The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic Celecoxib equivalents of Pfizers blockbuster anti-inflammatory drug Celebrex®. Global sales of Celebrex® exceeded USD2.1 billion, in the first nine months of 2013, a 13% increase on the same period the previous year.
General information
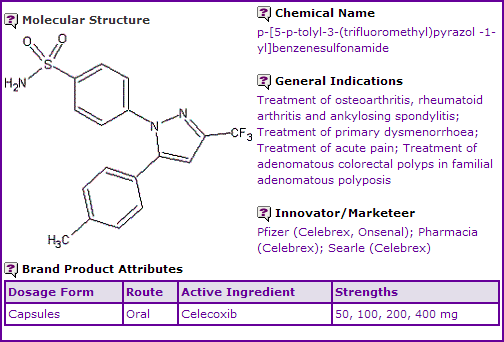
Celecoxib is a sulphonamide non-steroidal anti-inflammatory drug, which acts as a selective COX-2 inhibitor, marketed by Pfizer, Pharmacia and G.D. Searle (both subsidiaries of Pfizer), as Celebrex, Onsenal and Celebra. It is indicated for use in the treatment of osteoarthritis, rheumatoid arthritis, ankylosing spondylitis, acute pain and primary dysmenorrhea. The product is currently available in an oral capsule form in multiple dosage strengths. The Celecoxib molecule was first discovered through work arising from collaboration between Brigham Young University and G.D. Searle (a subsidiary of Monsanto at the time). Searle was later merged with Pharmacia then acquired by Pfizer from Monsanto.
Figure 1: General Information table for Celecoxib
INN Constraint Comment
Patent protection via the patent family, US16059493A (30/11/1993), is the primary hurdle for companies wishing to develop generic equivalents, as it provides protection for the Celecoxib molecule and its use in the treatment of arthritis or inflammation (Fig. 3). Originally expiring in November 2013 in the US, paediatric extensions have extended the expiry of the molecule and composition patents, US 5,466,823 (823) and US 5,563,165 (165), a further six months, until May 2014 (Fig. 3). Teva has already tested the waters by filing an ANDA with a paragraph IV certification, however the validity of claims in the molecule and composition patents have been upheld following appeal to the US Court of Appeals for the Federal Circuit.
In the same proceedings, US 5,760,068 (068), expiring in December 2015 following grant of additional six months of paediatric extension, was deemed invalid based on obviousness-type double patenting. Pfizer sought to correct prosecution errors and it was subsequently replaced with USRE44048 (048) in March 2013. This patent provides protection for the current authorised indicated uses of Celecoxib in the treatment of inflammation or an inflammation related disorder specifically arthritis (osteo, rheumatoid or juvenile), pain, menstrual cramps spondyloarthropathies, fever, and colorectal cancer. Searle and Pfizer moved quickly to assert their intellectual rights filing a complaint against Lupin, Teva, Mylan Watson and Apotex in the District Court of Virginia upon grant of 048. This will potentially prevent generic competition from launching their product at risk beyond the expiry of 823 and 165 in May 2014. Therefore it is considered that the Use patent, 048, may further constrain generic entry until its expiry in December 2015.
The equivalent molecule, composition and use patents in Australia, Canada and Europe are due to expire on 14 November 2014. In Europe, a supplementary protection certificate (SPC) for the French designation of EP0731795 extends the term until December 2014. However, GenericsWebs proprietary SPC analyser has identified it as using the later Swedish market authorisation as the first market authorisation date for the basis of its expiry calculation, instead of the earlier Swiss date. Therefore it may be susceptible to challenge and/or review. SPC applications in Germany and the UK have lapsed or been withdrawn.
Families with priorities US2849496P (15/10/1996) and US94992297A (14/10/1997) protect the use of Celecoxib in the treatment of adenomatous polyps. Celecoxib had been indicated for use in the reduction of familial adenomatous polyposis as an adjunct to surgery and endoscopic surveillance. In Europe, it was issued with a market authorisation as an orphan designation under exceptional circumstances as the condition is sufficiently rare such that the usual safety and efficacy requirements were relaxed. However, Pfizer was never able to provide enough evidence to support this indication and therefore the indication has been withdrawn in all major jurisdictions. Subsequently, the US patent 5,972,986 was removed from the FDA Orange Book following the withdrawal of the indication. At the present these patents are not considered to constrain generic entry however, if Pfizer is able to provide more evidence for the use in familial adenomatous polyposis and is granted market authorisation, then these patents will be a constraint to generic competition for this indication.
Patents in the US94978092A (22/09/1992) family have been identified as Key due to litigation for US member 6,048,850, after the University of Rochester sued Searle for patent infringement. In 2004, The Court of Appeals for the Federal Circuit, ruled in favour of Searle, holding in essence that the university had claimed a method requiring, yet provided no written description of, a compound that could inhibit COX-2 and therefore the patent was invalid. Therefore, the family is not considered to be a constraint to generic entry.
Family members of US11033398P (30/11/1998) are not considered to be a constraint to generic competition because the protected technologies are likely to be circumvented.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Celecoxib
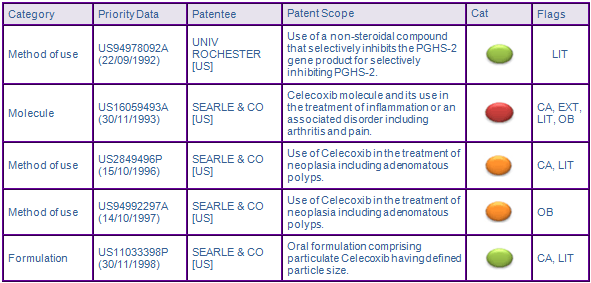
Figure 3: Patent Family View priority application US16059493A
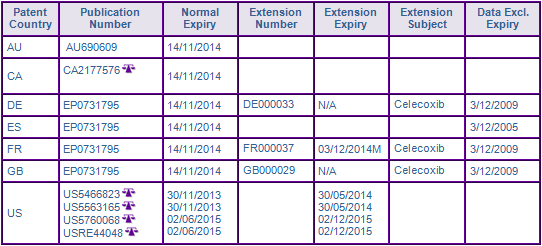
Figure 4: Marketing Authorisations for products containing Nevirapine in Key Countries
Amongst the US approvals, tentative approvals have been granted to Teva, Mylan, and Watson for their generic equivalents to Celebrex®. Originally Teva filed an application with paragraph IV certifications against the molecule patent however the court ruled that the claims were valid and therefore Teva and other companies will have to wait until expiry until its expiry to launch their products. Additionally, patent protect by way of the reissued Use patent 048 may prevent final FDA approval until December 2015.
In the UK, Teva and Pfizer have been granted approval to market a generic product. It is likely that more generic competitors will receive market approvals for generic Celecoxib, as indicated by other European markets, leading up to the expiry of the molecule patent.
Pfizer has also gained generic market authorisations in Canada. Pfizer and Searle filed Orders of Prohibition against Novopharm, Mylan, Apotex and Teva following their submission of ANDS to manufacturer generic versions. In the Novopharm case, the Court of Appeals reversed the Application Judges previous decision of invalidity for the Celecoxib molecule patent, preventing them from obtaining a Notice of Compliance until the expiry of the patent.
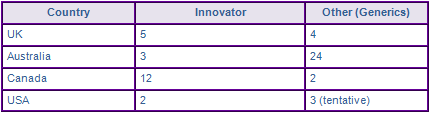
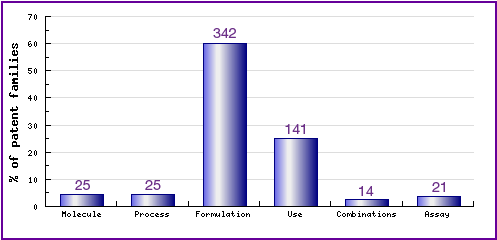
Figure 5: Top Patent Applicants
This figure is a representation of patentees who have filed the most patent families for this INN (Fig. 5). Note that applications from Pfizer, Pharmacia and Searle make up 19% of the total number of patents applied for, while University of Texas and McNeil are mostly general NSAID formulations. However, a large portion of the patent landscape is made up of non-innovator filings, which is to be expected in the popular anti-inflammatory market.
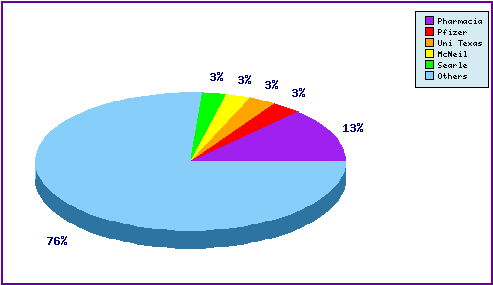
Figure 6: Patent Filing Trend
Figure 6 represents the timing of the earliest priority filing date for each patent family identified for this molecule as well as the type of claims found in the applications. Note the significant increase in filing immediately after approval and strong filing numbers since then.
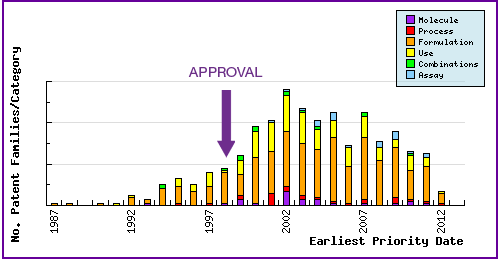 Figure 7: Patent Category Distribution
Figure 7: Patent Category Distribution
The types, number and relative distribution of patents that have been filed for Celecoxib obtained via a comprehensive patent search (Pipeline Developer report). Note the dominance of patent filing of formulation. Much of this represents general patenting that are related to either NSAIDs, COX inhibitors or patents that use Celecoxib as an example but do not claim Celecoxib specifically. However, there has been some attempt by generic competitors in developing technology to overcome the difficulties of formulating oral dosage forms of Celecoxib due to its low solubility and compressibility, and to circumvent innovator patents. Formulation and Use type patents by the innovator can be indicative of likely lifecycle management of a product as the innovator seeks to maintain their monopoly. In October 2009, Pfizer applied to extend the indications of Celebrex® to include chronic pain, however authorisation for this indication is pending the results of further clinical trials. Some patents obtained by the innovators may protect this indication, so may be important for generic developments in the future.
In summary, patent protection for the Celecoxib molecule is currently constraining generic entry, and will do so in most major markets until November 2014. Attempts to extend the protection by an SPC have been successful in France, extending the term until December 2014. However, the SPC application used the later Swedish market authorisation date for purposes of calculation the extended term instead of the earlier Swiss market authorisation which would have been valid in Liechtenstein, so may be susceptible to invalidation. In the US, the validity of patents protecting the Celecoxib molecule were upheld, preventing the grant of ANDAs until May 2014. However, Searle and Pfizer gained a valid reissue of 068, a Use patent covering all the currently marketed indications for Celecoxib. This potentially extends their monopoly until December 2015 in the US. Searle and Pfizer were quick to assert their intellectual property rights and, potentially preventing generic competitors launching at risk, filed patent infringement suits against Lupin, Teva, Watson and Apotex. The market should be very open to competition after this time as no other patent significantly constrains the market authorised product. A number of generic competitors are already setting themselves up in what is likely to be a very hard fought generic market.
Do you want find out more about how we can assist your generic drug development?
Share this article:
To register for GenericsWeb's free monthly newsletter 'INNsight', click here