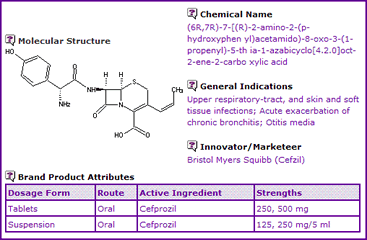
This month's Drug in Focus analyses the patent landscape surrounding Cefprozil, based on information contained in the GenericsWeb Pipeline Selector report, with a view to launching generic equivalents and the difficulties that may be faced in the process. The antibacterial molecule Cefprozil (Cefzil) was launched in the US during December 1991 and indicated for treatment of respiratory tract, skin and soft tissue infections, acute exacerbation of chronic bronchitis and Otitis media. The Cefprozil General Information (Figure 1) indicates the active ingredient is available as tablets or in solution for oral administration.
Figure 1: Cefprozil General Information
The Regulatory Data demonstrates that data exclusivity has expired in all major territories. The market authorisation data shows four companies in Canada and eight companies in the US hold licences for generic versions of this product which follows the order of data exclusivity expiry. Cefprozil has not been authorised in Australia, even for the innovator, which explains why there no generic versions of this product on the market.
Table 1: Cefprozil Data Exclusivity

The Key Patent Indicator (Table 2) for Cefprozil confirms that the patent protection for the Cefprozil molecule has now expired in all major territories. The patent remained in force a year longer in Great Britian compared to the other countries due to an extension. Even with an extension of term the US was the first country to lose protection in 2005, which explains the currently existing generic competitors. The disparity in patent expiry dates is due to the differences in calculation of standard patent terms and/or rules applying to term and allowability of extensions in these countries.
Table 2: Cefprozil Key Patent Indicator
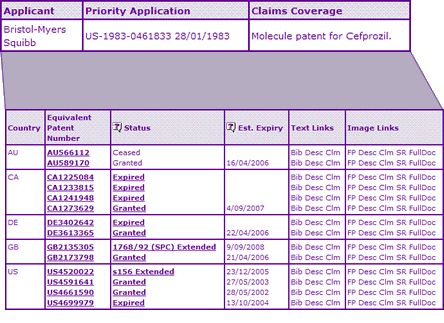
The absence of any litigation implies solid patent protection of the Cefprozil molecule, which is confirmed by the innovator also holding a patent for a crystalline solvate, however there are no other constraining patent families identified by the Key Patent Indicator.
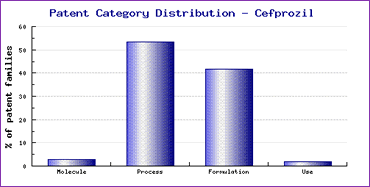
The Patent Risk Analysis section, based on comprehensive patent data (details of which is accessible in the corresponding Pipeline Developer subscription) indicates that patenting activity relating to this drug is heavily focused on methods of API synthesis and formulations, with over 90% of the total patent families falling into on of these two categories (Figure 2). The vast majority of the formulation patents focus on oral administration of the active ingredient. This means a significant amount of attention should be paid to identifying and evaluating the process and formulation patents to ensure that any generic product does not contain an active ingredient manufactured by a protected process or formulation for oral administration at the time of commercialisation.
Figure 2: Cefprozil Patent Category Distribution
Furthermore the patent filing trends for this product show that both process and formulation patents had a resurgence from 2000 to 2003 meaning that if any of these patents are granted they will be in force well beyond the molecule patent expiry. Thus a considerable amount of effort would be required to develop a non-infringing API for launch, in a product, at the end of 2008.
Figure 3: Cefprozil Patent Filing Trends
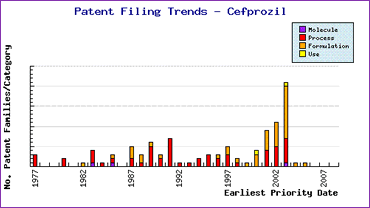
The patent filing trends indicate that patenting in relation to this molecule jumped dramatically several years ago, post launch (generally attributable to generic competition), with an emphasis on formulation patents.
In summary, the market for Cefprozil has been very well protected by the innovators beyond the life of the active ingredient patents. Although later patenting strategies did not include uses of Cefprozil, the large number of patents regarding manufacturing processes may limit the amount of generic competition by eliminating the commercial viability of supply and/or by restricting substitution in respect of certain indications. This is highlighted by the relative lack of interest by generic competitors filing secondary patents. However, the entry of generic competition in most major markets upon molecule patent expiry is inevitable where such competitors have undertaken expert research and evaluation of the patent issues surrounding this product many years ago, and produced a sound intellectual property strategy.
Comprehensive data for patent families relating to Cefprozil, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports, which include twelve monthly updates, to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
For questions and comments about this article please contact the author at s.robinson@genericsweb.com