|
Welcome to the December edition of INNsight; this month's Drug In Focus is Lansoprazole, Anna McKay discusses new European data exclusivity provisions, and Peter Wittner analyses why he got sore feet at the CPhI Exhibition.
If you were forwarded this email and would like to receive future editions of INNsight, become a member by clicking here.
As with all aspects of GenericsWeb we love to learn from your feedback; please email us your comments about our newsletter format, article content or any other matter.
| 'Drug In Focus' is written by Leighton Howard, a patent information expert with extensive experience in the generic pharmaceutical industry. He has worked within the professional patent information industry and major generic pharmaceutical firms. He is the founder of XIP Pty Ltd, the professional patent search firm responsible for researching all patent data found in GenericsWeb Pipeline Patent Intelligence. Please email any comments or queries. |
|
Lansoprazole
Patents covering the anti-ulcer active ingredient Lansoprazole (Zoton, Prevacid) expire in some major European markets this month, which should result in some generic competition. Based on information contained in the GenericsWeb Pipeline Selector report for Lansoprazole, this months Drug In Focus analyses the patent landscape surrounding this product with a view to launching generic equivalents.
The Lansoprazole General Information (Table 1) indicates that the active ingredient is available generally in several oral dosage forms, all of them employing an enteric-coating to prevent release of the active ingredient until it reaches the intestine. An intravenous dosage form is also available in the US.
Table 1: Lansoprazole General Information
The Key Patent Indicator (Table 2) for Lansoprazole confirms that the SPC extensions on the Lansoprazole active ingredient patent expire in Germany and the UK in early December 2005, whilst the Australian and US equivalent patents have been extended until 2009. Disparity between patent expiry dates across these countries is due to the later marketing approval dates in the US and Australia, which are used to calculate patent term extensions, whereas a later expiry date of June 2006 in Canada is due solely to the manner in which standard patent terms are calculated.
Table 2: Lansoprazole Key Patent Indicator
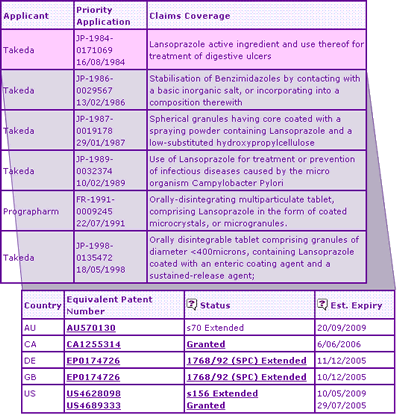
In addition to the molecule patent family, five other constraining patent families are identified in the Key Patent Indicator, relating to stabilisation of the active ingredient, preparation of granules, orally disintegrating formulations and use for treatment of Campylobacter infections. The most significant patent is likely to be that relating to stabilisation of the active ingredient by using a basic salt of calcium or magnesium (magnesium carbonate is used in the innovator product), as a stable active ingredient is clearly a requirement for a generic product. These patents expire in 2007 in Europe after opposition by Hässle was withdrawn, and offer protection until 2008 in the US.
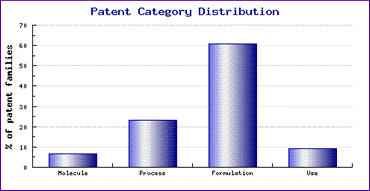
Although these patents may represent a barrier to launch of generic oral Lansoprazole dosage forms in major markets, it is not likely that they will prevent generic competition completely as our Patent Risk Analysis section, based on comprehensive patent data (details of which is accessible in the corresponding Pipeline Developer subscription) indicates that patenting activity relating to formulations containing this active ingredient is extremely high (Figure 1). Whilst this means that caution is necessary with regard to developing non-infringing formulations, it also suggests that such strong development by generic competitors will likely result in at least a handful of non-infringing generic competitors.
Figure 1: Lansoprazole Patent Category Distribution
However, a further patent family broadly claims the use of coated microcrystals or microgranules in tablets that disintegrate in less than 60 seconds. The European patent has survived strong opposition that initially resulted in revocation, but the Board of Appeal later cancelled this decision. Due to the acid-labile nature of the active ingredient these patents appear to protect the orodispersible dosage form from generic competition until 2012 in many major territories, but more proactive generic competitors will no doubt seek to circumvent them.
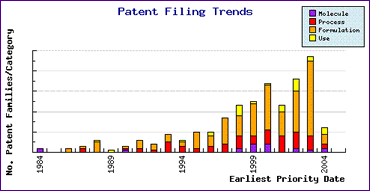
This highly protective situation has arisen by proactive life cycle management by Takeda, as demonstrated by the Patent Filing Trends graph (Figure 2). This shows a great deal of patenting activity prior to launch, particularly with respect to formulations. Although this will not extend their monopoly on the active ingredient completely, it is likely to reduce the amount of generic competition Takeda will face and should therefore reduce related price erosion. The ensuing post-launch rally of patenting by generic companies demonstrates the magnitude of problems faced in attempting to launch on day one after API expiry, and highlights the need to address patent issues early in the generic development cycle.
Figure 2: Lansoprazole Patent Filing Trends
In summary, the market for Lansoprazole has been well protected by the innovators beyond the life of the active ingredient patents, given that this type of compound had been known for some time (e.g. Omeprazole). Later patenting of formulations will provide limited protection as a few generic companies are likely to circumvent the key patents with their own innovative solutions, but orodispersible dosage forms may be protected for extended periods if patents remain in force.
Comprehensive data for patent families relating to Lansoprazole, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. During the month of December, GenericsWeb are offering a 20% discount on the standard price of a Pipeline Developer subscription to Lansoprazole. GenericsWeb Pipeline Selector and Developer reports are available for any active ingredient upon request.
Click here for more info on GenericsWeb's Pipeline Selector and Pipeline Developer patent intelligence.
Leighton Howard
Managing Director
GenericsWeb
December 2005
l.howard@genericsweb.com
Back to top
|
DEVELOPMENTS AT GENERICSWEB |
|
New article: How to Avoid Patent Infringement in Developing APIs and Generics
This article is based on a presentation given by Leighton Howard at IPC Shanghai in September 2005. Click here to download the article in PDF.
New presentation: Managing Patent Information needs for Generic Pharmaceutical Development
Based on a presentation given by Leighton Howard at CPhI/ISCE, November 05. Click here to download the article in powerpoint.
Examine legal statuses at the click of a button
Saving hours of legwork and allowing ease of analysis, a new feature to our Pipeline Patent Intelligence enables our clients to instantly view the legal status of equivalent patents all over the world. Click here to read more.
One-click access to patent claims, descriptions, bibliographic details and more
You can now click through to the various sections of patent documents by clicking on the 'text' and 'image' links provided in Pipeline Developer reporting. Click here to read more.
Litigation alerts keep you in-the-know
Litigation alerts will appear on all new Pipeline Developer subscriptions, allowing you to keep up to date with the latest cases. Click here to read more.
Extended patent family equivalents allows quick analysis of global patent coverage
For our Pipeline Developer subscribers, to quickly ascertain the patent situation around the world, extended patent family equivalents are now displayed in the 'bibliographic details' screen of each patent family. Click here to read more.
Thank you for visiting us at CPhI/ICSE
Our third year of exhibiting at CPhI/ICSE was a tremendous success. The stand was constantly busy and it was great to meet some of our many new clients. If you would like a demonstration of our patent intelligence but were too busy at the event or did not attend, please email us to schedule a time. |
Any questions? Click here to see our FAQs.
Back to top
If you are new to GenericsWeb you may not know that there is daily generics news archived at our site:
Click here for this month's major generics news headlines
| Contributor Anna McKay specialises in IP Law and Strategy for Pharma companies. As an English solicitor, she won many leading patent cases before the Patents Court, Court of Appeal, EPO, and House of Lords for generic companies. She now works independently, advising companies worldwide on IP exploitation and strategy. |
|
If you would like to contact Anna to give feedback or enquire about her services, please email anna@annamckay.com
Directive 2004/27/EC
New European Data Exclusivity Provisions
New legislation relating to the registration of generic products came into force throughout Europe on 30th October. Directive 2004/27 introduces important amendments to the Community Code, Directive 2001/83. It introduces new time periods for data exclusivity, introduces a new definition of a generic product, and contains Bolar provisions. In this article, I have concentrated on summarising changes which are relevant to generic companies, in particular, those relating to data exclusivity. A longer article can be found on my website.
Provisions for data exclusivity and abridged registration procedures were introduced at a time when the EEC was expanding, and having to cope with different cultures relating to regulation and Intellectual Property. The legislation adopted in 1987 allowed countries to choose the period of protection they afforded data protection six or ten years, and thus offered some accommodation for different attitudes towards the protection of innovators data. Of course, it resulted in regimes applying in different countries.
The 2004 Directive has harmonised time periods for registration, and that is undoubtedly its main achievement. No one can take advantage of that until 2013 it only applies in relation to reference products which are authorised after 30th October 2005.
Period of Data Exclusivity
Article 204/27 states, briefly, that:
An applicant is not required to provide the results of pre-clinical tests and clinical trials if the product is a generic and the reference product has been authorised for at least eight years in a Member State.
A generic cannot be placed on the market until ten years from initial authorisation of the reference product.
The ten year period is extended to eleven years if the original holder of the marketing authorisation obtains a new therapeutic indication which brings a significant clinical benefit in comparison with existing therapies.
If a company wants to apply for a generic product within the first eight years, it must do its own safety and toxicology work, and its own clinical trials (if it can). This is not always possible. There may be ethical reasons against carrying out trials, and of course, costs may be prohibitive for a generic product. After eight years, the regulatory authorities can cross-refer to the innovators data on safety and toxicology, and the generic company must wait another two years before it can actually market the product. In other words, the innovator has at least ten years of market protection even if his patent has passed into the public domain.
If an innovator obtains an authorisation for one or more new therapeutic indications which are held to bring a significant clinical benefit in comparison to existing therapies, he is entitled to another year of protection, taking the total to eleven years. It seems that that extra year applies to all indications, including the indications which were initially permitted. The MHRA has said that in its view significant clinical benefits require that no product containing the same active substances has previously been authorised in the relevant indication and/or extended to new categories of patients. I am sure there will be argument here! It seems that if a product is used off label then formal application later made for that indication, the innovator should not be entitled to the additional one year, but my guess is that innovators will want that extra year in such a case. We shall have to wait and see.
Essentially Similar cf Generic
Before 2004/27 the abridged procedure was available for essentially similar products. In 2004/27, instead of descriptions of essential similarity, the abridged procedure is stated to be available for a generic of a reference medicinal product
. A generic medicinal product is defined as a product which has the same qualitative and quantitative composition in active substances and the same pharmaceutical form as the reference medicinal product, that is bio-equivalent with the reference medicinal product and has been demonstrated by appropriate bioavailability studies. The different salts, esters, ethers, isomers, mixtures of isomers, complexes or derivatives of an active substance should be considered to be the same active substance, unless it differs significantly in properties with regard to safety and/or efficacy. In such cases, additional information providing proof of the safety and/or efficacy
must be supplied by the applicant. The various immediate-release oral pharmaceutical forms shall be considered to be one and the same pharmaceutical form. Bioavailability studies need not be required if the applicant can demonstrate that the generic medicinal product meets the relevant criteria as defined in the appropriate detailed guidelines.
The definition of a generic product takes into account much of the past ECJ case law. It is on all fours with the Generics, and Novartis cases. However, it does not include all dosage forms of the product and in that respect differs from the Generics case, in which the ECJ decided that An [essentially similar product] may be authorised under the abridged procedure
for all dosage forms
authorised for that product.
Hybrid Abridged Procedures
Article 10.3 of the directive states:
"In cases where the medicinal product does not fall within the definition of a generic medicinal product
or where bio-equivalence can not be demonstrated through bioavailability studies, or in case of change of the active substance(s), therapeutic indications, strengths of pharmaceutical form or route of administration, vis a vis the reference medicinal product, the results of the appropriate pre-clinical tests or clinical trial must be provided. "
Does it matter that products that have a different indication, strength, form or route of administration have to be dealt with under this section, rather than being treated as a generic? It is certainly different. Under the earlier legislation, products incorporating these differences might have been regarded as essentially similar, rather than being considered under the Proviso., European legislation is to be construed purposively, and the purpose of the Directive is to avoid repetitive tests without overriding cause and paragraph 3 requires appropriate pre-clinical tests or clinical trials. In some cases, results will simply not be appropriate, and it should not matter whether they are regarded as inappropriate because the product is a generic, essentially similar, or whether it is covered under this section.
At the same time, it does not fit with the words of the Advocate General in APS, when he said There are good reasons against requiring an application of a product which was essentially similar to a variant of a reference product to proceed under the Proviso. The Proviso operates under circumstances where bridging data are required because of a difference between the new product and the earlier products, or products to whose data reference is made. Where Product C claims essential similarity to Product B, which is a variant of Product A, no additional data were required. There was therefore no need to proceed under the Proviso.
Reference Product
2004/27 introduces provision for a European reference product. A generic manufacturer can now apply for a marketing authorisation in any Member State and rely on the dossier of information already submitted from the European Reference Product in another Member State. The other Member State must supply all the relevant documentation requested.
Up to now, some regulatory authorities, including the UK, would not supply confidential information on UK reference products: Now, it is obliged to support generic abridged applications in other Member States. There may be more extensive information possessed by the authorities in one country than in the country from which the application is made and a generic company could apply for registration on the basis of the reference product in the country with more extensive information, perhaps enabling it to market a product with different dosages, and broader indications than the innovators own product in that country. Essentially, it seems to me that generic companies ought to be able to choose to refer to the most attractive marketing authorisations for their purposes, and to have their registrations reflect the scope of such authorisations.
Bolar Provision
Long after the US adopted its Bolar Provision allowing a generic pharmaceutical company to conduct tests on a patentable compound before patent expiry, Directive 2004/27 provides for something similar. Until now, different countries have had different provisions as to the kind of work which might be carried out during patent term. Patent laws contained an experimental use exemption, but different countries have different views as to what is permitted under this exemption, and in practice, many companies carried out their clinical trials outside the EC. New Article 10.6 states :
Conducting the necessary studies and trials
and the consequence of practical requirements should not be regarded as contrary to patent rights or to SPCs for medicinal products.
National patent laws will have to be amended to take account of 2004/27, and there is no definitive list of the kind of studies and trials or consequential practical requirements which fall within exemption. There is a risk that each country will interpret this differently. The UK, and several other countries, has decided to adopt the wording of the Directive as it stands into Patent Law. In the UK, Section 60(5) of the Patents Act 1977 is to be amended so that the Bolar Exemption is added to the list of exemptions. The wording of the statute would thus accurately reflect the Directive and it would be up to case law to determine exactly what this means.
The UK Patent Office has stated that in its view, the exemption should be interpreted to cover only abridged and hybrid abridged applications for marketing authorisations in the EEU and these include:
The manufacture and importation of active substances in sufficient quantities to conduct trials and validate the manufacturing process in accordance with regulatory requirements.
The development of the final pharmaceutical form of the actual substance.
The conduct of pre-clinical tests, clinical and bioavailability trials and stability studies.
The manufacture and supply to the regulatory authorities of samples of active substances, precursors, intermediates, impurities and finished product samples.
The compilation and submission of a marketing authorisation application or variation application.
The UK Patent Office believes that activities undertaken for purposes other than applying for the marketing authorisation will not be exempted, e.g. tests to see whether the generic company can manufacture to suitable quality. It is not clear whether the subjective intention of the manufacturer is to be taken into account. If a company intends to apply for a marketing authorisation, but changes its mind, will its activities infringe patents or not? The MHRA believes that the Bolar Provision encompasses aborted development.
Where can we expect conflict in future cases?
Hybrid Abridged Procedure Article 10.3.
I think that there will be arguments as to whether all applicants under the Hybrid Abridged Procedure must supply data, or whether they must supply data only if it is appropriate to do so, and if it is appropriate to supply data, exactly what data is appropriate. There will be special difficulties where bioequivalence can not be demonstrated.
Reference Product
Current periods of data protection and market exclusivity are not harmonised. Where an abridged application is made in one Member State, and relies on a data belonging to another, what period applies? Is it the period of protection in the State for which the application is made, or the Reference State? There is no Europe wide agreement. The MHRA believes that the period of protection applied by the Authorising State is relevant, not that of the Reference State. This seems sensible to me: any contrary interpretation would undermine the ability of the Member State to choose the period of protection. But it is not clear.
Bolar Provision
Exactly what is permitted? Are subjective intentions to be taken into account? Does the provision apply solely to generic products, or also to those covered by Article 10(3), which replaces the proviso?
Additional one year period of protection
What constitutes a significant clinical benefit?
I may write more when there are further developments! And I would love to hear from you if you have matters to discuss.
December 1st 2005
Anna McKay
www.annamckay.com
Back to top
| Contributor Peter Wittner has been in the pharmaceutical industry for 30 years. He has worked for the former Evans Medical and then Norton Pharmaceuticals (now part of IVAX) where he was responsible for European Sales & Marketing. After leaving Norton Peter set up his own consultancy in 1993 and operated independently until 1996 when he joined the Indian company Ranbaxy to set up the infrastructure of their new subsidiary. For the last 7 years he has been a consultant in the field of generics. |
| If you would like to contact Peter to give him feedback or enquire about Interpharm Consutlancy, please email him at peter@interpharm-consultancy.co.uk
CPhI then and now
Like many readers of this article, I spent the first few days of November walking around the CPhI and ICSE exhibitions in Madrid. I imagine that like me, many readers also found themselves at the end of the day with sore feet and aching joints from all the walking around. Add to this all the weight of giveaways and literature that we pick up during the exhibition and it is not surprising that we reach the end of the exhibition feeling that we have run or walked a marathon and feeling ready to collapse.
It seemed to me that part of the problem is that the exhibition appears to be growing significantly larger each year. I therefore thought it worthwhile to look back at previous years exhibitions to determine whether this is just a case of me growing old and feeble or whether CPhI had really expanded significantly.
The good news is that I am not growing as old as I feared, but rather that the statistics show that CPhI itself has really grown. Since I have the CDs from 1997 and 2001 that the organisers used to issue, (and which I personally find more convenient than the on-line catalogue) I was able to find some figures to compare to those for 2005.
It is of course possible that my database queries did not produce accurate results, but they seem to show that exhibitor numbers have changed as follows:-
|
Country |
1997 |
2000 |
Change 97
-2000 % |
2005 |
Change 2000
- 05 % |
|
Total |
680 |
932 |
+37.1% |
1,508 |
+ 61.8% |
|
China |
39 |
222 |
+469.2% |
578 |
+160.4% |
|
India |
59 |
75 |
+27.1% |
163 |
+117.3% |
|
Italy |
68 |
55 |
-19.1% |
77 |
+40.0% |
|
Korea |
1 |
16 |
+1500% |
35 |
+118.8% |
|
Spain |
24 |
25 |
+4.2% |
52 |
+108.0% |
There is actually a point to this exercise other than proving that I am not imagining things as a number of interesting issues emerged from the analysis.
Firstly, exhibitor numbers have grown over 8 years by 122% from 680 in 1997, to 1,508 in 2005. I do not know what the comparable figures were in 1997, but the organisers say that over 20,000 visitors attended the exhibition this year. The significance of this is that the exhibition was originally set up to provide a forum where suppliers of APIs to the generic industry could meet. Thus, the growth in exhibitor numbers must be seen as reflecting the growth in both the size and importance of the generic industry itself.
Secondly, parallel to the growth of exhibitor numbers overall is the almost explosive growth in the numbers of suppliers from China, India and Korea. At the same time, the numbers of suppliers from the more traditional API source countries of Italy and Spain has remained almost stable or at best increased slightly. This must be very convincing evidence of the movement of the focus on manufacture and supply for APIs and to a degree generic formulations, from Western Europe to the Far East.
In choosing the example countries to use for these comparisons, my main criterion was countries that are associated with the supply of APIs to the generic formulation industry. While it is true that, in percentage terms, the Italian and Spanish presence has grown, their numbers are still very small compared to those of the new API source countries.
Trying to extrapolate from previous trends in order to predict future trends is always risky. I will nevertheless risk predicting that formulations manufacturers in the newer API source countries will try to act in the same way as many Italian and Spanish API manufacturers who have moved on into producing formulations to benefit from the better margins and higher value.
The phase has clearly started as shown by the many Indian generic companies establishing their presence in Europe and the USA. Many of them started by producing APIs, then built formulation plants. They used these initially to carry out contract manufacturing for Western firms and then started to sell their formulations directly into the generic market places.
It is probably a fairly safe bet to assume that the Korean and Chinese API manufacturers who have seen this happen will also want to emulate them and follow the same path. This in turn raises two significant questions.
1 when will this happen? and
2 what will be the effect on prices in the Western generic markets?
With regard to when, there is something of a consensus that China is about 10 years behind India but Korean firms may well be closer to India because the political and economic systems are more open than in China. This in turn is likely to give Western customers for formulations a little more confidence, but there seems little doubt that eventually Chinese formulations will appear in European pharmacies.
Looking still further ahead in the evolutionary cycle of the generic industry, where will generic companies turn after China in their search for still cheaper supplies?
If you have any comments or questions on this article, or would like to suggest a topic for an article, please feel free to contact me on
peter@interpharm-consultancy.co.uk.
Peter Wittner
December 1st 2005
www.interpharm-consultancy.co.uk
Back to top
|
GENERICS EVENT WATCH DECEMBER '05 - DECEMBER '06 |
Biotech and Pharmaceutical Patenting
30-31 January, Paris: This 2 day conference is specifically designed to provide you with a practical and intensive review of the very latest developments in patent law and patent office practice, the important business considerations that must underlie your biotech and pharmaceutical patenting strategies, and the current tactics that are proving successful for the leading players in biotechnology today.
2nd International Forum on Business Development in API and GMP Fine Chemicals
23-24 January, Rome: Developments, trends and case studies.
Pak Pharma
17-19 January, Karachi: Serves as a source of providing business opportunities to the various stakeholders in the Pharmaceutical Industry of Pakistan.
Generics 2006
12-13 January, Barcelona: Topics include: Challenges and Opportunities in Spanish Generic Market, New Data Exclusivity for Generics, Tactics Regarding Portfolio Selection - Global Generic Company Experience, Tactics Regarding Portfolio Selection Small/Middle Size Company Experience, and Role of Corporate Brand and Individual Brands in Generics.
Chemical World 2005
15-19 December, Ahmedabad: Exhibition on Chemicals, Chemical Plant & Equipment, Pharmaceutical and Process Industry.
Interchimie Maroc 2005
7-10 December, Casablanca: Mediterranean Exhibition for Chemical, Pharmaceutical & Cosmetical Technologies.
Generics IP
8-9 December, Berlin: Getting the most out of data exclusivity.
Regulatory Challenges in the CEE Pharmaceutical Market
5-8 December, Warsaw: Seminar titles include; Assess the Impact of Parallel Trade on your Business, and Build a Robust Pricing Strategy & Tackle The Reimbursement Crisis.
If you would like us to include details of any additional events that are relevant to the generics industry in our future newsletter please email us.
Back to top
"The impact of patent expiries on sales of major drugs" Urch Publishing, September 2005.
The period 2005 to 2009 will see the expiration of patent protection for 39 major drugs in the United States, an average of 8 a year. With 2004 seeing a decline in the number of NCEs approved down to 23 from 27 in 2003, there is clear evidence that the loss of revenue from patent protected drugs will impact upon the growth of the industry when so few new drugs are reaching the market.
Back to top
|
The GenericsWeb Patent Intelligence Newsletter is available in both HTML-format (text & images) and text-format. If you have any problems with the format, please let us know by emailing: info@genericsweb.com . If this information is not relevant or of interest, please accept our apologies.
Disclaimer: Please note that all information provided in this newsletter is considered accurate at the time of writing but is not guaranteed and may change over time. Opinions expressed in this newsletter are those of the authors and are not intended to represent legal or investment advice. Information should be verified for accuracy and advice should be sought from a suitable professional before acting upon any information contained herein. |
|
GenericsWeb
|
|
GenericsWeb is a division of XIP Pty Ltd responsible for provision of information products dedicated to the generic pharmaceutical industry.If you are interested in contributing to the GenericsWeb newsletter or would like to discuss promotional or partnership opportunities please feel free to contact us.
email: info@genericsweb.com
web: www.genericsweb.com
tel no.: +61 2 9818 6111
fax no.: +61 2 9818 7786
Address: PO Box 202
Balmain NSW 2041 Australia |
To unsubscribe from this newsletter list, click here: ##Unsubscribe Link##
|
CONTENT
Patents
Drug In Focus: Lansoprazole
Resources
Events Watch
Industry Reports
|