The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of a generic Zanamivir product for GlaxoSmithKlines (GSK) antiviral flu treatment Relenza®. Global sales in 2011 reached £27 million, a 79% decrease from the previous year due to the loss of pandemic flu sales.
General information
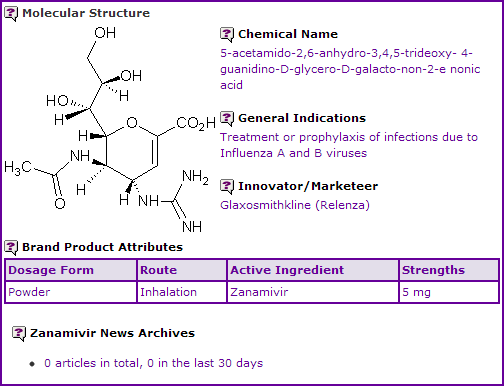
Zanamivir is indicated for the treatment and prophylaxis of influenza A and B viruses and is available as a powder for inhalation in 5 mg strength for use in a Diskhaler device. It was licensed by Biota to GSK in 1990 and was the first neuraminidase inhibitor launched worldwide in 1999.
Figure 1: General Information table for Zanamivir
INN Constraint Comment
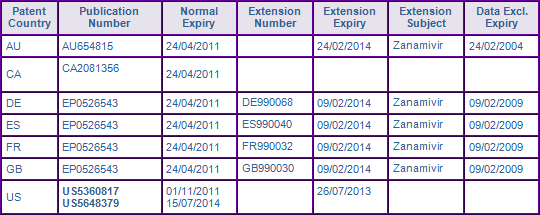
Patent protection via patents in the family AUPJ980090A (Figure 2) for the Zanamivir molecule expired in Canada in April 2011, while the maximum 5 year patent term extension was granted in Australia and most major European countries, extending protection until February 2014 (Figure 3). The US member 5360817 was extended for 633 days under 35 U.S.C § 156, and expired in July 2013. However, the later expiring FDA Orange Book (OB) listed patent 5648379, which also claims the Zanamivir molecule and its use for treating a viral infection, is considered to further constrain generic entry until July 2014.
Patents in family GB9325841A (Figure 2) seek to protect the crystalline form of Zanamivir used in the marketed dosage form. While generally not considered to be a constraint to generic competition, patents claiming the molecular form can still extend the product monopoly, as it may be preferred due to its increased stability and bioavailability. No Paragraph IV certifications have been identified for Zanamivir, suggesting that generic companies will wait for the expiry of these patents. Patents from this family are in force until December 2014 in all major markets.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Zanamivir
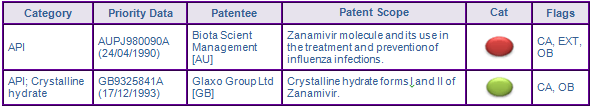
Figure 3: Patent Family View priority application US34097089A
Although data exclusivity has expired in all major territories, there are currently no authorised generic forms or market approvals for a Zanamivir equivalent (Figure 4). This is mainly due to unexpired patent protection in relation to the Zanamivir molecule, thus constraining entry.
Delays to generic entry can also be attributed to the additional costs of device dependent dosage forms. This has been previously observed for other inhalable dosage forms, such as Serevent® (Salmeterol), another GSK product which is available in a 0.05 mg powder for inhalation. Serevent® was authorised in the US in 1997, with patents protecting the Salmeterol molecule expiring in 2005 and 2008 in Europe and the US, respectively. However, in the US there has still been no generic entry for this dosage form. This can be attributed to the OB listed patents relating to the Diskus inhaler device. Thus, in the cases of Zanamivir and Salmeterol, generic companies must take into account the higher developmental costs associated with circumventing innovator patents with a device, while still being able to offer their product at a competitive price.
Figure 4: Marketing Authorisations for products containing Zanamivir in Key Countries

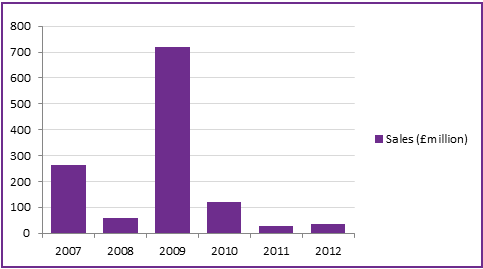
Global sales figures reflect the fluctuation in demand due to government stockpiling and predicted risk of influenza in a given season (Figure 5). This is strongly reflected in the 2009 sales, which generated £720 million as a result of the global pandemic threat of influenza A H1N1 (swine flu).
The significant decrease in sales since 2009 are, in part, due to the risks associated with the inhalable administration of Zanamivir, and competition from Oseltamivir (Roches Tamiflu®) and other influenza vaccines. However, the availability of both vaccines and antiviral drugs are required to effectively prevent or combat an influenza pandemic. Furthermore, the European Medicines Agency has recommended stockpiling more than one influenza drug to cope with emerging resistance, which has been observed due to overuse of Oseltamivir. Studies have also shown that Zanamivir is more effective than Oseltamivir, and is therefore likely to remain a key component for government strategies against influenza. GSK commenced some late stage lifecycle management to try and overcome the drawbacks associated with powder inhalation and potentially increase their market share, commencing phase III clinical trials of an intravenous dosage form. This may provide a more attractive future avenue for generic companies to enter the market with a Zanamivir product.
Figure 5: Global Zanamivir sales revenue
This figure is a representation of patentees who have filed the most patent families for this INN (Figure 5). Note that Lilly the innovator has 41% of the filing relating to Raloxifene and the prominence of the generic companies Teva, Hexal and Sandoz in the top 5. The appearance of Pfizer in the top 5 can be attributed to general patenting relating to its competing SERM products, Fablyn® (Lasofoxifene) and Conbriza® (Bazedoxifene).
Figure 6: Top Patent Applicants
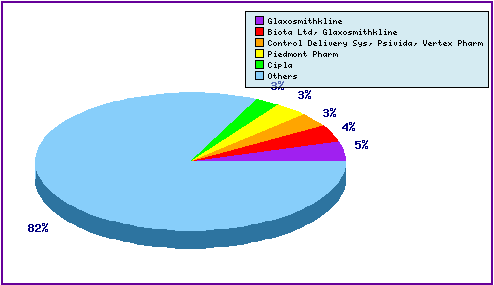
Figure 7 represents the timing of the earliest priority filing date for each patent family identified for this molecule, as well as the type of claims found in the applications. Note the strong representation of use and formulation patents from the time of approval, which mainly relate to antivirals and sialic acid inhibitors.
Figure 7: Patent Filing Trend
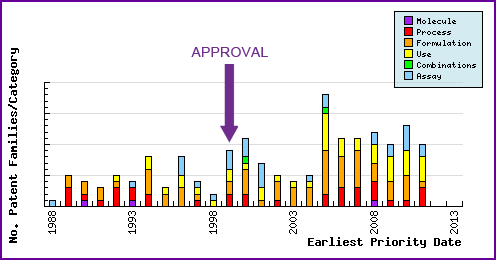
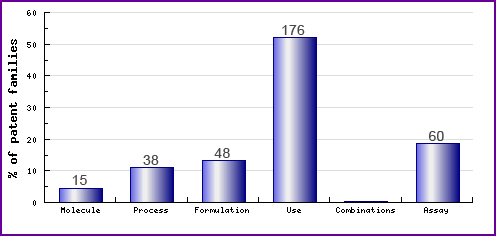
Figure 8 represents the type, number and relative distribution of patents that have been filed for Zanamivir obtained via a comprehensive patent search (Pipeline Developer report). Note the prominence of formulation, use and process patents. The patent landscape for Zanamivir in regard to formulation and use reflects general patenting around antivirals. However, certain patent families filed by the innovator relating to the use of an inhalable formulation comprising Zanamivir may prove difficult to circumvent, and may further extend protection in certain markets. Therefore, generic competitors should carefully consider these patents when looking to develop and launch their products. The process patents are generally for the preparation of sialic or neuramic acid derivatives and analogues which form the core structure of Zanamivir.
Figure 8: Patent Category Distribution
In summary, patent protection for GSKs Zanamivir has expired in Canada, and will remain in force in Australia and major European markets until February 2014, and in the US until July 2014. With the continual threat of an influenza pandemic and increasing resistance to main competitor Oseltamivir, the demand for highly effective antivirals such as Zanamivir is likely to remain. Phase III development of an intravenous formulation by GSK also indicates their ambitions for Zanamivir. These reasons, along with the impending expiry of key patents and the lack of generic competition, make Zanamivir an attractive avenue for generic manufacturers to gain a share of the antiviral market.
Do you want find out more about how we can assist your generic drug development?
Share this article:
To register for GenericsWeb's free monthly newsletter 'INNsight', click here