The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of a generic Raloxifene product for Eli Lillys blockbuster selective oestrogen receptor modulator (SERM) Evista®. Global sales declined 6% in Q1 2013 generating US$240.6 million.
General information
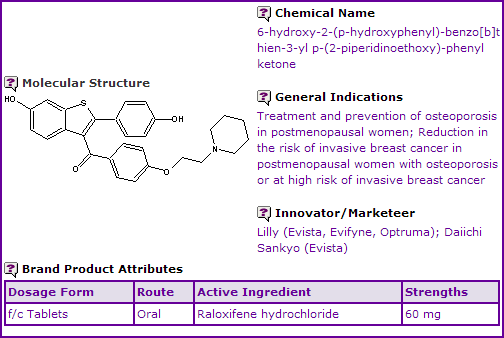
Raloxifene is commercially available in the hydrochloride salt form as an oral film coated tablet in 60 mg strengths for use in the treatment and prevention of osteoporosis in postmenopausal women. In certain markets, including the US and Australia, it is also indicated to reduce the risk of invasive breast cancer in postmenopausal women with osteoporosis or at high risk of invasive breast cancer. Raloxifene was developed and marketed by Elli Lilly (Lilly) who, in July 2006, entered into a marketing and distribution agreement with Daiichi Sankyo for major European markets.
Figure 1: General Information table for Raloxifene
INN Constraint Comment
Patents in the family US24633381A, which protect the Raloxifene molecule and its hydrochloride salt, have expired in all major markets. Patents protecting the molecule are usually considered to be the most significant barrier to generic entry. However, as indicated by the red constraint indicators (see Figure 2), patent protection conveyed by patent families US92093392A and US20501294A protecting methods of use and the dosage regimen for Evista® are considered to constrain generic entry beyond the expiry of patents in the molecule family in this case.
Patent family US92093392A seeks to protect the main indicated use of Evista® in the treatment and prevention of osteoporosis, and is therefore considered to be a constraint to generic entry. While the US members of the family expired in December 2012, the protective term of the European member, EP0584952 (952), has been extended by SPC in France, Germany, Spain and the UK, expiring in early August 2013 (see Figure 3). The French and Spanish designation of 952 has been revoked following nullification actions filed by Teva in the respective countries. The designations being declared invalid on 20 March 2012 by the Tribunal de Grande Instance of Paris and 31 July 2012 by the Pamploma Commercial Court for lack of inventive step. Similarly, patent nullification proceedings have been filed in Germany. In addition, opposition was filed with the EPO by Teva and Tecnimede Sociedade Tecnico-Medicinal to its divisional patent, EP1438957, which was subsequently revoked. This highlights the importance of monitoring and using the EPOs opposition period to revoke weak patents, as opposed to the national litigation route, which comes at much higher cost and often leaves many smaller markets with protection in place. Teva, launched their Raloxifene products in Spain at risk prior to the conclusion of revocation proceedings, triggering Lilly to file an injunction which was ultimately dismissed. In Canada, the allegation of non-infringement and invalidity to CA2101356 was upheld following the filing of an Order of Prohibition by Lilly in response to applications for Notices of Compliances by Novopharm (Teva Canada) and Apotex.
US patent RE39,050 of family US20501294A is considered to further constrain generic entry until March 2014. The patent protects the dosing regimen of administering 60 mg/day Raloxifene hydrochloride in the treatment and prevention of osteoporosis. The patent was upheld follow a paragraph IV challenge brought by Teva and was affirmed after appeal on 1 September 2010. Patent family members in Europe, Canada and Australia were withdrawn prior to grant. The above two cases outlines how vast differences in patent protection across different countries can arise.
Remaining patent families US28017380A, US99522292A, US3002887A, US30823594A, US20491594A and US1416796P are generally not considered to be a constraint to generic competition as the protected technologies are likely to be circumvented.
NCE data exclusivity is generally not considered to be a constraint to generic competition as it has expired, however in the US, Lilly was awarded seven years of orphan data exclusivity pursuant to FDC Act §526(a)(2)(B) for 'cost recovery', where incidence of the disease in the population is 200,000 or more, for the indicated uses of Raloxifene for reducing the risk of invasive breast cancer in postmenopausal women with osteoporosis or at high risk of invasive breast cancer (see Figure 3). This will prevent approval of generic competitors applications for generic Raloxifene equivalents carrying those indications until September 2014.
Litigation Comment
As highlighted above, extensive patent litigation has been identified in the US for patents in the families US92093392A, US20501294A and US1416796P, which relate to authorised uses and administration of Raloxifene. Lilly filed an infringement suit against Teva after they filed an ANDA with paragraph IV certifications to patents listed in the FDA Orange Book. In September2009, the United States District Court of Southern District Indiana (District Court) found the patents USRE39049, USRE38968 and US6906086 of US92093392A protecting Raloxifene hydrochloride for use in the treatment and prevention of osteoporosis, and USRE39050 of US20501294A relating to the administering of a 60 mg/day dose, were upheld as valid and infringed by Teva. Teva appealed the decision with the US Court of Appeals for the Federal Circuit, which affirmed the District Courts decision in September 2010, preventing final FDA approval until March 2014. In regard to patents US6458811, US6797719 and US6894064 from the family US1416796P, which expire in March 2017, claiming Raloxifene hydrochloride having defined particle size and its use, the District Court found them to be invalid for lack of obviousness and lack of enablement.
In Australia, Lilly filed a patent infringement suit against Apotex in May 2012 in the Federal Court of Australia in regard to Australian patent 658075, which claims the use of Raloxifene in the treatment and prevention of osteoporosis. This resulted in Apotex agreeing not to manufacturer, market and sell their Raloxifene product and withdraw applications to list their product on the Pharmaceutical Benefit Scheme until the conclusion of the infringement proceedings.
Of note are the families with priorities US28017380A and US3002887A, both included as 'Key' due to patent infringement litigation brought against Lilly for its manufacture and sale of Evista®. An infringement suit was brought about by Arid in regard to US6410516 of US28017380A, which claims methods of inhibiting gene expression by reducing the activity of the NF-kB transcript pathway. In May 2006, a jury upheld Ariads complaint and found Lilly to have infringed the patent. The decision was overturned on appeal by bench trial, which found the patent to be invalid for lack of inventive step.
In addition, on 23 December 2008, Lilly was found not to have infringed US patent member 5,075,321 of family US3002887A, or to have misappropriated the asserted invention or trade secrets related to the manufacture and sale of Evista®, pursuant to a stipulation entered into with the plaintiffs University of Pennsylvania, Dr Alan Schreiber and ZaBeCor. Although litigation with regard to these patents appears to have reached an end in the US, it is always worthwhile considering whether the innovators may pray on less financially resourced generics companies in markets outside of the US in an attempt to asserting their intellectual property rights, making identification of these potential risks of high importance.
Figure 2: Key Patent Indicator; Key patent families protecting Raloxifene products
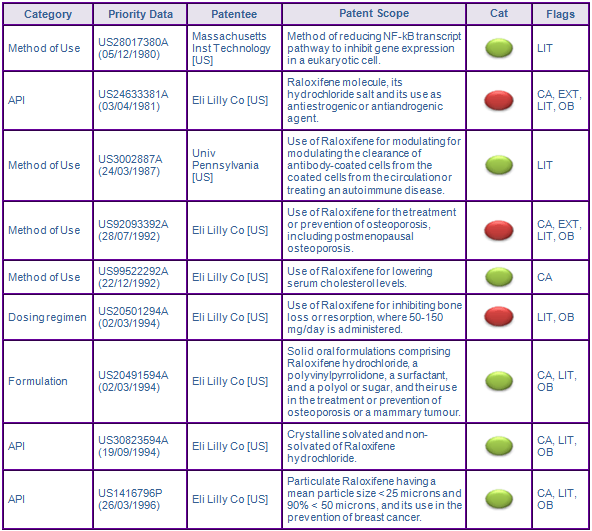
Figure 3: Patent Family View priority application US92093392A
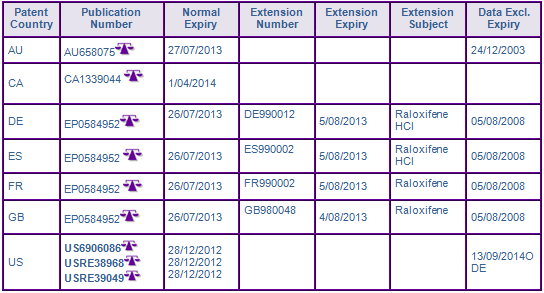 ODE Indicates expiry of 7 year orphan data exclusivity period in the US
Figure 4: Marketing Authorisations for products containing Raloxifene in Key Countries
ODE Indicates expiry of 7 year orphan data exclusivity period in the US
Figure 4: Marketing Authorisations for products containing Raloxifene in Key Countries
Amongst the US approvals Teva and Invagen have secured tentative approvals for their 60 mg Raloxifene hydrochloride tablets. Final FDA approval and launch in the US is expected to following the expiration of dosage regimen patents in March 2014.
The presence of generic Raloxifene products in Europe can be attributed to the revocation of use patents, their perceived invalidity and unenforceability, and soon expiring patent protection. Several companies, including Synthon, Stada Arzneimittel and Pharmathen have been granted market authorisations in the UK, whilst Teva launched their Raloxifene product at risk on the 1 January 2013, after being granted a market authorisation via the centralised procedure on the 29 April 2010. Sandoz, Teva and Biogaran are amongst the generic competitors to file a 'declaration de commercialisation' for their Raloxifene products following the revocation of patents in France.
In other markets, there is a presence of generic Raloxifene products on the market in Canada following invalidation of Canadian patents and the issuance of Notice of Compliance. However due to the ongoing litigation in Australia generic Raloxifene product are currently not available on the ARTG, a situation which may change upon expiry of key patent AU658075 in July 2013.
 Figure 5: Top Patent Applicants
Figure 5: Top Patent Applicants
This figure is a representation of patentees who have filed the most patent families for this INN (Figure 5). Note that Lilly the innovator has 41% of the filing relating to Raloxifene and the prominence of the generic companies Teva, Hexal and Sandoz in the top 5. The appearance of Pfizer in the top 5 can be attributed to general patenting relating to its competing SERM products, Fablyn® (Lasofoxifene) and Conbriza® (Bazedoxifene).
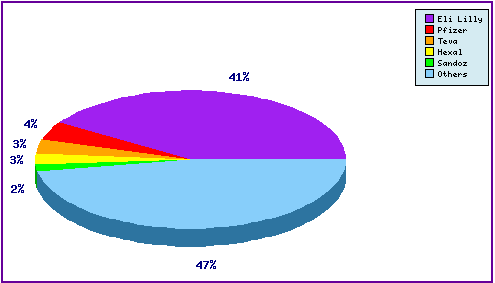 Figure 6: Patent Filing Trend
Figure 6: Patent Filing Trend
The figure below represents the timing of the earliest priority filing date for each patent family identified for this molecule as well as the type of claims found in the applications. Note the large numbers of filings of use patents in the 5 years preceding the launch of Evista®, the majority of which were filed by Lilly. Post launch patent filing relating to formulations represent generic companies trying to circumvent innovator patents and protect their technological developments, to allow launch of generic products after expiry of patents in the constraining families.
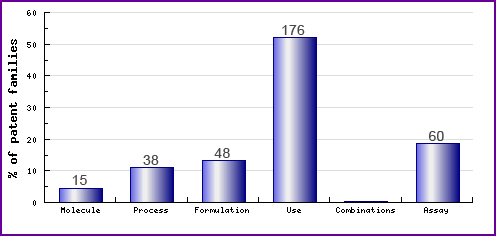
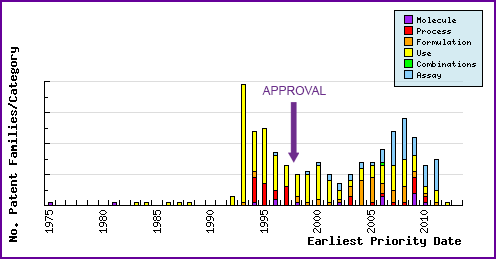 Figure 7: Patent Category Distribution
Figure 7: Patent Category Distribution
Figure 7 represents the types, number and relative distribution of patents that have been filed for Raloxifene obtained via a comprehensive patent search (Pipeline Developer report). Note the prominence of use patents, approximately half which were filed by Lilly, mostly relating to unauthorised uses of Raloxifene. This shows the significant lengths an innovator company goes to, during the development of a drug, protecting their research developments and the potential to extend the indications for Raloxifene. Most of the other use patents relate to general patenting in regard to SERMs or oestrogen agonists.
In summary, patents protecting the Raloxifene molecule and its hydrochloride salt have expired in all major markets. However, patents protecting the main indicated use of Raloxifene in the treatment and prevention of postmenopausal osteoporosis and its dosage regimen have provided some constraint to generic competition beyond the expiry of molecule patents. In the US, these patents have proved to be a barrier to generic competition, after being upheld after paragraph IV challenges by Teva, preventing final FDA approval until March 2014. In Europe declaration of invalidity has allowed generic entry in some markets, but not all. This demonstrates the benefits of method of use patents in maintaining the innovators monopoly beyond molecule patent protection and data exclusivity provisions. In addition to constraining generic entry, it can be costly to generic companies having to delay the launch of their generic products until the conclusion of revocation proceedings, receiving injunction orders restraining them from the manufacture, marketing and/or sale of their generic equivalents, and other penalties awarded for patent infringement. Orphan data exclusivity in the US will prevent approval of ANDAs having the indicated use of Raloxifene in the reduction of invasive breast cancer until September 2014, again demonstrating the validity of life-cycle management strategies based on therapeutic use.
Do you want find out more about how we can assist your generic drug development?
Share this article:
To register for GenericsWeb's free monthly newsletter 'INNsight', click here