CPhI India how does it compare?
A couple of months ago I wrote about my impressions of CPhI in Madrid and my feeling that it had grown too big. Last month, I attended CPhI India in Mumbai and these are a few personal impressions of the event and how it compared to the one a couple of months earlier in Madrid.
Sitting here and looking out over a quiet, snow-covered London, it is difficult to remember the warmth and vibrancy and noise of Mumbai, but here are a few impressions that have stuck in my memory. I went expecting CPhI India to be different and of course it was, while nevertheless retaining many of the elements of the European version.
The first impression was surprise at seeing how rough and uneven the surfaces were both inside and outside the event. Some attempt was made to disguise them with carpets but even so it was easy to trip over humps made by electricity cables under the carpets while walking around the event and I certainly managed to do so. A British Health & Safety inspector would not have been amused.
The bigger surprise, though, was at how small the CPhI part of the exhibition was compared to Madrid. In October, the event filled 7 halls of which 6 were CPhI with one shared by ICSE, P-MEC and BioPh whereas all the CPhI exhibitors in Mumbia were concentrated into one hall. The P-MEC part by contrast filled two halls, but that was because many of the exhibitors were displaying fully functioning products lines in massive stands that sprawled over a very large area. The display was actually fascinating to watch and very impressive, but probably more difficult to achieve at the European version because of the much larger number of exhibitors and presumably a much higher cost per m2 of exhibition space.
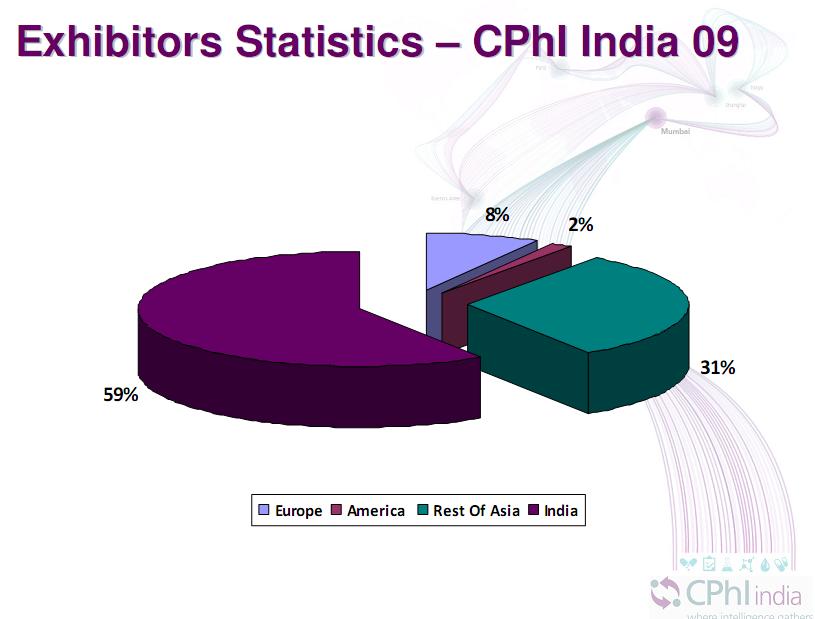
In total, there were 752 exhibitors, around one third of the number that exhibited in Madrid. The graphic above is taken from a presentation made by Mr. M. Gandhi, Managing Director of the event at the Indian PharmaSummit.
The Rest of Asia category was almost exclusively Chinese exhibitors who were the second largest presence after local Indian companies. Chinese companies seem very keen to turn the tables on India by copying the Indian model of pile it high and sell it cheap that India used so successfully to penetrate Western markets. The difference is that the Chinese are also taking the battle into Indias home territory and appears to be seeking and taking an increasing share of the Indian domestic market for intermediates and API building blocks.
In so far as is it possible to compare the European and Indian versions, here are a few statistics.
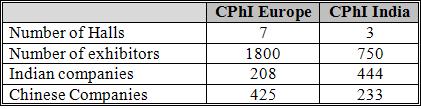
It was also interesting to see the roughly two hundred Indian companies that exhibited in Mumbai who had not made it to Madrid. Out of these companies that are perhaps still in their infancy and keen to copy the successes of their compatriots such as Dr.Reddys, Sun and CIPLA some of tomorrows industry leaders will emerge, but they will find it tough going.
Many of them told me that they aspire to obtain EU-GMP approval from the MHRA or other EU regulators in the hope that this will unlock the European marketplace for them. This marketplace though is now much tougher and more competitive than it was when Ranbaxy, Dr. Reddys and others first entered it. At that time, their main obstacle was overcoming the prejudice and perception that Indian quality was somehow rather doubtful and unreliable. Prices were still relatively high in Germany and the UK, but countries such as France, Italy and Spain had plenty of branded generics on the market, and little appetite for unbranded generics.
Now, however, the atmosphere has changed and while these countries represent a significant growth opportunity as generic penetration increases, they are clamping down on costs with tight price controls, reference price schemes and competitive tendering for generics. It is a tough environment, but some of the companies that exhibited in Mumbai will eventually make the leap into Europe and the US, although they will only succeed if they mentally prepare themselves for a tough fight.

