|
Ke Wang joined GenericsWeb in January 2008 as a patent searcher and analyst. Ke studied biochemistry and biotechnology, and since graduating from university has worked in the biopharmaceutical industry as a therapeutic protein purification technologist. Ke now utilises his knowledge and technical experience in the pharmaceutical industry to provide comprehensive and reliable patent information to GenericsWeb clients. |
 |
Drug In Focus: Venlafaxine
Venlafaxine, or Effexor (ER) is a type of serotonin-norepinephrine reuptake inhibitor and is generally used for the treatment of a range of mental diseases including depression, panic disorder and generalised and social anxiety disorder. It generated revenue of USD 3.79 billion for its innovator, Wyeth, from the US market in 2007. As patent protection for this API has expired or is going to expire later in the year in some core territories, launching the generic version of Effexor will be of great interest of generic pharmaceutical manufacturers worldwide.
Based on information contained in our Pipeline Selector and Developer report for Venlafaxine, this months "Drug In Focus" analyses the patent landscape surrounding the Effexor product with a view to launching generic equivalents.
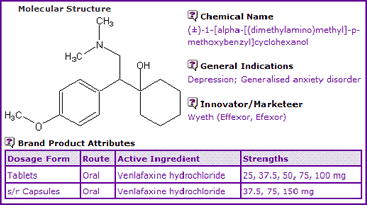
According to Table 1, Venlafaxine is available on the market as oral tablets or sustained release capsules with its hydrochloride salt being the active ingredient.
Table 1: Venlafaxine General Information
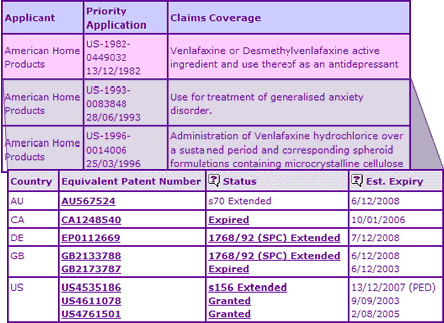
The Key Patent Indicator (Table 2) shows that patent protection in key territories expires in December 2008, having benefited from a full 5-year extension in most countries where such extensions are applicable. It is also worthwhile noting that Wyeth did not designate the UK on their EP patent application, and instead filed separate applications in the UK. Although there could be many reasons for this filing strategy, it does minimise the impact of a possible revocation of a key patent centrally via the opposition procedure at the EPO. Notably, the active ingredient patent has expired in the US and Canada due to differences in patent terms and extension provisions, thus an generic activity in these markets can be used as an indication of potential activity in later expiring EU markets.
Table 2: Venlafaxine Key Patent Indicator
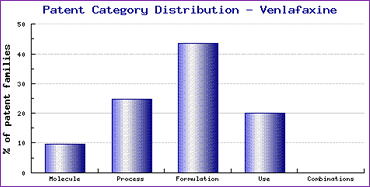
Beside the molecule patent family, two other families have been listed in the Key Patent Indicator, claiming use for the treatment of generalised anxiety disorder and extended release formulation of Venlafaxine respectively, suggesting that Wyeth has made some effort to extend the lifecycle of this product by filing patents to protect the other aspects of the developmental process. The formulation patent listed very specifically claims an extended release formulation (contains defined amount of microcrystalline cellulose) which appears to leave generic competitors possibilities to circumvent by formulating this active ingredient differently. This could be confirmed by the Patent Category Distribution chart (Figure 1) which indicates that patents protecting formulations of Venlafaxine account for more than 40% of all relevant families identified and through exploring the Pipeline Developer report it is noted that most of these formulation patents were filed by generic companies. Nevertheless, since this key formulation patent will be in force until 2017 in major markets it has triggered a number of litigations between Wyeth and generic companies, as discussed later. The use patent family specifically protects only some of the approved indications (generalised anxiety disorder & panic disorder). Having expiry in 2014 in most major markets, this patent will likely affect the indications carried on generic products launched where such patents are in force, said products only carrying the indication for treatment of depression.
Figure 1: Patent Category Distribution
The three patent families listed in the Key Patent Indicator are the only families provided by the FDA Orange Book. Interestingly, a family applied by Wyeth specifically claiming hydrochloride monohydrate salt of Venlafaxine has been found and listed in Pipeline Developer. This family is not included in the Orange Book because applications in all four territories (Australia, Canada, Europe, and the USA) are at their pending stage. However, once the applications are granted by the corresponding patent offices it would become a significant constraint for generic manufacturers because only hydrochloride salt is currently approved by the regulatory bodies worldwide. In addition, a number of patent families specifically claiming various salts and polymorphic forms of Venlafaxine have been identified by the Pipeline Developer. When combined with the charts and graphs, it is clear that caution should be exercised in determining the protection of a marketed product using the Orange Book. Each of the patents listed should be investigated for relevance to a desired generic product and it must be acknowledged that many patents protecting the marketed product held by the innovator and by other generic manufacturers will not be listed. Although GenericsWeb recognises that this may be a good starting point for IP due diligence in relation to a generic development, it should never be the end point.
Table 3: Existing Authorisation Holders

Since the Regulatory Information section of the Selector report shows that data exclusivity has expired in all major territories patent protection appears to be the only restriction that prevents generic versions from launching in the major EU markets. As indicated in the Marketing Authorisation Holders section (Table 3), several generic companies such as Teva, Impax, Sun and Mylan have been granted current marketing approval in the US and/or Canada suggesting that the listed formulation patent is able to be circumvented, at least in these territories. It is important to note that Wyeth has tried to delay those generic challenges by filing lawsuits against generic Venlafaxine manufacturers, mainly for infringement of the key patent protecting the extended release formulation. Interestingly, Wyeth has settled the litigation with Impax (for its filing of an ANDA for generic versions of Effexor including a Paragraph IV certification) by granting a licence that permits Impax to launch Venlafaxine extended release capsule formulation on or after 01/01/2011. Similarly, at least 11 generic companies have marketing approval in the UK and are apparently just waiting for the UK SPC to expire suggesting that the EU markets will go generic in a very similar fashion to the US And Canada.
Figure 2: Venlafaxine Patent Filing Trends
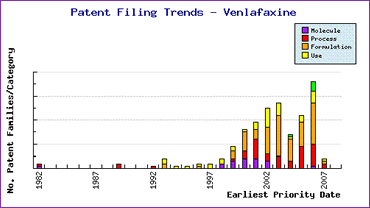
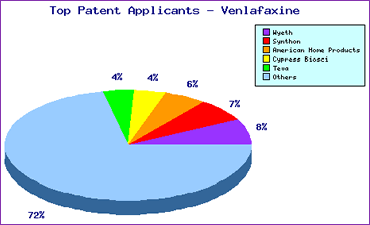
Subsequent to launch, a significant increase in filing activity can be seen (Figure 2), indicating generic interest in the product compounded by the complex patent landscape laid down by the innovator. The Top Patent Applicants analysis (Figure 3) reveals that the innovator has filed only 14% of the published applications indicating that attention has been attracted from generic competitors around the world.
Figure 3: Venlafaxine Top Patent Applicants
In summary, it appears that Wyeth has undertaken a considerable amount of lifecycle management in regards to Venlafaxine and has laid down a number of constraints for generic competitors to overcome should they seek to market a generic product on the day of expiry of the active ingredient patent. However, as generic competitors become more sophisticated at circumventing or challenging patents, such innovator patents become less troublesome and of little use in preventing generic competition. Of course, none of this would be possible without first comprehensively identifying the relevant patents for the development from the 50 million+ published patent documents worldwide, and keeping this information up to date. This is not an area where leading generic companies take chances so they use experts such as GenericsWeb to assist in this area.
Comprehensive data for patent families relating to Venlafaxine, based on professional patent searching, may be accessed by subscribing to the GenericsWeb Pipeline Developer report which include twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
For questions and comments about this article please contact the author at k.wang@genericsweb.com.
|