|
Welcome to the January edition of INNsight; this month's Drug In Focus is Tamsulosin, Anna McKay discusses contracts, and Peter Wittner considers the future of the US generics industry.
If would like to receive future editions of INNsight, become a member by clicking here.
As with all aspects of GenericsWeb we love to learn from your feedback; please email us your comments about our newsletter format, article content or any other matter.
| 'Drug In Focus' is written by Leighton Howard, a patent information expert with extensive experience in the generic pharmaceutical industry. He has worked within the professional patent information industry and major generic pharmaceutical firms. He is the founder of XIP Pty Ltd, the professional patent search firm responsible for researching all patent data found in GenericsWeb Pipeline Patent Intelligence. Please email any comments or queries. |
|
Tamsulosin
Patents covering the alpha-adrenergic antagonist active ingredient Tamsulosin (Flomax) are close to expiry in European markets. Based on information contained in the GenericsWeb Pipeline Selector report for Tamsulosin, this months Drug In Focus analyses the patent landscape surrounding this product with a view to launching generic equivalents upon expiry of these patents.
The Tamsulosin General Information (Table 1) indicates that, this active ingredient is approved in the form of the hydrochloride salt in a sustained release oral dosage form for the treatment of benign prostatic hyperplasia and associated urinary disorders. Both the salt and the indications were disclosed in the active ingredient patents and should therefore fall into the public domain upon expiry.
Table 1: Tamsulosin General Information
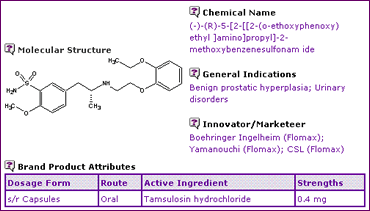
The Key Patent Indicator (Table 2) for Tamsulosin confirms that the extensions on the Tamsulosin active ingredient patent expire in Germany and the UK in early February 2006, whilst the Australian and Canadian equivalents have already expired.
Table 2: Tamsulosin Key Patent Indicator
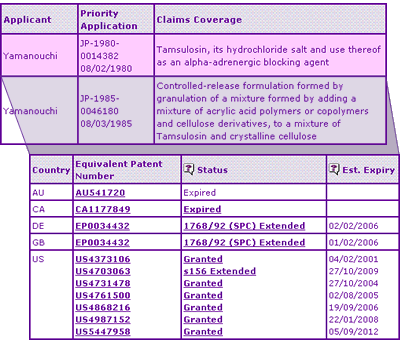
A number of US patents protect the Tamsulosin active ingredient including US 5447958, which has a normal expiration in September 2012 and relates specifically to the R(-) enantiomer present in the Flomax product. This patent is not listed in the Orange book and, providing no terminal disclaimer has been applied, will expire after the 2009 expiry date of the extended US 4,703,063. It is not certain without further investigation whether US 5,447,958 will affect generic competition beyond 2009, but this example highlights the importance in seeking further patent information beyond that disclosed in the Orange Book to identify potential risks.
In addition to the molecule patent family, one other constraining patent family has been identified by the Key Patent Indicator, relating to sustained release dosage forms of Tamsulosin. Whilst the early expiry of the US equivalent patent (relative to the extended active ingredient patent) will have little effect on the US generic market, the March 2006 and January 2010 expiry of the respective European and Canadian equivalents may act to delay generic competition in these markets. This is seemingly the case in Canada with no generic equivalents yet available despite the active ingredient patent having expired, possibly due to the broad claims issued in the formulation patent. In Europe, however, the corresponding patent is narrower and may be circumvented more easily by generic competition who are keen to secure a one-month head start by doing so and enabling launch on the active ingredient expiry date.
Figure 1: Tamsulosin Patent Category Distribution
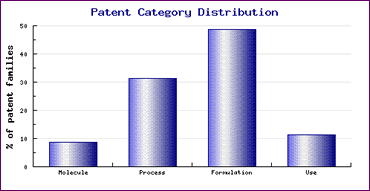
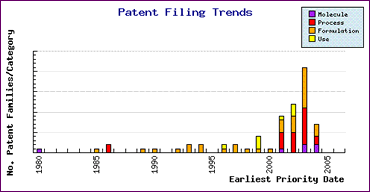
Study of the Patent Risk Analysis section, based on comprehensive patent data (details of which is accessible in the corresponding Pipeline Developer subscription) indicates that patenting activity relating to this drug is mainly concentrated on formulations (Figure 1). The timing of this activity is shown by the Patent Filing Trends graph (Figure 2) which highlights the significant increase in patent filings as generic competitors develop new processes and formulations to gain competitive advantage and sought protection in return for their efforts. This indicates that caution is necessary with regard to both process and formulation patents to ensure that all relevant patents have been considered. It is also interesting to note that the small amount of R&D effort by the innovator in 1985/86, prior to launch of Flomax, has resulted in patents which may afford them additional market protection, in some countries.
Figure 2: Tamsulosin Patent Filing Trends
In summary, certain markets for Tamsulosin have been well protected by the innovators beyond the life of the active ingredient patents by later patenting of the commercial sustained release formulation. In Europe, this will provide only limited protection as some generic companies are likely to circumvent the patents and launch in February 2006, whilst the remainder will wait an additional month to launch with technology disclosed in the expiring patent. In Canada, the broad nature of the formulation patent has served Yamanouchi well in maintaining its monopoly, and may do so until 2010. In the US, the patent filing procedure has allowed a substantial protection period that will end in 2009 at the earliest, unless legal challenges are successful.
Comprehensive data for patent families relating to Tamsulosin, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb are pleased to offer a 20% discount on the standard price of a Pipeline Developer subscription to Tamsulosin until 15th February 2006. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
Click here for more info on GenericsWeb's Pipeline Selector and Pipeline Developer patent intelligence.
Leighton Howard
Managing Director
GenericsWeb
January 2006
l.howard@genericsweb.com
Back to top
|
DEVELOPMENTS AT GENERICSWEB |
|
Generics industry country, company and market analysis now available at GenericsWeb.
| In GenericsWeb.com's new 'RMF' section you can now find valuable industry research from reputed publishers. Our generics industry dedication means that you will always find a great range of up-to-date reports relevant to your work. Payment can be made by credit card through an SSL secure system. Click here to browse the collection. |
 |
SSL security for all GenericsWeb customer data
 |
Our ordering system, customer details and all patent data are now SSL secure. Combined with our existing multi-level, company-administrator controlled, password protected login system, you can have complete confidence in our ability to protect your data. |
The following articles were popular downloads from the last issue of INNsight. Download them now if you have not already done so:
'How to Avoid Patent Infringement Needs in developing APIs and Generics' ; based on a presentation given by Leighton Howard at IPC Shanghai in September 2005; and 'Managing Patent Information needs for Generic Pharmaceutical Development; based on a presentation given by Leighton Howard at CPhI/ISCE, November 05.
| Any questions? Click here to see our FAQs.
Back to top
If you are new to GenericsWeb you may not know that there is daily generics news archived at our site:
Click here for this month's major generics news headlines
| Contributor Anna McKay specialises in IP Law and Strategy for Pharma companies. As an English solicitor, she won many leading patent cases before the Patents Court, Court of Appeal, EPO, and House of Lords for generic companies. She now works independently, advising companies worldwide on IP exploitation and strategy. She is also a qualified mediator. |
|
If you would like to contact Anna to give feedback or enquire about her services, please email anna@annamckay.com
Contracts: avoiding the pain
Happy New Year!
This month Ive decided to write a different kind of article. I am hardly going to mention pharmaceuticals. There will be nothing at all that specifically relates to generic medicines, yet I hope you will find it interesting and useful. I would like you to tell me!
This article is for those of you who are involved with the business relations of your companies, and because of its nature and length, much is left out. Please do not take it as comprehensive legal advice.
I want to write about contracts, and how to agree and record them. You may think that contracts are for lawyers, but actually, all of us deal with them in employment, in buying and selling, agreeing practices with other companies, conducting our business. All of that involves contracts.
Some companies have a policy that all contracts should involve lawyers and I dont think any succeed. Almost inevitably, even if initially arrangements are formally recorded, variations occur, are welcomed or condoned, often without formal amendment to the document. In other companies executives have a policy of trying their hardest to avoid lawyers and formal documentation whenever they can, perhaps relying on relationships to make things work. Legal documents and good relationships should not be a contradiction, and I would like to offer some suggestions as to how to narrow the gap.
I am prompted to write this article because I have just been, dealing with YET ANOTHER badly drafted contract. In this case, it was a distributorship agreement which looked, on its face, quite well written. It was a perfectly valid contract, however, it failed to cover certain matters which were absolutely essential to the distributor. The manufacturer wasnt happy with it either it was saddled with a deal which locked it into a price and duration which did not enable it to recover tooling costs. By the time I got involved the executives concerned were spitting at each other. The distributor had to re-negotiate, and achieve more flexibility, at costs which were 25% higher than those originally agreed, and for a duration which was several times longer. This provided it with a viable product, from which both parties could make money and avoided a situation which seemed likely to result in the receivership of one party, and substantial irrecoverable costs on the part of the other. Much of the pain, stress and lost sales could have been saved if the parties had done a good job of agreeing the contract in the first place.
With pharmaceuticals, the available market is usually big enough to make both parties happy. There is no point in entering into a contract unless the product or service which you are buying or selling is going to achieve a benefit for both parties.
Formation of Contracts
A contract is created when there is an agreement between two parties. Under English law, there has to be an offer and acceptance. All major terms have to be agreed. Minor terms may be implied by a course of conduct, what is normal in the business and others (e.g. that products are of merchantable quality, fit for the purpose for which they are sold) may be implied by law. If fundamental terms are not agreed (price, identity of goods, that kind of thing) then there will be no contract.
Binding contracts can be oral. Under English law, only contracts relating to land must be in writing. It is a common misconception that contracts have to be written in order to be binding. This is not so, but of course it is much easier to clearly identify the terms of a contract which has been recorded in writing. E-mails and other informal messages can be useful evidence as to the terms of a contract which has not been formally recorded.
In common law countries (England, USA), there is a great deal of importance on the literal wording of the contract. Civil law, which applies throughout Europe, and in many other countries, considers matters from a more philosophical viewpoint. Civil lawyers look at what was intended, rather than being limited by what was said. There are many advantages in this approach, and it can be less predictable give less commercial certainty. Contracts made under civil law can be much shorter than those which are governed by common law.
What should you do in order to create a good contract?
Decide what do you want
Work out what you want, and ask for it. You may get it much more often than you expect. (A philosophy for life, as well a piece of legal advice!) Often, this is where contracts fall down. The matter may not be thought through properly, or one party may not ask for everything he wants, perhaps because he thinks he is more likely to get it if he asks later, or that it will be easier to agree a particular aspect of the transaction without lawyers. Possibly, but if you adopt this course of action, do it with your eyes open, knowing that you may not be able to agree what you want later on. Make sure that what you have agreed is comprehensive in its own right so that, as it stands, you have a contract that can be performed.
Simplicity and Clarity
When you have agreed a position with the other party, express it simply and clearly. You do not have to use legal language. Indeed, in my opinion, its better if you dont. No Latin! What is needed is a clear document which those who are involved with the matter day to day can understand and use. A document which has to be taken to lawyers for a translation will be a source of expense and stress. The shorter the better, in my view. If you know that something is implied by the law which is to govern the contract, then you dont need to put it down in writing. If you are not sure, include it.
Governing Law, Disputes, and Choice of Forum
In contracts made between parties from different countries, it is very important to include a governing law clause specifying the law which applies to the contract. You may choose a legal system in the jurisdiction of one of the parties, or pick an unrelated jurisdiction. To the extent that you can, work out how you will solve problems at the time you write the contract. That will make any dispute far quicker and easier to resolve, and much less expensive. Different kinds of problems may arise within a contractual relationship, and if you can provide a mechanism for sorting these out without litigating, you may save much pain and expense. So for example, you can provide that disputes as to quality are referred, in the first instance, to a third party who you both trust, that financial queries are settled by an agreed accountant, and so on.
What courts, if any, will you use in case of dispute? Do you want courts that are quick, is it important that the courts have a knowledge of pharmaceuticals? Pick appropriately. Different legal systems have different strengths. English courts, for example, tend to be quick, skilled and reliable. They are highly regarded and may be persuasive in other jurisdictions. However, determining disputes before English courts is an expensive matter. Civil courts tend to be less predictable (because of the different manner of interpretation) may be slower, and in some cases, are less inclined to grant injunctions. However, there are many variations.
You may want your matter determined by arbitration, particularly if you want to keep facts confidential. Some forms of arbitration, for example ICC application, can be slow and expensive.
There are many different fora. Here in London we have a specialised arbitration court. Provided you are specific enough, and have arbitrators who consent, you can be very flexible as to how you run an arbitration.
You may decide that you want to mediate disputes. This can have many advantages and costs are certainly one of them. If parties need to think out of the box, look at flexible options; mediation can be a very good solution. The amount of preparation, and hence cost, which is necessary for mediation is likely to be far less than that required for court proceedings or arbitration parties may be willing to agree matters which a court would require to be proved.
Mediation is particularly useful where a dispute involves a breakdown in communication, or emotional issues. Dont think that commercial disputes do not involve emotions! They do, and generally, there is no room for this aspect of the parties conflict in court. Furthermore, dealing with emotional issues in court is a very expensive business. The kind of emotional issues which you may meet in a dispute involving pharmaceutical products may include, for example, the pride/arrogance of an inventor or proprietor of a patent, the refusal of a management team to admit its mistakes, an entrenched culture of positive thinking which is so strong that it flies in the face of logic, the need of one management team to assert its difference from previous management, the need of a party to show that it is meaner and more aggressive than the other. I have met all of these in the course of litigation for generic companies against innovators. I have seen disputes which have been fought at vast cost, and never should have been, and disputes which have been settled at the court door when settlement should have occurred months earlier. There are circumstances where acknowledging emotional issues can lead to a much faster resolution.
Often, originators do not settle with generic pharmaceutical companies, even when their legal case is poor, for one very good reason : profit. Stopping a generic product from coming onto to the market, even by a few weeks, may mean a great deal of money, and be commercially sensible even when a patent is untenable.
In discussing contracts, I have picked a few issues, understanding of which I think would lead to less expensive, easier, commercial relationships. Complex pharmaceutical contracts are likely to involve both commercial personnel and lawyers and simple contracts may be agreed without lawyers in both cases I believe that a greater understanding of the risks and benefits by all concerned would be useful. Let me know what you think, and do contact me if you want advice on your contracts!
PS. I will be speaking at the EGA Conference on the 1st February about injunctions. If you have anything that you think would be useful for me to include in that talk, do let me know.
Anna is an independent adviser on Intellectual Property matters to the pharmaceutical industry. She has studied in England and France and has conducted many cases for generic companies. She is a qualified mediator.
Anna McKay
January 2006
anna@annamckay.com
Tel: +44 (0)208 347 8734
www.annamckay.com
Back to top
| Contributor Peter Wittner has been in the pharmaceutical industry for 30 years. He has worked for the former Evans Medical and then Norton Pharmaceuticals (now part of IVAX) where he was responsible for European Sales & Marketing. After leaving Norton Peter set up his own consultancy in 1993 and operated independently until 1996 when he joined the Indian company Ranbaxy to set up the infrastructure of their new subsidiary. For the last 7 years he has been a consultant in the field of generics. |
|
If you would like to contact Peter to give him feedback or enquire about Interpharm Consultancy, please email him at peter@interpharm-consultancy.co.uk
A shaky future for US companies?
In earlier articles, I have mentioned the wave of consolidation that has been sweeping over the world generics industry in the last few years.
Companies such as Teva and Sandoz have made major acquisitions around the world, as have several Indian companies and just recently the Icelandic company Actavis when it acquired the generics business of Alpharma. Everybody, it seems, is pursuing a strategy of growth through overseas acquisitions except the US companies.
Looking at the US market specifically as it was at the end of 2004, the market leader Teva is Israeli. The second placed company Watson as well as Mylan and Barr are indeed all American domestic companies but where are they on the world generic stage?
Go a little further down the ladder of leading generic companies in the US market and names like Sandoz (Swiss owned), IVAX (now Israeli owned), Alpharma (Icelandic owned) and Merck (German) appear. There are indeed still many US owned companies in the market but the overseas presence is very significant and, of course, many more companies are setting the sights on a US market entry.
Foreign ownership of the leading generic companies in any country is nothing unusual as the growing internationalisation of the generic industry means that most of the top ten companies in countries such as UK, Netherlands for example are foreign owned. This form of consolidation is what has brought about the emergence of generic superheroes such as Sandoz and Teva and is in effect a version of the Darwinian law of the survival of the fittest.
A major driving force behind this consolidation has been the need for rapid growth combined with the opportunity to make a relatively easy (if costly) entry into overseas markets. There are other factors that have driven the wave that have been discussed in detail in earlier articles but let us concentrate on these two particular factors and examine their relevance to the US situation.
Interestingly, it was the US generic companies themselves that were the first to adopt the strategy of expansion through acquisition in the early 1990s as the US generic industry really started to move. The provisions of the Hatch-Waxman Act of 1984 stimulated its initial growth during the 80s, but by the early 90s, organic growth was not enough and so the bigger companies started to buy up the smaller companies. Mylan, Watson and Barr led the way but their focus was entirely on the domestic market with targets such as Bertek, Circa, Duramed, Hickam, Makoff, Royce, Somerset, Penederm and Schein being swallowed up.
The only exception to the US-focussed acquisition trail was IVAX, which did make acquisitions in Europe and South America, although another exception joined it recently when Perrigo bought the Israeli generic company AGIS.
Apart from this, the leading US domestic generic companies have used acquisition to fuel domestic rather than international expansion. This must raise the question of whether they have thereby left themselves rather vulnerable and so liable to succumb to market and competitive pressures.
As things stand, these companies are now completely dependent on one market the US generic market. Since the market was valued by IMS at around US$ 18 billion this is perhaps nothing to worry about. However, the market is also proving increasingly attractive to all sorts of overseas, and thus lower-cost, manufacturers. The numbers of these competitors will inevitably rise and as they compete more vigorously on price, how will the domestic companies cope?
In addition, it is no secret that voices are being raised in the US at both public and governmental levels, asking why US drug prices are so high and calling for them to be controlled. So far, Big Pharma has managed to fight off these calls for lower prices but eventually they must succumb to the same sort of controls that have been implemented across Europe and elsewhere. When this happens, there will inevitably be knock-on consequences for the generic industry.
If the prices of the big brands fall, so will the prices at which generics can launch, thus further eroding the profitability of the domestic manufacturers. And what if other European cost control methods such as reference prices and generic substitution are introduced thereby further pushing down generic prices?
All these factors seem to suggest that the remaining independent US generic manufacturers will find the going harder and harder in the coming years. This must inevitably call into question their continuing independence.
Tevas acquisition of IVAX has demonstrated quite clearly that size is no defence against being acquired. So it is now worth speculating about how long Mylan, Watson, Barr and other smaller US companies can remain independent and who will eventually be the suitor that seeks their hand in marriage
If you have any comments or questions on this article, or would like to suggest a topic for an article, please feel free to contact me on peter@interpharm-consultancy.co.uk.
Peter Wittner
January 2006
www.interpharm-consultancy.co.uk
Back to top
|
GENERICS EVENT WATCH JANUARY '06 - FEBRUARY '06 |
Generics 2006
12-13 January, Barcelona: Topics include: Challenges and Opportunities in Spanish Generic Market, New Data Exclusivity for Generics, Tactics Regarding Portfolio Selection - Global Generic Company Experience, Tactics Regarding Portfolio Selection Small/Middle Size Company Experience, and Role of Corporate Brand and Individual Brands in Generics.
Pak Pharma
17-19 January, Karachi: Serves as a source of providing business opportunities to the various stakeholders in the Pharmaceutical Industry of Pakistan.
2nd International Forum on Business Development in API and GMP Fine Chemicals
23-24 January, Rome: Developments, trends and case studies.
Biotech and Pharmaceutical Patenting
30-31 January, Paris: This 2 day conference is specifically designed to provide you with a practical and intensive review of the very latest developments in patent law and patent office practice, the important business considerations that must underlie your biotech and pharmaceutical patenting strategies, and the current tactics that are proving successful for the leading players in biotechnology today.
5th EGA Annual Regulatory Affairs Conference 2006
1-3 February, London: Incorporating EGA's 2nd Legal Affairs Forum.
Brand Defense and Lifecycle Management for Mature Drugs
9-10 February, Philadelphia: Protect Brand Revenue through Defensive Patenting, Franchising, Reformulation and Market Segmentation.
PHARMtech Congress CEE 2006
22-23 February, Warsaw: The opportunity to meet senior representatives from leading vendor companies at home in Central and Eastern Europe.
If you would like us to include details of any additional events that are relevant to the generics industry in our future newsletter please email us.
Back to top
|
In GenericsWeb.com's new 'RMF' section you can now find valuable industry research from reputed publishers. Our generics industry dedication means that you will always find a great range of up-to-date reports relevant to your work. Payment can be made by credit card through an SSL secure system. |
 |
Back to top
|
The GenericsWeb Patent Intelligence Newsletter is available in both HTML-format (text & images) and text-format. If you have any problems with the format, please let us know by emailing: info@genericsweb.com . If this information is not relevant or of interest, please accept our apologies.
Disclaimer: Please note that all information provided in this newsletter is considered accurate at the time of writing but is not guaranteed and may change over time. Opinions expressed in this newsletter are those of the authors and are not intended to represent legal or investment advice. Information should be verified for accuracy and advice should be sought from a suitable professional before acting upon any information contained herein. |
|
GenericsWeb
|
|
GenericsWeb is a division of XIP Pty Ltd responsible for provision of information products dedicated to the generic pharmaceutical industry. If you are interested in contributing to the GenericsWeb newsletter or would like to discuss promotional or partnership opportunities please feel free to contact us.
Email: info@genericsweb.com Web: www.genericsweb.com
Tel no.: +61 2 9818 6111
Fax no.: +61 2 9818 7786
Address: PO Box 202
Balmain NSW 2041 Australia |
|
CONTENT
Patents
Drug In Focus: Tamsulosin
Resources
Events Watch
Industry Reports
|