The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic equivalents of Mercks blockbuster non-nucleotide reverse transcriptase inhibitor (NNTRI), Efavirenz.
General information
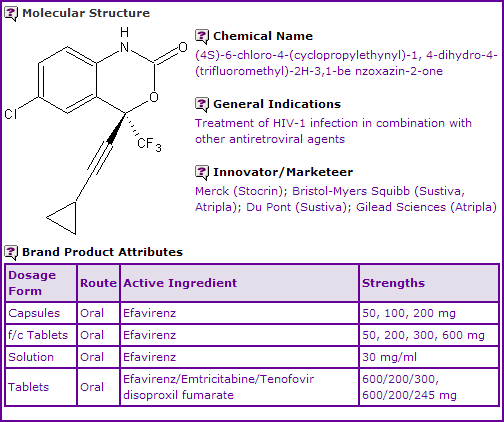
Efavirenz is currently available in several oral dosage forms. The single-active product is marketed in some territories (including United States, Canada and six major countries of the European Union) by Bristol-Myers Squibb (BMS) under the brand name Sustiva®, and in others by Merck Sharpe & Dohme under the brand name Stocrin®. In 2004, a collaborative deal struck between Merck, BMS and Gilead, lead to the development and commercialisation of Atripla®, a fixed dose combination tablet of Efavirenz, Emtricitabine and Tenofovir. All products are indicated for the treatment of HIV-1 infection.
Figure 1: General Information table for Efavirenz
INN Constraint Comment
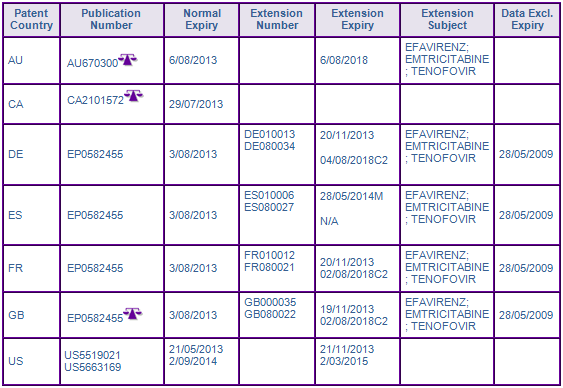
According to the key patent indicator (Figure 2), the patent protecting the Efavirenz molecule in the family US92660792A (07/08/1992) expired in Canada in August 2013 following the normal term, however the US members have been extended by paediatric extension for a further 6 months, extending protection until March 2015.
The Australian equivalent has a normal expiry in August 2013, but is subject to an application for extension of term based on the TGA approval of Atripla, which would expire in 2018, if granted. Whether this application would, in fact extend the portion of the patent scope relating to products containing Efavirenz as a single active ingredient is yet to be determined.
In Europe, granted SPCs seek to extend patent protection of products containing the single active ingredient until November 2013 in major European member states. As identified by GenericsWebs proprietary Validity Flags, the Spanish SPC amongst these expires in May 2014 due to using the later centralised EU market authorisation date as the basis of its expiry calculation instead of the earlier Swiss date used by other SPCs attributed to patents in the family. Such SPC expiry dates are susceptible to challenge and/or review.
Additional SPCs protecting the combination of Efavirenz, Emtricitabine and Tenofovir have also been granted in Germany, France and the UK citing the Efavirenz molecule patent and expire in August 2018. GenericsWebs proprietary SPC analysis has again flagged these SPCs as being a C2 category, suggesting that the claims of the basic patent do not specify or identify all components of the combination that is the subject of the SPC, and therefore the SPC may be invalid. The claims of the basic patent EP455 seek to protect the use of Efavirenz with other nucleoside analogues being active against HIV, of which Emtricitabine is one. However, this does not specify the combination with the other active component Tenofovir, which is a nucleotide analogue. The Spanish SPC application was rejected. Any attempts to invalidate these SPCs on the basis of Medeva will likely be met with fierce resistance, with Gilead reporting global sales of Atripla generating US$1.82 billion, the combination product outselling the single active form of Efavirenz. Due to three active ingredients being present in the combination product, the SPC situation in Europe is extremely complex with applications made across a number of patent families, many of which have questionable validity. However, the European market appears to be full of opportunities for the fully-informed.
Patents in the family US44024603P (14/01/2003) seek to protect the combination of Emtricitabine and Tenofovir fumarate and its combination with Efavirenz, so therefore may constrain generic competition for an Atripla equivalent product until expiry. The European member of this patent family, however, was revoked following opposition filed by Teva and Generics UK (Mylan), this decision being subject to appeal. The corresponding US patent application is still undergoing examination. Normal expiry dates of patents in this family are in 2024 if challenges to their validity are unsuccessful, so would not be eligible for any Patent Term Extension based on the first approval dates for Atripla in various markets.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Efavirenz
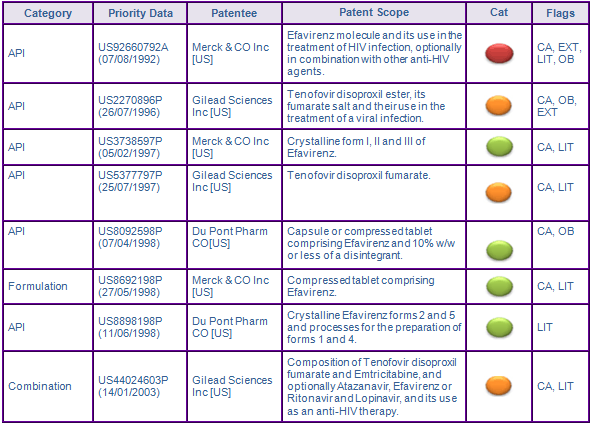
Figure 3: Patent Family View priority application US92660792A
Marketing Authorisations
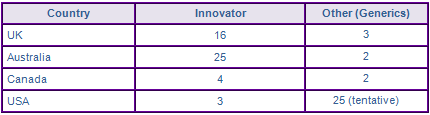
In the USA several companies have gained tentative approval for the single active Efavirenz tablet and capsule dosage forms. Many of these tentative approvals are dependent upon expiry of US patents in family US3738597P, protecting three crystalline forms of Efavirenz. Matrix, Teva, Cipla and Aurobindo have also gained a tentative approval for generic version of Atripla. Several other generic companies have taken advantage of the 505(b)2 pathway reviewed under the Presidents Emergency Plan for AIDS Relief (PERFAR) and gained tentative approvals for fixed dose combinations and co-packaged Efavirenz with other anti-retroviral drugs, but are also subject to similar constraints as the ANDA counterparts. Litigation is underway with regard to these patents in the US following Hetero and Ciplas filing of an ANDA with paragraph IV certification. No generic competitors have secured a market authorisation for an Atripla equivalent product in Europe, due to data exclusivity provisions preventing filing an application until December 2015, however it appears that many competitors have obtained marketing authorisation and will launch imminently an equivalent to Efavirenz in Europe.
Figure 4: Marketing Authorisations for products containing Efavirenz in Key Countries
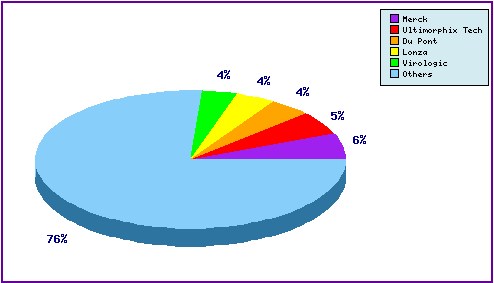
A representation of patentees who have filed the most patent families for this INN is shown in Figure 5. Amongst the top 5 filers are the innovator Merck, however the majority of patent filings stem from a diverse range of companies, whose presence can be attributed to patenting regarding their own anti-retroviral agents in what is a very crowded technological area. This result in very complex patent analyses.
Figure 5: Top Patent Applicants
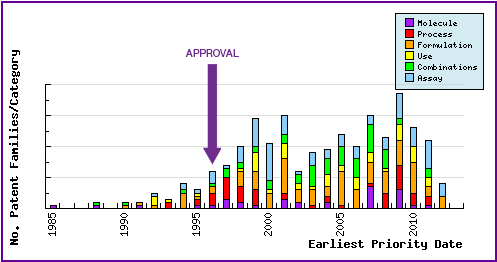
The Patent Filing Trends graph represents the timing of the earliest priority filing date for each patent family identified for this molecule obtained via a comprehensive patent search (Pipeline Developer report) as well as the type of claims found in the applications. Note the filing of many molecular forms prior to commercialisation of the product and the strong responsive patenting by companies post-launch, particular in the area of molecular forms, formulations and combinations. This demonstrates that the innovator has placed considerable effort in protecting as many facets of the Efavirenz product as possible prior to public disclosure.
Figure 6: Patent Filing Trend
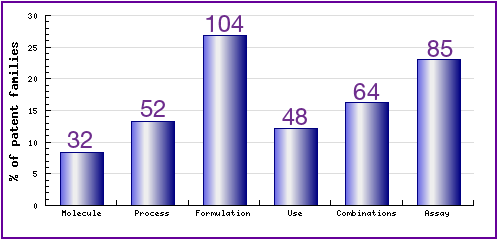
The types, number and relative distribution of patents that have been filed for Efavirenz are shown in Figure 7. Note the presence of relatively high activity in regard to the Efavirenz product. This can be attributed to generic companies seeking to circumvent innovator patents as well as work by other innovators in the field of antivirals generally. Generic competitors should carefully consider these patents when looking to develop and launch their own co-formulated or co-packet products.
Figure 7: Patent Category Distribution
In summary, patent protection for the Efavirenz molecule is currently constraining generic entry in Europe and the US. Whilst polymorph patents appear to be a significant constraint beyond the molecule patent (until 2018) in the US, the corresponding European patent does not carry the polymorph claims so is much less restrictive, allowing launch of the single-active product immediately after molecule patent expiry in November 2013. BMS and Gilead have gone to great lengths to protect their blockbuster combination product, Atripla, by filing SPC applications instead of relying solely on their later combination patent. However, the validity of some of those SPCs appears uncertain due to the claims of the patent not specifically disclosing the combination product; the validity of the combination patent is also being questioned in certain countries. If unchallenged, SPCs will extend protection for Atripla until July 2022 in Europe. In addition, the combination patent will offer protection until 2024 if upheld following the various attacks underway on its validity. Regardless of the patent situation, data exclusivity provisions available in the EU will prevent the filing of a generic application for an Atripla product until December 2015, meaning generic competition for the combination will not be available until at least late 2016.
Do you want find out more about how we can assist your generic drug development?
Share this article:
To register for GenericsWeb's free monthly newsletter 'INNsight', click here