|
Welcome to the October edition of INNsight; this month's Drug In Focus is Glimepiride, Anna McKay discusses when SPCs can be granted, and Peter Wittner discusses the growth of generics in Europe.
If you were forwarded this email and would like to receive future editions of INNsight, become a member by clicking here.
As with all aspects of GenericsWeb we love to learn from your feedback; please email us your comments about our newsletter format, article content or any other matter.
| 'Drug In Focus' is written by Leighton Howard, a patent information expert with extensive experience in the generic pharmaceutical industry. He has worked within the professional patent information industry and major generic pharmaceutical firms. He is the founder of XIP Pty Ltd, the professional patent search firm responsible for researching all patent data found in GenericsWeb Pipeline Patent Intelligence. Please email any comments or queries. |
|
Glimepiride
Patents covering the antidiabetic active ingredient Glimepiride
(Amaryl, Dimeryl) are due to expire in December 2005 in major
European markets and Australia. Based on information
contained in the GenericsWeb Pipeline Selector report for
Glimepiride, this months Drug In Focus analyses the patent
landscape surrounding this product with a view to launching
generic equivalents in those major markets.
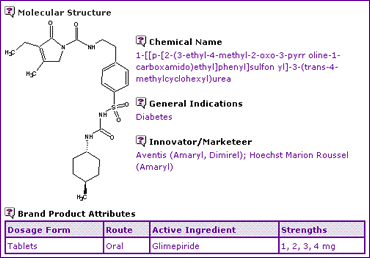
The Glimepiride General Information (Table 1) indicates that only an oral dosage form is currently marketed in four strengths, containing Glimepiride base as the active ingredient.
Table 1: Glimepiride General Information
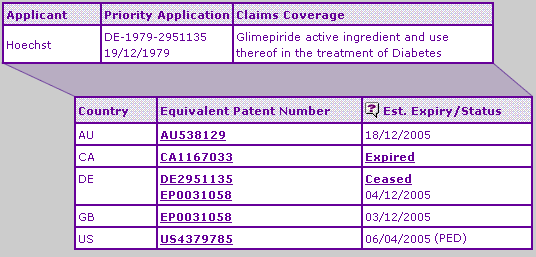
The Key Patent Indicator (Table 2) for Glimepiride confirms that the SPC extensions on the Glimepiride active ingredient patent expire in Germany and the UK in early December, after a full five-year extended term. The equivalent Australian patent expires on December 18th due to a later TGA authorisation date, whilst the protection based on the US equivalent is extended by virtue of a paediatric extension until October 6th 2005.
No other key patent families are identified, which indicates that development of an equivalent to the brand product will not prove to be too difficult for generic competitors.
Table 2: Glimepiride Key Patent Indicator
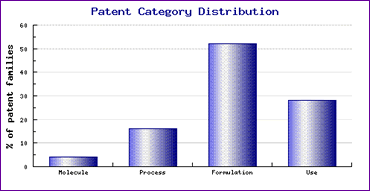
Study of the Patent Risk Analysis section, based on comprehensive patent data (details of which are accessible in the corresponding Pipeline Developer subscription) indicates that patenting activity relating to this drug is concentrated in the formulation category (Figure 1).
Figure 1: Glimepiride Patent Category Distribution
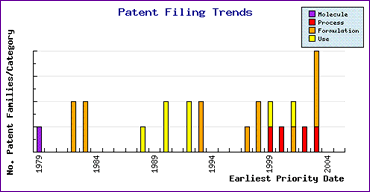
Study of when these formulation patent applications
were filed using the Patent Filing Trends graph (Figure 2)
indicates that they were filed prior to 1985 and would therefore
ordinarily expire in Europe prior to the active ingredient
patent (the corresponding US equivalents may have later expiries
due to their term being calculated from the issue date).
The reason for the very early relevant patent filings is possibly
due to broad claims encompassing the sulfonylurea class of
antidiabetics generally rather than Glimepiride specifically,
which may indicate the right to use the technology disclosed
therein upon expiry of the relevant patents. The Filing
Trends graph also shows later-filed formulation patents, as
well as some unexpired API synthesis patents suggesting that
caution should be exercised in ensuring that all relevant
patents have been considered. Although such patents
may represent a stumbling block to developing a generic
Glimepiride product in major markets, it is not likely that
they will prevent generic competition to any significant degree.
Figure 2: Glimepiride Patent Filing Trends
Despite the limited subsequent patent protection of the Glimepiride product by the innovator, it us unlikely that we will see generic competition in Europe until some time after expiry of the active ingredient patent. This unusual situation is brought about by the length of time between patenting the active ingredient and the date of obtaining first marketing authorisation as outlined in the Glimepiride First Marketing Authorisation table (Table 3).
Table 3: First Marketing Authorisation Table

In some major European generics markets, such as the UK, Germany and France, regulatory authorities are not allowed to accept applications for generic products that rely on data submitted by the originator until 10 years after the first EU marketing authorisation has expired. This restriction on filing for generic approval means we are not likely to see approval of the first generic Glimepiride products until the second half of 2006, allowing for a 12-18 month approval period.
(To download GenericsWeb's guide to calculating data exclusivity in Europe, please click here)
Such data exclusivity provisions carry a much shorter term of 5 or 6 years in certain European countries (e.g. Austria, Ireland, Spain etc.), Australia and the USA. The launch date of generic equivalents in these countries is therefore likely to be determined by the expiry of the active ingredient patent term, unless the application is made via an EU centralised procedure (in which case the 10 year term applies).
In summary, the market for Glimepiride has not been well protected by the innovators beyond the life of the active ingredient patents. However, due to the first EU marketing authorisation of products containing this active ingredient late in the life of these patents, it should not experience generic competition in some major EU markets until some time after this, perhaps late 2006. This will provide up to an additional year of monopoly profits for the innovator beyond the patent term expiry in those markets.
Comprehensive data for patent families relating to Glimepiride, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. During the month of October, GenericsWeb are pleased to offer a 20% discount on the standard price of a Pipeline Developer subscription to Glimepiride. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
Click here for more info on GenericsWeb's Pipeline Selector and Pipeline Developer patent intelligence.
Correction:
In the Drug in Focus section of our September newsletter we inaccurately stated the following: "The Key Patent Indicator (Table 2) for Lacidipine confirms that the SPC extensions on the Lacidipine active ingredient patent expire in Germany and the UK in early November 2006" The Key Patent Indicator table for Lacidipine referred to in the newsletter clearly showed the expiry dates as being November 2005. We apologise for this typographical error in the discussion, and for any concern it may have caused to those planning a European launch of Lacidipine in November 2005.
Leighton Howard
Managing Director
GenericsWeb
September 2005
l.howard@genericsweb.com
|
DEVELOPMENTS AT GENERICSWEB |
Meet us in Madrid at the CPhI / ICSE exhibition
If
you are attending the CPhI / ICSE exhibition in Madrid, November
1-3, then please take the opportunity to meet the GenericsWeb
team at Stand H41, Hall 5. We would be delighted
to show you why our customers value our patent intelligence
so highly by means of a demonstration. Please email
us if you would like to schedule a time to
meet. Click
here to register for the event.
New Pipeline Developer reports
Our patent department has recently completed Pipeline Developer reports for Irbesartan and Nebivolol. The comprehensive patent information and analysis contained in our Pipeline Developer reports for these and many more drugs can be provided within one business day.
Handy web resource
The 'resources' section of GenericsWeb now contains quick links to Generic Pharmaceutical companies, associations around the world, and also generics related conferences and exhibitions. Click here to explore this resource.
Any questions? Click
here to see our FAQs.
If you are new to GenericsWeb you may not know that there is daily generics news archived at our site:
| Contributor Anna McKay specialises in IP Law and Strategy for Pharma companies. As an English solicitor, she won many leading patent cases before the Patents Court, Court of Appeal, EPO, and House of Lords for generic companies. She now works independently, advising companies worldwide on IP exploitation and strategy. |
|
If you would like to contact Anna
to give feedback or enquire about her services, please email
anna@annamckay.com
SPCs When Can They Be Granted?
Chirons Application to The Patent Office.
Chirons application : ReFacto
Earlier this month, the Hearing Officer of the UK Patent Office gave his decision in relation to an application by Chiron and Novo Nordisk (the Applicants) for an SPC for their product ReFacto. Two other products containing the same active ingredient already had SPCs, and there was a question as to whether the Applicants were entitled to an SPC given that Council Regulation (EEC) No. 1768/92 concerning SPCs for medicinal products (the Medicinal Products Regulations) does not appear to allow the grant of a certificate for a product which is already the subject of a granted certificate.
The Applicants had applied for an SPC on 17th March 2003 in relation to a patent granted on the 18th September 2002. The first authorisation for ReFacto was stated to be an EMEA authorisation. Two other SPCs based on the same EMEA authorisation for other products containing the same active ingredient had already been granted to Genetics Institute and Karbivitrum and the Examiner rejected the Applicants application on the basis that there were previous SPCs, granted before the basic patent had been granted to the Applicants, and before they had lodged their application for an SPC. The Hearing Officer considered the appeal.
Article 3 of the Medicinal Products Regulations
On the face of it, the wording of Article 3 of the Medicinal Products Regulations which states that an SPC may be granted If, amongst other criteria, the product has not already been the subject of a certificate, would seem to preclude the grant of an SPC to the Applicants, however, all European legislation must be construed purposively, or, as the Applicants argued, teleologically. In other words, the Tribunal or Court must look to its underlying general principles when seeking to find the meaning of a provision.
This kind of construction differs from the traditional literal approach of English Law, and the US approach.
Teleological construction
Because the provision should be interpreted teleologically, the Hearing Officer considered the recitals to the Medicinal Products Regulations, Travaux Preparatoires, and jurisprudence of the European Court of Justice.
The Recitals state, amongst other things that:
Medicinal products, especially those that are the result of costly research, will not continue to be developed in the Community and in Europe unless they are covered by favourable rules that provide sufficient protection to encourage such research
The period that elapses between the filing of an application for a patent for a new medicinal product and authorisation to place the medicinal product on the market makes the period of effective protection under the patent insufficient to cover the investment put into the research
.
The Hearing Officer also took account of the case of Biogen v- Smith Kline Beecham (C181/95) (Biogen). Biogen wanted an SPC based on patents which it had licensed to SKB. Institut Pasteur, which had also licensed patents on the same product to SKB already had an SPC, and SKB argued that the Medicinal Products Regulation permitted only one certificate to be granted for each product that is to say, each identical active ingredient, even when the product in question was based on several patents. The matter was referred to the ECJ, which allowed Biogens application and stated that under Article 3(c) of the Regulation
only one certificate may be granted for each basic patent, Advocate General Fennelly, considering the same case, went further, concluding that the fact that the same product was covered by several basic patents belonging to different holders did not preclude the grant of an SPC to each holder of a basic patent. He stated that the text of the Regulation applies simply to a simple situation, in which basic research, product development, production marketing are vertically integrated: where the holder of the patent or patents relating to a medicinal product, the marketing which has been authorised in the Member State is also the holder of relevant marketing authorisation. The regulation was obviously drafted on the basis of this classic model. Article 1(c) may be thought to assume that in a case where there are numerous patents, possibly of different kinds, these will be held by a single holder who is in a position to choose between them and to designate one as the basic patent for the purpose of the procedure for the grant of a certificate
The assumption becomes more important in Article 3 ( c), which requires that
.the product has not already been the subject of a certificate
.
The Applicants also referred to the Dutch Sigma-Tau Decision. That was a decision of the Dutch Patent Office nullifying a rejection under Article 3(c) of an application for an SPC by Sigma-Tau on the basis that the Wellcome Foundation already had an SPC on the product. The UK Patent Office is not bound by the decisions of other patent offices, and these are taken into account. In the Sigma-Tau case, in contrast to the application being considered by the Hearing Officer, applications for SPCs were co-pending.
Finally, the Hearing Officer affected by the Advocate Generals comment that a restriction of supplementary protection to just one patent in each Member State would almost certainly result in the fragmentation of the market.
Hearing Officers conclusions
Taking all of these factors into account (recitals, Advocate-Generals comments on the meaning of 3(c), and fragmentation of the market, Sigma-Tau decision) and despite the ECJs comment in Biogen that under Article 3(c) of the Regulation
only one certificate may be granted for each basic patent, the Hearing Officer concluded that It makes no sense to impose an arbitrary rule that certificates could only be granted to more than one applicant if the corresponding applications were co-pending
.such an arbitrary rule sits ill with the general principle of legal equity mentioned by Advocate General Fennelly.. [there is] no justification for rejecting the application on the grounds of a Supplementary Protection Certificate having already been granted to the same product, but to a different patent holder when the application was lodged. On the contrary, it seems to me that to reject the application in these circumstances will run contrary to the purpose of the Medicinal Products Regulation, when viewed in the light of Biogen
It is necessary to consider how [Article 3(c)] impinges on application such as the present one, filed after an earlier application for the same product had been granted.
The Hearing Officer stated I take the view that the ECJ intended that it is not permissible for one and the same patent holder to multiply extend the protection for a medicinal product over time by obtaining successive certificates
All holders of basic patents may be granted a Supplementary Protection Certificate, but only one certificate may be granted for that product to each
..The duration of a Supplementary Protection Certificate should be such as to provide adequate effective protection
.it would be unacceptable
for this duration for one and the same medicinal product to be exceeded. Therefore, it should be possible for all patent holders to benefit from supplementary protection of the same product, but the balance between the interests concerned should not be taken too far towards patent holders by allowing them to muliply extend their protection for the product over time by obtaining successive certificates.
Generic Companies take note
The Hearing Officers decision makes rather heavy reading. Clearly, it extends the monopoly afforded to ReFacto. Generic versions of ReFacto will not be able to be marketed until the Applicants SPC expires, notwithstanding the expiry of the SPCs granted in respect of basic patents granted to Genetics Institute and Kabivitrum. Generic versions of the products sold by Genetics Institute and Kabivitrum should be able to be marketed. I have no idea of the differences between the products or the basic patents protecting them, and whether, in practise, the distinction is clear. I think that the decision of the Hearing Officer in this case is probably correct. It does re-enforce the fact that generic companies must be extremely careful in deciding whether or not they are free to market a product. They must check basic patents and SPCs granted to all of those selling products containing a particular active ingredient. It is not enough to assume that they are free to market if the SPC of one party selling the product has already expired.
If you have any questions concerning this decision, I would be happy to consider them.
Anna McKay
October 2005
www.annamckay.com
| Contributor Peter
Wittner has been in the pharmaceutical industry
for 30 years. He has worked for the former Evans Medical
and then Norton Pharmaceuticals (now part of IVAX) where
he was responsible for European Sales & Marketing.
After leaving Norton Peter set up his own consultancy
in 1993 and operated independently until 1996 when he
joined the Indian company Ranbaxy to set up the infrastructure
of their new subsidiary. For the last 7 years he has
been a consultant in the field of generics. |
| If you would like to contact Peter to give him feedback or enquire about Interpharm Consutlancy, please email him at peter@interpharm-consultancy.co.uk
Generics then and now
While looking for some information, I came across a copy of the NERA report entitled POLICY RELATING TO GENERIC MEDICINES IN THE OECD COUNTRIES commissioned by the EU and delivered to them in December 1998 by NERA (National Economic Research Associates).
The purpose of this report was amongst other things to conduct an examination of the role of generics in medical practice in the regions studied, including how far the authorities are promoting the use of generic medicines
and also to look at the technical requirement for licensing and marketing authorisation, issues relating to intellectual property and R&D.
What is particularly interesting about this report now is how it gives a snapshot of the state of European and world generics in 1997 and early 1998. In its summary, NERA stated In the EU, the weighted average of generics in all prescribed medicines by value was 15 per cent in 1996 - 1997 with generics shares being highest in Germany (39 per cent), Denmark (38 per cent) and Finland (32 per cent). It was lowest in Spain (insignificant), Portugal and Italy (3 per cent) and Sweden (4 per cent). It also noted Outside Europe, the weighted average by value for those OECD countries for which we have data is 10 per cent. It was highest in Canada (15 per cent) followed by the USA (11 per cent).
| |
Market Share By Value (%) |
| Country |
NERA 1997 |
EGA 2004 |
| Austria |
9 |
7.8 |
| Belgium |
6 |
6.1 |
| Denmark |
38 |
32 |
| Finland |
32 |
7.6 |
| France |
2 |
6.4 |
| Germany |
39 |
22.7 |
| Ireland |
13 |
5.4 |
| Italy |
3 |
2.1 |
| Netherlands |
13 |
19.8 |
| Portugal |
3 |
8.8 |
| Spain |
0 |
5.2 |
| Sweden |
4 |
12.3 |
| United Kingdom |
22 |
20.6 | Sources: NERA - 1997 data, EGA - 2004 data
|
Have things changed since then? The answer as always is Yes and No. The table shown on the left compares the position in Europe as seen by NERA with the current situation as seen by the EGA (European Generic Association. This is in turn based on data supplied to them by individual country associations to reflect the market position at the end of 2004.
There is a difficulty in that the two are not directly comparable as each uses a slightly different definition of what is a generic with NERA, for example, including branded generics in the numbers. In addition NERA made a number of assumptions and calculated estimates, as they could not find any published data. Indeed, the authors comment, Volume data as to generics market penetration and the shares of INN and branded generics are too incomplete for conclusions to be drawn.
Nevertheless, and treating the comparisons with suitable caution, there are some signs of change. The figure for France of 2% in 1997 is probably roughly correct and similarly the estimate of 0% for Spain is likely to be accurate. The 3% generic market share for Italy and Portugal in 1997 is almost certainly inaccurate as it was probably closer to 0% for both at that time. |
Assuming that all the respondents to the recent EGA survey were working on the same basis of a generic being a product known and prescribed by its INN name, then the picture for these same four countries in 2004 is somewhat different, but not hugely so. Although there must be some doubt concerning the accuracy of the NERA figures, there is no doubt that a generic market has arisen in each country although it is not yet particularly large in any of them, with market shares of only a few percent.
The countries have much in common from a generic perspective as all have made significant efforts in the last few years to create a generics industry and promote generics use as a cost control measure. They have succeeded in this to the extent that all have introduced measures to facilitate generic registrations, encourage generic prescribing and dispensing and introduced reference-pricing schemes.
Local generics industry observers though suggest that while the government measures to promote generics have been welcome, their efforts to tightly control pricing have been less welcome and could even prove counterproductive. The feeling is that the price squeeze has been too tight and that there is a risk that the newborn generics baby will be strangled, if not at birth, then at an early age.
Has anything changed in the larger, more mature markets? Well, market share has certainly not dropped from 32% in Finland to 7.6%, as it is unlikely that the figure was ever as high as 32%. It is similarly unlikely that generic market shares have fallen in Denmark, Germany or the UK as the table implies at first glance. Market shares in volume terms have been steadily rising in all these countries although it is fair to say that market share in value terms have certainly lagged behind due to pricing pressures, but they have not lagged so far as to result in a significant fall when compared to 1997.
In summary, NERAs comments were certainly correct about the negligible sizes of the generic market in the countries shown as being at the bottom of the generic ladder with only a few percent market share. On the other hand, the figures that NERA provided can probably not be relied upon as being exact but rather as being illustrative. Nevertheless, things have changed as market shares have risen from negligible to small which is at least a form of progress.
The full report is available at:
http://pharmacos.eudra.org/F2/pharmacos/docs/nerareport.pdf
If you have any comments or questions on this article, or
would like to suggest a topic for an article, please feel
free to contact me on peter@interpharm-consultancy.co.uk.
Peter
Wittner
28th September 2005
www.interpharm-consultancy.co.uk
GENERICS EVENT WATCH AUGUST - SEPTEMBER 2005
|
EURO Plx 26 Porto
30 November - 1 December, Lisbon:
Proposed orientation of EuroPLX 26 is towards collaborative agreements in dossiers, custom synthesis, contract manufacture, R&D services, and supply of APIs.
11th Annual EGA Conference
21-22 November, Berlin:
'Ensuring a Competitive Environment for Generic Medicines in Europe', this two-day conference of the European Generic Medicines Association (EGA) will focus on measures to ensure a positive competitive environment for the sustainable delivery of cost-effective generic medicines. Topics include pricing and reimbursement, regulatory and IP issues, and current German healthcare reforms. The conference will end with a panel discussion with leading CEOs from the European generic medicines industry.
5th Annual Forum on Generic Drugs
17-18 November, Washington: Topics include 30 month stays and 180 day exclusivities, forfeiture events, medicare reforms, authorized generics, market excusivity, generic biologics, and the Indan generics market.
CPhI / ICSE / P-Mec
1-3 November, Madrid: International Exhibition on Pharmaceutical Ingredients and Intermediates / Contract Services / Pharmaceutical Manufacturing. Look for GenericsWeb at Stand H41, Hall 5.
China-Pharm 05
25-28 October, Shanghai: The 10th China International Pharmaceutical Industry Exhibition Participation of the Federal Ministry of Economics and Labour
EuroPLX 25
8-9 October, Zurich: Proposed orientation of EuroPLX 25 is towards collaborative agreements in prescription drugs, patented or generic, as well as drug delivery technology.
Pharma India
1-3 October, Delhi: International Exhibition & Conference for Pharmaceutical Industry, Raw Materials, Manufacturing and Packaging Industry.
If you would like us to include details of any additional
events that are relevant to the generics industry in our future
newsletter please email
us.
"The impact of patent expiries on sales of major drugs" Urch Publishing, September 2005.
The period 2005 to 2009 will see the expiration of patent protection for 39 major drugs in the United States, an average of 8 a year. With 2004 seeing a decline in the number of NCEs approved down to 23 from 27 in 2003, there is clear evidence that the loss of revenue from patent protected drugs will impact upon the growth of the industry when so few new drugs are reaching the market.
|
The GenericsWeb Patent Intelligence Newsletter is available in both HTML-format (text & images) and text-format. If you have any problems with the format, please let us know by emailing: info@genericsweb.com . If this information is not relevant or of interest, please accept our apologies.
Disclaimer: Please note that all information provided in this newsletter is considered accurate at the time of writing but is not guaranteed and may change over time. Opinions expressed in this newsletter are those of the authors and are not intended to represent legal or investment advice. Information should be verified for accuracy and advice should be sought from a suitable professional before acting upon any information contained herein. |
|
GenericsWeb
|
|
GenericsWeb is a division of XIP Pty Ltd responsible for provision of information products dedicated to the generic pharmaceutical industry.If you are interested in contributing to the GenericsWeb newsletter or would like to discuss promotional or partnership opportunities please feel free to contact us.
email: info@genericsweb.com
web:
www.genericsweb.com
tel no.: +61 2 9818 6111
fax no.: +61 2 9818 7786
Address: PO Box 202
Balmain NSW 2041 Australia |
|
CONTENT
Patents
Drug In Focus: Glimepiride
Resources
Events Watch
Industry Reports
|