|
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
Drug In Focus: Tolterodine
The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic equivalents of Pfizers blockbuster antimuscarinic agent Tolterodine, indicated for the treatment of overactive bladder and associated symptoms. Pfizer reported total global sales in 2010 of US $1bn, a revenue decline of 12% compared to 2009.
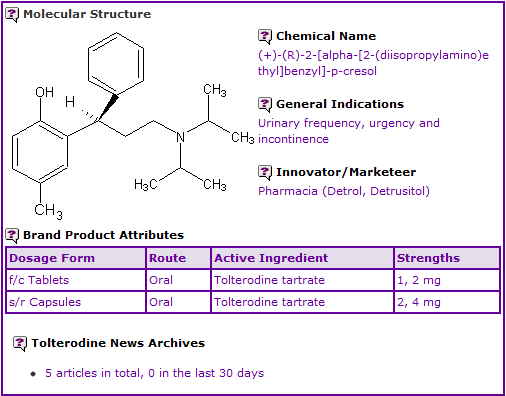
General information
Tolterodine is currently available in film coated oral tablet and sustained-release oral capsule formulations.
Figure 1: General Information table for Tolterodine
INN Constraint Comment
At the time of publication, patent protection of the Tolterodine molecule in most major territories either remains in force or has been extended by Supplementary Protection Certificates (SPCs) by virtue of family SE8800207A. Loss of protection will occur from 2012 to 2015 in Core territories, Canada being the latest due to a 17 years from grant expiry calculation (See Figure 3).
A second patent family, SE9203318A, is identified as a result of an FDA Orange Book listing for patent US 5,559,269 which was subsequently re-issued as US RE39,667 and then as US RE40,851, neither of which were updated in the Orange Book. These patents claim Tolterodine metabolites, and the use of Tolterodine in their multi-step synthesis and as such this family is not considered constraining to the launch of generic Tolterodine products.
The third patent family, SE9802864A, is derived from four separate PCT applications and the patents have expiries of 2019 in most major territories. This family, containing FDA Orange Book and Canadian Patent Register listed patents, relates to the controlled/sustained-release oral Tolterodine formulations having specific release characteristics and their use in the treatment of urinary incontinence. Patents in this family are the subject of litigation in the US and opposition in Europe by generic competitors and therefore may be considered a strong constraining factor for the prevention of generic sustained-release formulations of Tolterodine. A broad European patent in this family has recently been granted and is still within the opposition period. It is unlikely to proceed unopposed by major generic manufacturers as it appears to be the only major constraint to generic sustained release Tolterodine products in Europe.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Tolterodine
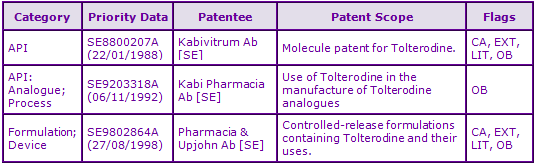
Figure 3: Patent Family View priority application SE8800207A
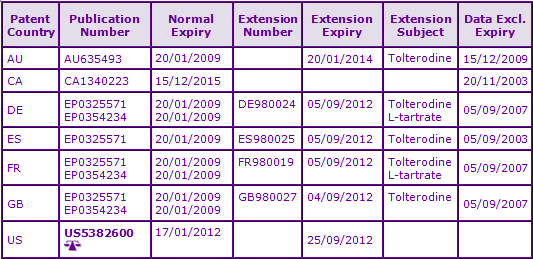
Tentative FDA approvals for generic immediate-release oral tablet formulations have been granted to Ranbaxy and Sandoz. Several Paragraph IV filings have been identified (Impax, Mylan, Teva, Sandoz), relating to both the immediate-release and sustained-release formulations. In the UK, several generic marketing authorisation holders have been identified (Caduceus, Hexal, Ratiopharm, Sandoz and Teva) however none relates to the sustained-release formulations (See Figure 4).
Figure 4: Marketing Authorisations for products containing Tolterodine in Key Countries

Figure 5: Top Patent Applicants
Represents patentees who have filed the most patent families for this INN. Note the prominence of generic players in the top five e.g. tentative FDA approval holders Ranbaxy And Sandoz (LEK) as well as the diversity of applicants represented by the large portion outside the top five.
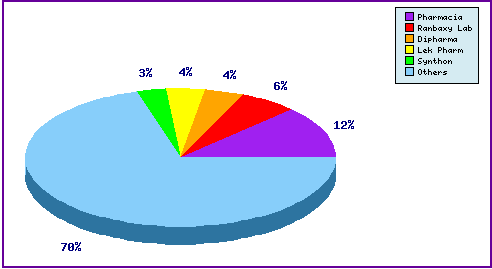
Figure 6: Patent Filing Trend
Represents the timing of the earliest priority filing date for each patent family identified for this molecule as well as the type of claims found in the applications. Since the approval of Tolterodine (1998 in the US/UK) there has been consistent patent activity in the area of molecular form and process. Furthermore, since the subsequent approval of the sustained-release formulation (2000/2001 in the US/UK) several generic companies have submitted patent filings relating to modified-release formulations.
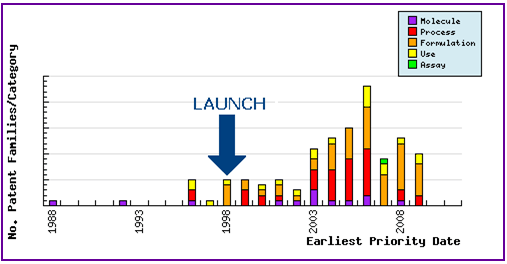
Figure 7: Patent Category Distribution
The types, number and relative distribution of patents that have been filed for Tolterodine obtained via a comprehensive patent search (Pipeline Developer report). Of the formulation patents identified, the majority specifically relates to oral modified-release formulations, however interest has been identified in other administration techniques, including transdermal formulations.
In summary, the molecule patent in force in most major countries represents a barrier to generic competition; however the expiry of this molecule protection from September 2012 in Europe and the US means that generic competition, at least to the immediate-release formulations of Tolterodine, is likely to be very strong as evidenced by the generic marketing authorisation holders in the UK and the Paragraph IV filings in the US.
Generic sustained-release formulations will likely be prevented until the end of the decade as a result of protection of the release profile, however this depends on the outcome of litigation in the US concerning the Detrol LA formulations which are currently on-going, and whether the key European patent is successfully opposed.
Do you want find out more about how we can assist your generic drug development?
Share this article:
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
|