The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic equivalents of Boehringer Ingelheims non-nucleoside reverse transcriptase inhibitor (NNRTI) Viramune® (Nevirapine).
General information
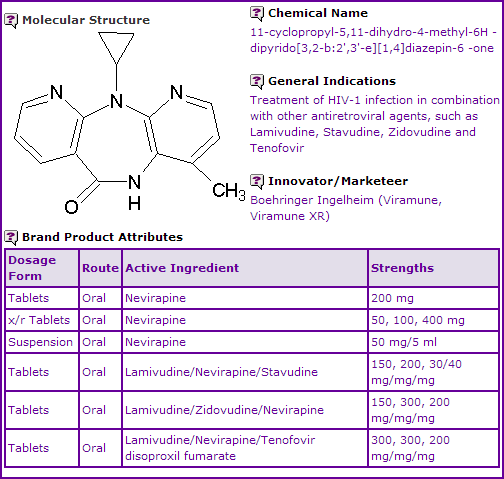
Nevirapine is co-indicated for use with other anti-retrovirals in the treatment of HIV. It is currently available as immediate release (IR) tablets in 200 mg strength, as a 50mg/5mL oral suspension and as extended release (XR) tablets in 50, 100 and 400 mg strengths. US sales of the 200 mg IR tablets generated approximately US$116.6 million for the 12 months ending March 31, 2012, according to IMS Health. Nevirapine was discovered by Boehringer Ingelheim and was the first NNRTI authorised by the FDA for treating HIV-1 infection and AIDs.
Figure 1: General Information table for Nevirapine
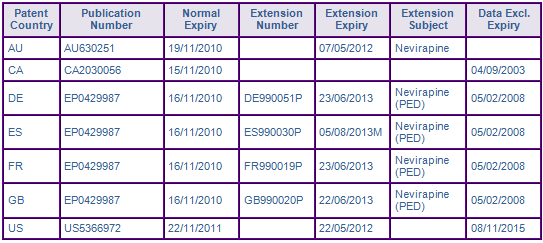
INN Constraint CommentPatent protection for the Nevirapine molecule expired in the US and Australia in May 2012 after enjoying 6 month paediatric extension and 535 days of s70 extension, respectively. However, patent protection is still constraining generic entry in certain European countries after Boehringer Ingelheim successfully complied with an agreed paediatric investigation plan (PIP) and was awarded a 6 month extension to the SPC term (see Figure 3), effectively extending patent protection in Germany, France, and the UK until June 2013 and August 2013 in Spain. Genericswebs proprietary SPC analyser flagged the Spanish SPC, suggesting that the SPC expiry date is based on an incorrect first EU market authorisation date. In this case, the Spanish SPC expiry date, listed in the OEPM registry was based on the EU centralised approval for Nevirapine on 05/02/1998, not the earlier Swiss approval date of 23/12/1997, which was recognised in the EU state of Liechtenstein. The later expiring SPC term in Spain can be attributed to OEPM basing its calculation on the first EU centralised date rather than the earlier Swiss authorisation date used by other IPOs in their calculations. Those wishing to launch generic Nevirapine in Spain should verify the status and expiry of the SPC with OEPM prior to launch.
Patent families US5680397P and US94276507P protect oral formulations for Nevirapine. US5680397P claims an oral suspension formulation and a method of preparing Nevirapine hemihydrate. The family was identified through the inclusion of Mexican patents listed under article 47-bis of the Regulation of the Law of Industrial Property (data for Mexico and over 40 other countries are available for these key patent with an extended Pipeline Developer subscription). The list was established to help COFEPRIS identify any intellectual property effecting the market authorisation of a drug, so as not to infringe any patentees rights. In January 2010 the Mexican Supreme Court allowed the inclusion of compositions, formulation, and uses in the list prior to this decision only API patents where included. However, the family is not considered to be a constraint to generic competition as the protected technology is likely to be circumvented by reformulation.
US94276507P seeks to protect the dissolution profile of the XR dosage form. However, in Canada, the only country to have granted a patent in the family so far, the claims are restricted to a formulation containing specific excipients, thus only protects the 400mg. The file wrapper for the US equivalent suggest the claims will have similar coverage, however the European application is yet to undergo examination, hence the XR dissolution profile claims remain in the application. Whilst patent protection is not considered to be a constraint in the US and Canada, as the claims are likely to be circumvented there is some uncertainty with regard to the future scope of the claims in Europe, which may deter some companies from developing the XR dosage form. In the US, a further barrier to entry is the 3 years of data exclusivity awarded to new dosage forms, will block generic competition for the XR dosage form until 2014, and 2015 for indications covering the paediatric population.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Nevirapine
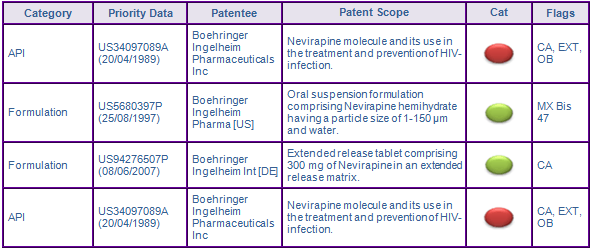
Figure 3: Patent Family View priority application US34097089A
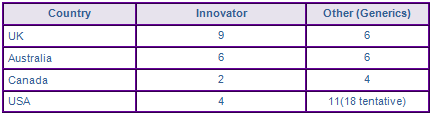
Amongst the US approvals, Apotex, Aurobindo, Cipla, Strides, Hetero Labs, Prinston, Mylan, Micro Labs and Sciegen were awarded market authorisations for the 200 mg IR tablet upon patent expiry in May 2012. Aurobindo was also the only generic company to be awarded a market authorisation for the oral suspension formulation. Tentative approvals have been awarded to generic companies for their fixed dose combinations with Lamivudine and Stavudine, Lamivudine and Zidovudine and co-packaged 200 mg IR Nevirapine tablets with a Lamivudine and Zidovudine or Lamivudine and Tenofovir disoproxil co-formulated tablets. These have been granted by the 505(b)2 pathway reviewed under the Presidents Emergency Plan for AIDS Relief (PERFAR). Under the PERFAR program a tentative approval means that the product meets all the requirements of the FDA but may not be marketed in the US due to patent protection or other regulatory exclusivities but may be purchased outside the US in certain developing countries.
In the UK, generic authorisations have been awarded to Mylan, Hetero, Disphar, Aurobindo (Milpharm) and Morningside healthcare for the 200 mg IR formulation. Teva have also received a market authorisation for the 200 mg IR tablets via the centralised procedure. Generic authorisations were also awarded to Aurobindo and Teva for the 200 mg IR tablets following the expiry of the API patent in Canada in November 2010.
These generic approvals are indicative of the likely competition Nevirapine will face across Europe upon Paediatric extension expiry, however no approvals for the XR dosage form have been identified. In addition to any patent/IP barriers identified above, this is likely because the Viramune XR product was launched in 2011 and generic companies have not had sufficient time to develop an equivalent based on the reference product.
Figure 4: Marketing Authorisations for products containing Nevirapine in Key Countries
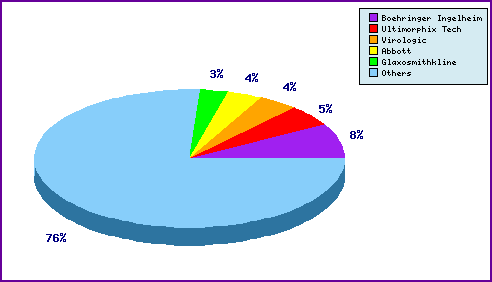
This chart shows a representation of patentees who have filed the most patent families for this INN. Overall there are over 190 individual filers. Amongst the top 5 filers are the innovator Boehringer Ingelheim and other innovator companies such as Abbott (Ritonavir/Lopinavir) and Glaxosmithkline (Efavirenz), whose presence can be attributed to general patenting regarding their anti-retroviral agents.
Figure 5: Top Patent Applicants
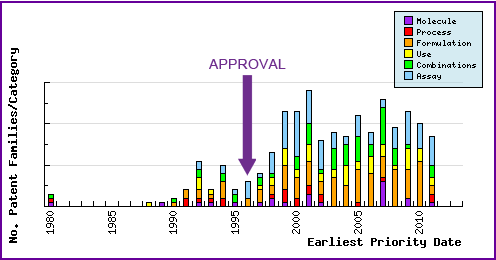
This graphic represents the timing of the earliest priority filing date for each patent family identified for this molecule as well as the type of claims found in the applications. Note the strong responsive patenting by companies post-launch, particular in the area of formulations and combinations.
Figure 6: Patent Filing Trend
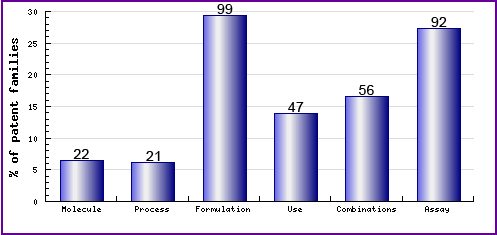
This chart shows the types, number and relative distribution of patents that have been filed for Nevirapine obtained via a comprehensive patent search (Pipeline Developer report). Note the dominance of formulation patents (see Figure 7), the majority relate to general formulating markedly for oral formulations. Combination patents filed relate to the use and co-formulation of combinations of anti-retrovirals. Generic competitors should carefully consider these patents when looking to develop and launch their own co-formulated or co-packet products. The presence of relatively high activity in regard to the molecule can be attributed to the common indicated use of anti-retrovirals, such as Nevirapine, used in combination with other anti-retrovirals for the treatment of HIV. Nevirapine is generally given as a treatment option in patients with lower CD4+ cell counts and is also administered in combination with other antiretroviral, so to ensure the best treatment option is selected for an individual, patient suitability type assay patents have been of the interest of many applicants.
Figure 7: Patent Category Distribution
In summary, patent protection for the Nevirapine molecule has expired in Australia, Canada and the US leading to generic entry for the Nevirapine 200 mg IR tablet and the oral suspension formulation in those countries. However, patent protection remains in force in most major European markets due the granting of a 6 month paediatric extension to the SPC. Several generic companies have gained market authorisations for the 200 mg IR tablets in the UK, this is the indicative of the generic competition Viramune will face follow patent expiration. Late stage lifecycle management by the innovator by way of launching Viramune XR and programs to promote patient use of the dosage form may ease generic pressure due to loss of patent protection for other Nevirapine products, but only for a short amount of time. Depending how the sustained release profile patent claims are handled by the EPO examiner, the XR product may be protected in Europe for much longer. Other generic companies will take advantage of patent expiries to develop and launch their own combination products.
Do you want find out more about how we can assist your generic drug development?
Share this article:
To register for GenericsWeb's free monthly newsletter 'INNsight', click here