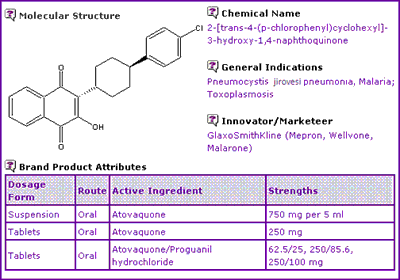
Subscribers to the Selector reports will notice that the data exclusivity information is now conveniently located within the Key Patent Indicator (KPI) (Table 2). This allows data exclusivity and patent expiry dates, including any extensions, to be viewed side by side on a country by country basis..
Table 2: Atovaquone Key Patent Indicator
This shows that data exclusivity for Atovaquone has expired in major territories such as Australia, Canada, Great Britain and the US. Interestingly in all four of these territories GSK is still the only marketing authorisation holder. The absence of a generics company holding a marketing authorisation when no data exclusivity exists may indicate patent constraints, possibly among other factors.
At this stage the KPI (Table 2) has identified 4 patent families critical to the production of Atovaquone. The active ingredient has been protected in the earliest of these with the other three protecting the use of Atovaquone for Pneumocystis carinii (jiroveci) infections, a specific composition of Atovaquone and use of Atovaquone in combination with Proguanil hydrochloride.
Patents from all the major territories in the first family, relating to the Atovaquone molecule, are due to expire in April this year with the exception of US5175319. The original expiry dates in Great Britain, France and Germany were extended from 2004 due to a 1768/92 (SPC) extension and the US5053432 patent was extended from 2008 by a 6 month paediatric extension. The remaining US patent identified by the KPI, US5175319, which was not listed in the Orange Book, expires in December this year and covers a key intermediate used in the preparation of Atovaquone. This patent may possibly present problems for a generic competitor seeking to launch Atovaquone in the US prior to expiry of this patent if the claimed intermediate is present in the generic product as an impurity.
The second KPI family, protecting the use of Atovaquone for Pneumocystis carinii (jiroveci) infections i.e. the sole indicated use for the Mepron product, are due to expire in Europe in August 2009 and during 2010 and 2011 respectively in the US and Canada. Later protection of this very specific use of Atovaquone may have been the result of the Pneumocystis genus being re-classified from protozoa to fungi (yeast). At the time the first KPI family was prepared Pneumocystis infections were thought to be protozoan and the use claim in the first KPI family reflects this by claiming use against protozoal infections. In any case, this patent family appears to protect the monopoly of the Mepron product beyond the expiry of the patents protecting the Atovaquone molecule per se.
Patents in the third KPI family relate to a specific formulation of Atovaquone and would require careful investigation to avoid infringement by generics companies looking to develop Atovaquone before this time. The claims protect microfluidised Atovaquone, Atovaquone of a certain particle size and methods of preparing either or both of these forms. The lipophilic properties of Atovaquone may make it difficult to formulate a suspension product by other methods, but given the specific claims within this patent family there would be opportunity and incentive to develop other alternative suspension formulations, especially since an oral suspension is currently the only dosage form in the US after the tablet form of Mepron was voluntarily withdrawn from the US market by GSK.
Of significant concern to generic competitors wishing to launch a combination of Atovaquone with Proguanil (hence its placement in Category 2 of the KPI), is the fourth KPI patent family, which contains patents protecting the use of a combination of these two active ingredients until expiry in 2013 in most countries.
Innovator companies are becoming much more diligent in protecting their monopoly by life cycle management activities such as filing patent applications protecting other molecular forms, formulations and new uses. In the case of Atovaquone, protection of the combination was a significant step with increasing resistance to monotherapies of other widely used anti-malarial drugs. Such patent-based strategies in the past have proven very successful in extending market share much further than simply relying on the molecule patent.
The Patent Risk Analysis section of the Pipeline Selector reports gives a graphical window into the comprehensive patent filings and provides further insight into the patent landscape around this molecule.
Figure 1: Atovaquone Patent Filing Trends
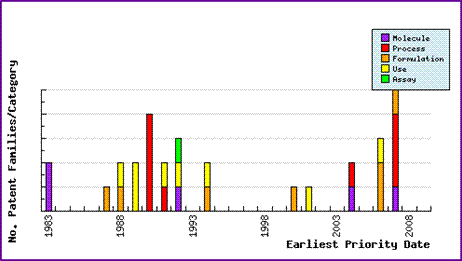
The Patent Filing Trends graph (Figure 1) shows a significant rise in patenting activity just prior to the drugs launch. Most of these can be assumed to be innovator patents, which indicate attempt to manage the life cycle of Atovaquone. The relatively even spread of molecule, process, formulation and use patent applications filed in this period shows that the innovator is attempting to gain the maximum protection across as many facets of the Atovaquone products as possible. Immediately following the drug's launch patent filing remained relatively dormant.
However, a sudden rise in patent filings in recent years suggests that generic companies are developing these products. The major activity has been biased towards process and formulation patents. As indicated above, generic companies may be opting to develop methods that will circumvent the later expiring, more specific innovator patents in these areas.
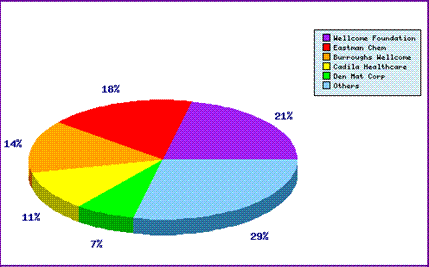
The Top Patent Applicants graph (Figure 2) confirms the patent filing trends by revealing GSK owned companies have filed 35% of the published applications relating to products containing Atovaquone, with the majority of these likely to be those filed prior to launch. Other applicants are responsible for the remaining applications including the majority of those published in recent times. In fact the largest group of applicants consists of a mixture of companies who have filed with only a few patents each.
This might suggest that the life cycle management activities of GSK have not been so vigorous or that the limited market size in western countries for products with these indications has curtailed R&D investment by the innovator. This may present a great opportunity for generic competitors to review the overall situation early and take advantage of possible weak points in the innovator patent portfolio. By monitoring the entire patent landscape surrounding Atovaquone and filing patent applications a company could circumvent the patent applications filed by the innovator and block other generic competitors from doing the same.
Figure 2: Atovaquone Patent Applicants
Comprehensive data for patent families relating to Atovaquone, based on professional patent searching, may be accessed by subscribing to the GenericsWeb Pipeline Developer report, now available in 'Core' and 'Extended' formats. Subscription includes twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector reports are also available in 'Core' and 'Extended' formats for any active ingredient upon request.
For questions and comments about this article please contact the author at s.robinson@genericsweb.com