|
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
Drug In Focus: Pramipexole
The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic equivalents of Boehringer Ingelheims blockbuster dopamine agonist Pramipexole which is indicated for the treatment Parkinson's Disease and Restless Legs Syndrome.
General information
An analysis of the currently marketed products containing Pramipexole shows that it is currently available as film-coated and extended-release oral tablets.
Figure 1: General Information table for Pramipexole
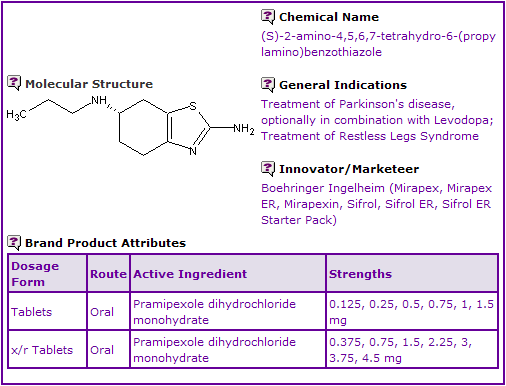
INN Constraint Comment
Ordinarily, the most significant patent family which may have prevented the launch of generics in certain countries would be the first API family (See Figure 2). This family contains patents with term extensions in several territories however all but one of these extensions has now expired; an SPC in Spain relating to a process patent still being valid until June 2012 (See Figure 3). Similarly, patents in the second family relate to Pramipexole transdermal therapeutic systems and have also now expired.
The third family with patent expiries expected in January 2018 may be considered to be partially constraining as it protects the use of Pramipexole in the treatment of Restless Legs Syndrome, which is one of the two authorised indications for the instant release dosage form. The extended-release formulation appears to only be indicated for the treatment of Parkinson's disease.
Boehringer Ingelheim reported a 16.6% reduction in sales (Euros) for Mirapex/Sifrol over the period 2009 to 2010; however in 2010 the Mirapex extended-release formulation received FDA approval and as a result the extended-release formulations may now be the focus for both innovator and generic companies alike due to the potential increased adherence to therapy. The final patent family listed relates to these extended-release oral formulations comprising Pramipexole with expiries of July 2025 in most countries and April 2028 in the US following an extensive patent term administrative adjustment. At this point, no generic activity has been identified for the extended-release formulations, however in the US there exists a Paragraph IV listing relating to the Mirapex ER formulations (see Figure 4).
Data exclusivity for Pramipexole has expired in almost all territories and as such is not a factor when considering generic entry. In the US, the Mirapex ER formulations have exclusivities until early 2013 for the new dosage form and treatment of signs and symptoms of advanced idiopathic Parkinson's Disease indication which are likely to prevent generic equivalents until their expiration.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Pramipexole
Figure 3: Patent Family View priority application DE3447075A
*The term of US 4,886,812 was originally awarded an s156 extension for Mirapex of 1,564 days, extending expiry to 25 March 2011; however this patent was subsequently the subject of a terminal disclaimer limiting its expiry to 8 October 2010, which corresponds to an equivalent 1,564 day extension to US 4,843,086.
Figure 4: US Paragraph IV filings for Pramipexole
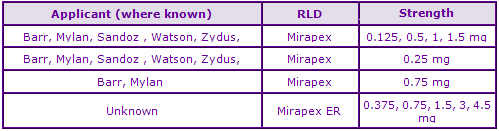
Figure 5: Marketing Authorisations for products containing Pramipexole in Key Countries
Marketing Authorisations approved in the UK since 2009 suggest that generic competition will be strong however, at the time of publication, no UK generics have been launched. The Canadian and US data highlights the presence of a large number of available generics.
*Data taken from UK electronic Medicines Compendium (eMC).
Figure 6: Patent Filing Trends
Represents the timing of the earliest priority filing date for each patent family identified for this molecule as well as the type of claims found in the applications. Note the consistent patenting by generic companies post-approval (1997 for Mirapex in the US), in the area of process, use and formulation.
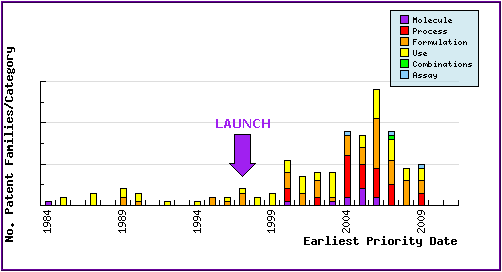
Figure 7: Patent Category Distribution
The types, number and relative distribution of patents that have been filed for Pramipexole obtained via a comprehensive patent search (Pipeline Developer report). Note that there exists a roughly equal number of use and formulation patents. Of the use patents identified, roughly 10% quote Boehringer Ingelheim as the patentee and relate to Parkinsons Disease, whilst of the formulation patent families identified, several generic companies (including Mylan, Alembic and Dr Reddys) are quoted as patentees in inventions relating to extended or prolonged-release dosage forms containing Pramipexole.
In summary, the large number of marketing authorisation holders identified in the UK and the number of generics available in Canada and the US is indicative of the interest shown by generic companies for Pramipexole. Most generics identified relate to the immediate-release formulations, however as yet no generics have been identified in the UK or Australia. The existence of Paragraph IV filings in the US corresponding to Mirapex ER highlights the current existence of interest in such formulations, even though the key patent family protecting these extended-release formulations is yet to remain in force for several years.
Do you want find out more about how we can assist your generic drug development?
Share this article:
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
|