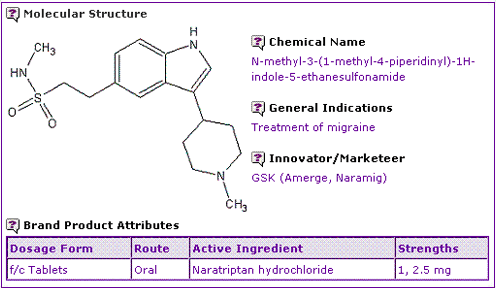
The Naratriptan General Information (Table 1) shows that only one oral dosage form, with the strengths of 1 and 2.5 mg is available in the form of film coated tablets. This dosage form contains Naratriptan hydrochloride, which is indicated for the treatment of migraine headaches.
Only one key patent family, which protects the Naratriptan active ingredient, has been identified by the Key Patent Indicator (Table 2). Within this family US5066600 claiming analogues of Naratriptan, already expired in November 2008 while US4997841 (841) claiming the Naratriptan active ingredient, salts, oral dosage forms with the strength of 0.1 to 100 mg and use for the treatment of migraine, will expire in early July 2010 due to the grant of a s156 patent term extension in January 2001. It can be seen from the Drug Master File (DMF) section of the Naratriptan Selector report that several generic companies are already prepared to supply Naratriptan hydrochloride manufactured in India to the US market once the 841 patent has expired.
Table 2: Naratriptan Key Patent Indicator
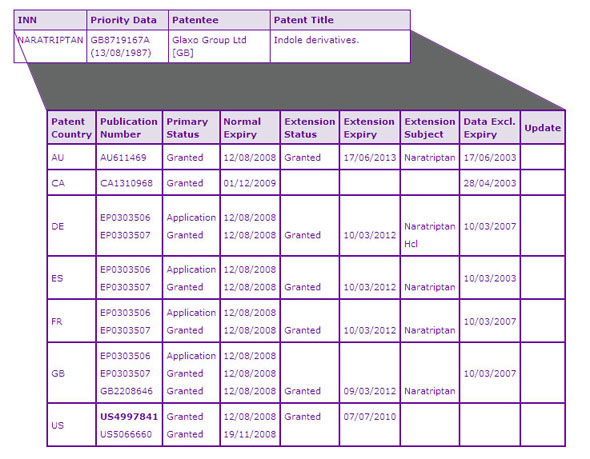
The term of the equivalent patents in this family, EP0303057 in Germany, France, and Spain, and GB2208646 in the UK has been extended through SPC until March 2012, therefore they provide much longer patent protection of the active ingredient in these countries compared to the US. However, the term of the equivalent patent in other European countries such as Croatia, Hungary, Ireland, Italy, Poland, Romania and Slovakia already expired due to no application of SPC or an absence of patent term extension provisions at the time of product launch. This provides a convenient springboard for the manufacturers located in these countries who may want to enter the major European and/or the US market.
In Australia the term of the equivalent patent has been extended through s70 extension provision until June 2013 while the Canadian equivalent, CA1310968, expired in December 2009 due to an absence of pharmaceutical patent term extension provisions. It can be seen from the Regulatory Information section that diligent generic companies such as Sandoz and Novopharm have already taken advantage of this opportunity in Canada and launched their generic versions on the next day of the expiry of this patent. Using the geographical closeness, these companies are likely to be the major candidates to penetrate the US market upon the expiry of the equivalent US patent. This is confirmed by the fact that Sandoz has recently obtained a tentative ANDA approval on this product.
Unlike the case of Eletriptan which is marketed by Pfizer, there do not appear to be any specific innovator patents protecting the polymorphic forms for Naratriptan. This suggests that GlaxoSmithKline has not been proactive in trying to extend and/or manage the lifecycle of this product, as such it will likely lose significant market share to a number of generic manufacturers upon the expiry of the patent protecting the active ingredient, and therefore significant price erosion is likely to occur where competition is a factor.
It can be seen from the Data Exclusivity column of Table 2 that the data or market exclusivity of the Naratriptan product expired more than 3 years ago in all countries. Therefore, this type of protection on the Naratriptan product will not constrain the launch of a generic version of this product.
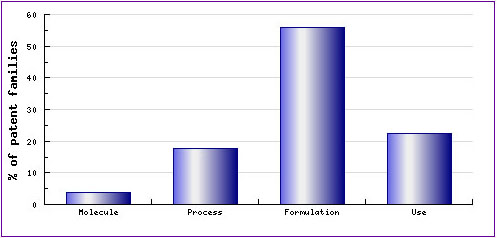
The question for generic companies would be whether they can launch a generic version of the Naratriptan product in the US market as soon as the US patent protecting the active ingredient expires since no data exclusivity exists and there is no patent protecting the polymorphic forms. The answer is not so simple because there could be other patents which are not identified as key patents but still protect the Naratriptan product from different perspectives such as processes for preparing the active ingredient, formulations or uses thereof. As can be seen from the Patent Category Distribution (Figure 1), there are a significant number of patent applications relating to formulations, occupying more than half of the total applications. According to information accessible from the corresponding Pipeline Developer report, most of them are related to an oral or general formulation which could further delay the launch of a generic version well after the expiry of the US patent protecting the active ingredient.
The question for generic companies in this situation would be whether they can launch a generic version of the Naratriptan product in the US market as soon as the US patent protecting the active ingredient expires since no data exclusivity exists and no patent protecting the polymorphic forms? The answer is not that simple because there could be other patents which are not identified as key patents but still protect the Naratriptan product from different perspectives such as processes for preparing the active ingredient, formulations or uses thereof. As can be seen from the Patent Category Distribution (Figure 1), there are a significant number of patent applications relating to formulations, occupying more than half of the total applications. According to information accessible from the corresponding Pipeline Developer report, most of them are related to an oral or general formulation which could further delay the launch of a generic version well after the expiry of the US patent protecting the active ingredient.
Figure 1: Naratriptan Patent Category Distribution
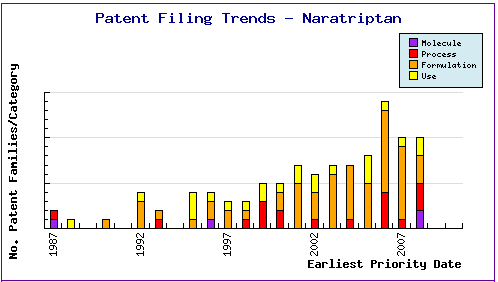
Patent Filing Trends (Figure 2) reconfirms that most patent applications since the major launch of this product in Europe in 1997 and in the US in 1998 have been focused on the formulations. The patenting activity around that period could be attributed to the innovator while the gradual increase of activity since then and the sharp increase in 2006 suggest that generic companies have become interested in developing and protecting their formulation for this product. This analysis can be confirmed by information accessible by subscribing to the corresponding Pipeline Developer report.
Figure 2: Naratriptan Patent Filing Trends
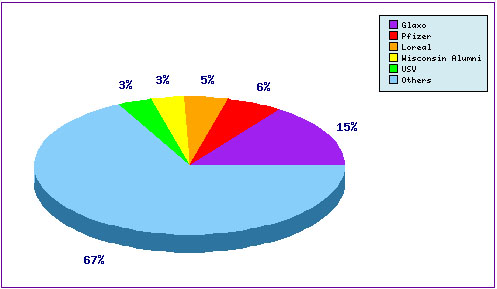
Generic interest on the Naratriptan product is further confirmed by the Top Patent Applicants (Figure 3) which shows that over 70% of the patent applicants are small generic companies while the innovator occupies 15% of the total applications. One of the generic companies who express a great interest on this product is USV who occupies 3% of the total patent applications and appears as one of the six DMF applicants.
Figure 3: Naratriptan Top Patent Applicants
In conclusion, the impending expiry of the US patent protecting the Naratriptan active ingredient will provide an opportunity to generic companies. Some companies appear to be in an advanced stage to launch their generic version in the US market by taking advantage of the less proactive approach in managing the lifecycle of this product by the innovator. Before making a decision to launch, however, they should carefully monitor a great deal of formulation related patents existing on this product, which potentially expire well after the expiry of the patent protecting the active ingredient.
Generic developers are strongly advised that a comprehensive search of the patent landscape surrounding Naratriptan is a crucial step before any technical development takes place, in order to reduce the risk of costly litigation. Obviously, this would not be possible without first comprehensively identifying relevant patents from the 50 million+ published patent documents worldwide.
Comprehensive data for patent families relating to Naratriptan, based on professional patent searching, may be accessed by subscribing to the GenericsWeb Pipeline Developer report, which includes 12 monthly updates, to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
For questions and comments about this article please contact me at d.oh@genericsweb.com.
To register for GenericsWeb's free monthly newsletter 'INNsight', click here