patent information industry and major generic pharmaceutical firms and now provides consultancy services to the generics industry and government organisations via his company XIP Pty Ltd. He is also the founder of GenericsWeb and remains instrumental in maintaining the high quality and accuracy of data contained in Pipeline Patent Intelligence. Please email any comments or queries. |
|
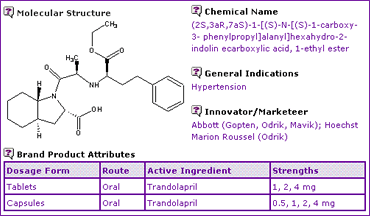
Trandolapril
In June 2004, Aventis and Abbott Laboratories reached an agreement for Abbott to acquire additional commercial rights and global manufacturing rights for the cardiovascular agent Trandolapril and the combination thereof with Verapamil. This followed on from an original co-marketing agreement between the two companies. Expiry of significant patents in major markets in 2007 would have been considered during negotiations to be a significant threat to the revenue streams generated by these products.
This months Drug In Focus analyses the patent landscape surrounding this product to determine how much of a threat generic competition really poses in 2007. This review is based on information contained in the GenericsWeb Pipeline Selector report for Trandolapril, but analysis of additional data may provide further insight.
The Trandolapril General Information (Table 1) indicates that the product is available in oral dosage forms, indicated for the treatment of hypertension and left ventricular dysfunction post-myocardial infarction.
Table 1: Trandolapril General Information
The First Marketing Authorisation Dates (Table 2) show that the Trandolapril molecule no longer benefits from data exclusivity in major territories, indicating a reliance on patents to protect its monopoly position. The Key Patent Indicator (KPI) (Table 3) for Trandolapril confirms that the extensions on the Trandolapril active ingredient patent expire in Germany and the UK in February 2007, whilst the corresponding patent in Australia expires in December 2007, due to its later approval date. It is interesting to note that, although the Australian product was approved two years later than the equivalent European product, the rules for extensions of term only allowed for an additional 10 months protection.
In the US, the molecule patent is due to expire in June 2007 based on the 17 years from grant term, and was not granted any extension of term. Additional US patents protect key intermediates in the preparation of Trandolapril and expire in April 2008. It is not thought that these will constrain generic competition completely in the US, but it may force API manufacturing to be carried out overseas until the patent expires.
Table 2: Trandolapril First Marketing Authorisation Dates
Table 3: Trandolapril Key Patent Indicator
The KPI also shows that European Patent protection of the ventricular dysfunction indication expires prior to the molecule patent extensions. However, in the US this indication will benefit from protection until April 2015. Again, it is not thought that this will significantly constrain generic competition in the US.
In addition to the above protection of the single-active product, the KPI shows that combinations of Trandolapril with Verapamil are well protected until 2012 in Europe and Australia, and until 2015 in the USA.
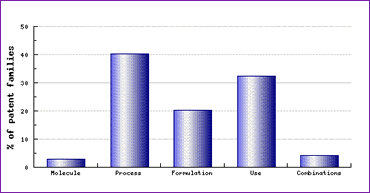
The absence of formulation and polymorph patents in the Key Patent Indicator is rare amongst the molecules currently under scrutiny by generic competitors. This signifies that developing and formulating a bioequivalent product will not be too challenging providing it is manufactured according to a non-infringing process. However, a look at the Patent Category Distribution (Figure 1) shows that a significant amount of patents have been applied for relating to preparation of this active ingredient, and formulating the final product.
Figure 1: Trandolapril Patent Category Distribution
Analysis of the Patent Filing Trends graph (Figure 2) shows that although some process patent were filed prior to 1986 (and will therefore have expired before molecule patent expiry), a significant number have been filed later in the products life cycle. This indicates that generic competitors have sought to circumvent key process patents secured by Aventis and Abbott, and have themselves sought to protect new solutions to the problem. Furthermore, similar trends are noted for formulations, although these may be a result of cross-patenting of other ACE inhibitor formulations. Launching generic products without firstly conducting a comprehensive search of the relevant patents as provided by a Pipeline Developer report is therefore very risky, and should not an exercise that is overlooked.
Figure 2: Trandolapril Patent Filing Trends
In summary, the market for Trandolapril has not been well protected by the innovators beyond the life of the active ingredient patents, and later patenting of indications, processes and formulations will provide only limited protection as the more prepared generic companies are likely to circumvent key patents. Hopefully Abbott predicted a significant erosion of revenues upon patent expiry when striking the deal with Aventis in 2004, as this appears to be extremely likely. However the profits gained by generic competitors will be restricted to those who have done their homework in this very crowded area of intellectual property protection, such that price erosion may be limited.
Comprehensive data for patent families relating to Trandolapril, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. Until the end of May 2006, GenericsWeb are pleased to offer a 10% discount on the standard price of a Pipeline Developer subscription to Trandolapril. GenericsWeb Pipeline Selector and Developer reports are available for any active ingredient upon request.
Please visit www.GenericsWeb.com to find our more about Pipeline Patent Intelligence and how it can assist your generic drug development. Register to receive our monthly newsletter INNsight, and a GenericsWeb staff member will call you to discuss your needs. |