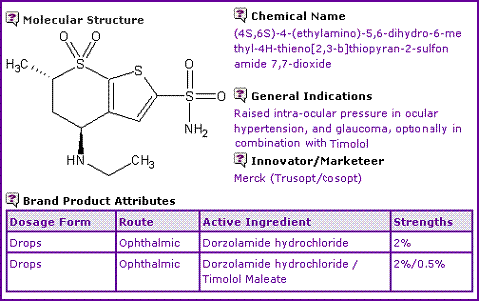
Table 1 shows the General Information for Dorzolamide extracted from Genericsweb's Pipeline Selector report. It indicates the sole dosage form for both Dorzolamide based products is opthalmic drops.
Table 2: Dorzolamide Key Patent Indicator
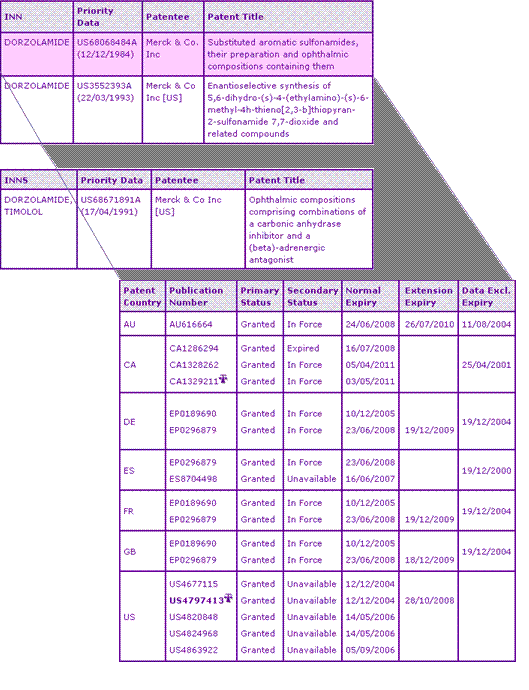
The Dorzolamide Key Patent Indicator (KPI) in Table 2 shows three patent families. Patents in the first family protect the Dorzolamide molecule. Even with an extension for US4797413, all US members of this family have now expired, more than 12 months previous to some of their European equivalents. This is against the trend, often commented upon and critisised, that US patents are afforded longer terms than their equivalents in other territories.
The British, German and French patents in this family are due to expire this month. AU616664 was originally due to expire in June 2008 but, due to a granted s70 extension, will now expire in July 2010. The extension of AU616664 only provided an additional two years of protection out of a maximum of five years, due to the first marketing authorisation for a Dorzolamide product in Australia being July 1995. Canada has the latest expiring member of the family with CA1329211 being valid until May 2011 due to its term being calculated from the issue date, however there is litigation surrounding this patent which will be discussed in more detail later. Litigation aside, fortunately for generic competitors in Canada, due to the absence of a patent extension scheme, the patent applicant will not be able to extend the life of this patent any further.
The second KPI family protects a general enantioselective synthesis method for Dorzolamide. Australian, Canadian, British and several European members of this family all expire uniformly in March 2014, while US5688968 expires in November 2014. Although this family will afford some protection for the preparation of Dorzolamide, generic competitors keen to enter the market could find alternative means of synthesis to circumvent this patent family all together. This family has been included in the Selector report as a key patent family due to an SPC application made in Hungary citing HU217363, implying that the commercial product is protected by this patent. Such methodology for identifying key patents, based on a range of different criteria, ensures the identification of more relevant 'key' patents than can be identified using sources such as Orange Book alone.
The final KPI family protects the combination of carbonic anhydrase inhibitor and a beta blocker. Granted SPCs in several European territories and an s70 extension in Australia protect the Cosopt combination product until March and August 2013 respectively. However, with litigation occurring at least in the UK, US and Canada, the patent landscape surrounding the Cosopt combination product is in a state of flux. In the US and Canada litigation also involved US4797413 and CA1329211 from the first KPI family.
The Pipeline Selector litigation alerts show that in October 2005, Hi-Tech Pharmacal filed the first ANDA for a generic version of the combination product Cosopt with paragraph IV certifications to the Orange Book-listed patents US4797413, US6248735 and US6316443. In January 2006, Merck sued Hi-Tech, asserting infringement of the US4797413 patent, protecting the molecule, but not the US6248735 and US6316443 patents protecting the combination. In April 2006, Merck filed a statutory disclaimer of the US6248735 and US6316443 patents with the USPTO, effectively dedicating its rights under the patents to the public. Shortly thereafter Merck wrote to the FDA and requested that the agency de-list those two patents from the Orange Book.
In October 2006, Merck was notified that Apotex had also filed an ANDA for a generic version of both Trusopt and Cosopt with paragraph IV certifications to the same three Orange Book-listed patents. In response, Merck sued Apotex in December 2006, again asserting infringement of the US4797413 patent (but not US6248735 and US6316443 for obvious reasons). Apotex agreed to be bound by the court decision on the US4797413 patent in the Hi-Tech case, but pursued declaratory judgment counterclaims of invalidity of the disclaimed patents.
The March 2007 ruling of the Federal Circuit in the Hi-Tech case affirmed, in Merck's favour, the patent term of the US4797413 patent with the expiry date of October 2008. Apotex's remaining declaratory judgment counterclaims of invalidity of the disclaimed patents were also dismissed on the basis that Merck had disclaimed both US6248735 and US6316443 patents.
The major outcome of this litigation in the US was Merck's formal disclaiming of the US6248735 and US6316443 patents. In doing so they could no longer enforce any claims from these patents, allowing generic competition into the US market after expiry of the US4797413 patent in 2008, three years earlier than what otherwise might have been the case, given the two US members in the family (US6248735 and US6316443) were due to expire in April 2011 (assuming the patents would be upheld in court). Parallel litigation is also currently before the Canadian courts, involving CA1329211 (protecting the molecule) and CA2065965 (protecting the combination). Since the expiry dates for these Canadian patents are May 2011 and April 2012 respectively the pending result will be eagerly anticipated by generics companies.
In the UK, the expiry of GB0296879 this month removed the molecule patent barrier, and Teva recently attacked GB0509752, protecting the Cosopt combination product. They sought revocation of Merck's patent based on obviousness and the court found in favour of Teva. For a full explanation of the decision and it's ramifications please refer to Duncan Curley's feature article in this month's INNsight 'UK Patents Court Opens Its Eyes to an Obviousness Claim'.
Given the expiry of key patents in the US has already occurred it's not surprising to find that the Pipeline Selector shows there are six other marketing authorisation holders in the US besides the innovator. All six competitors hold authorisations for both the single active and the combination product. Of the six competitors in the US the two biggest, Teva and Sandoz, also have market authorisations in the UK, since in the UK marketing authorisations may be granted prior to the expiry of the molecule patent, leaving the sponsor to choose when to market the product. This signals their intentions to enter that market as soon as possible and with the recent result in the UK courts regarding the patent protecting the combination product as discussed above, other generics companies may soon follow suit.
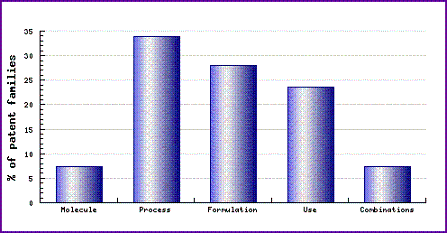
The Patent Risk Analysis section of the Dorzolamide Pipeline Selector report (Figure 1), based on comprehensive patent information (details of which are available by subscribing the corresponding Pipeline Developer report from GenericsWeb), indicates that the patenting activity relating to this drug has focused on methods of API synthesis, with one third of the total number of Dorzolamide related patent families falling into this category. Since the second KPI family, discussed above, protects synthesis of Dorzolamide it is not surprising to find a lot of activity in this category as other companies attempt to find their own method of producing the active ingredient.
Figure 1: Dorzolamide Patent Category Distribution
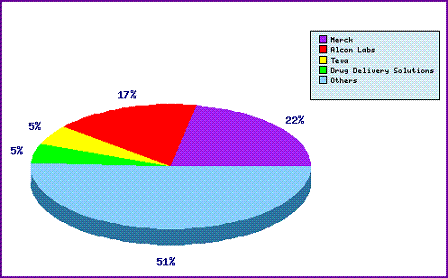
This is further confirmed by the Top Patent Applicants chart (Figure 2) which shows over half the patent applicants are small generics competitors while Teva is within the top five of filers for patent relating to Dorzolamide, likely as a result of holding US marketing authorisations and protecting its market share. Alcon is the second largest patent filer behind the innovator Merck. This is most likely due the fact Alcon, who specialise in opthalmic drugs, were the innovators of Brinzolamide, another carbonic anhydrase inhibitor, thereby resulting an overlap of patents generally claiming this class of active ingredient.
Figure 2: Dorzolamide Top Patent Applicants Analysis
In summary, the market for Dorzolamide has already been opened to generic competition in the US as a result of proactive generic companies, despite their actual loss in the courts. Several other companies have since seized the opportunities for marketing Dorzolamide based products and have taken steps to protect their place in the market. With their experience in the US these generics companies will be well placed to enter the markets of other territories as key patents begin to expire.
The possibilities of identifying a novel polymorphic form, developing alternative synthetic routes and methods of formulation all provide an excellent opportunity to review the patent landscape of Dorzolamide and develop strategies to fill any possible gaps and circumvent the patent applications filed by the innovator and block other generic competitors from doing the same. Several significant markets have not yet been open to generic competition however that is soon to change. Generic developers are strongly advised that a comprehensive search of the patent landscape laid around Dorzolamide is a crucial step before any technical development takes place in order to avoid costly litigation, particularly in regard to routes of synthesis. Obviously, this would not be possible without first comprehensively identifying relevant patents from the over 50 million published patent documents worldwide, and keeping this information up to date.
Comprehensive data for patent families relating to Dorzolamide, based on professional patent searching, may be accessed by subscribing to the GenericsWeb Pipeline Developer report which includes twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
For questions and comments about this article please contact the author at s.robinson@genericsweb.com