Leighton Howard is a patent information expert with extensive generics experience. He has worked within the professional patent information industry and major generic pharmaceutical firms and now provides consultancy services to the generics industry and government organisations via his company XIP Pty Ltd. He is also the founder of GenericsWeb and remains instrumental in maintaining the high quality and accuracy of data contained in Pipeline Patent Intelligence. Please email any comments or queries. |
|
Drug In Focus: Rosuvastatin
With underlying sales growth of 59% in 2006 and 33% in 2007, Crestor (Rosuvastatin Calcium) is rapidly climbing the list of high value medicines. Furthermore, a new indication for treatment of Atherosclerosis along with phase III studies underway for new indications relating to end stage renal disease and C-reactive protein (CRP) related disorders, plus a combination product with a fibrate could significantly increase the revenue of US$2.8 billion that AstraZeneca generated in 2007 from the Rosuvastatin molecule. Crestor is therefore attracting the attention of generic manufacturers around the world.
Based on information contained in the GenericsWeb Pipeline Selector report for Rosuvastatin, this months "Drug In Focus" analyses the patent landscape surrounding the Crestor product with a view to launching generic equivalents.
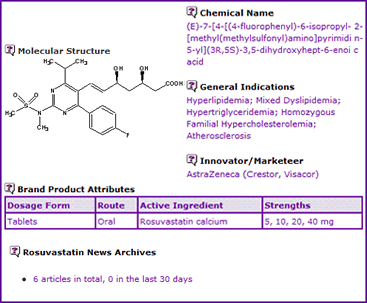
The Rosuvastatin General Information (Table 1) indicates that, at present, only an oral dosage form of Rosuvastatin is available in the form of a calcium salt and current approved indications include the treatment of atherosclerosis, which was approved late 2007 in the US.
Table 1: Rosuvastatin General Information
The Key Patent Indicator (Table 2) for Rosuvastatin confirms that the 5-year extensions on the Rosuvastatin active ingredient patent expire in Germany and the UK in June 2017, whilst the equivalent patent in the US has only been extended until January 2016 (3 ½ years in total). Due to an absence of pharmaceutical patent extension provisions in Canada, the patent protecting the molecule per se will expire there in July 2012, providing a convenient springboard for Canadian manufacturers who ultimately want to enter the US market.
Table 2: Rosuvastatin Key Patent Indicator
It is interesting to note that no equivalent patent protecting the molecule is identified in Australia. However, when considering data exclusivity in major territories, the first marketing authorisation of Rosuvastatin in Australia in May 2006 will preclude the filing for approval of generic equivalents with the TGA until 2011, with likely approval/launch timing of late 2012. Data exclusivity does not extend beyond the life of the patent in other key territories, but this Australian example demonstrates the need to consider all forms of intellectual property protection before devising a development and launch strategy.
Table 3: Rosuvastatin First Marketing Authorisation Dates
In addition to the molecule patent family, three other constraining patent families have been identified by the Key Patent Indicator relating to crystalline forms, formulations and indicated use of Rosuvastatin. This suggests that AstraZeneca have made some attempt to extend the lifecycle of this product by protecting more than one element of the marketed product, a suggestion that is reinforced by the patent filing trends graph (Figure 1). This graph clearly shows a significant amount of patent filing activity prior to the first major launch of the product in the Netherlands in 2002, most of which was undertaken by the innovator.
Figure 1: Rosuvastatin Patent Filing Trends
Subsequent to launch, a significant increase in filing activity can be seen, indicating generic interest in the product in combination with a complex patent landscape laid down by the innovator. The Top Patent Applicants analysis (Figure 2) reveals that generics giant Teva has filed almost as many published applications as AstraZeneca, with two more major generics firms hot on their heels.
Figure 2: Rosuvastatin Top Patent Applicants
Referring back to the Key Patent Indicator, one of the patent families listed, relating to a crystalline hydrate of Rosuvastatin calcium, was delisted from the US FDA Orange Book, possibly because the FDA approved label for the Crestor product clearly states that the active ingredient is a white amorphous powder and so does not actually protect the marketed product. When combined with the above graphs, it is clear that caution should be exercised in determining the protection of a marketed product using the Orange Book. Each of the patents listed should be investigated for relevance to a desired generic product and it must be acknowledged that many patents protecting the marketed product held by the innovator and by other generic manufacturers will not be listed. Although GenericsWeb recognises that this may be a good starting point for IP due diligence in relation to a generic development, it should never be the end point.
Although the formulation patent listed in the Key Patent Indicator appears to be very broad in scope (Rosuvastatin in combination with an inorganic salt in which the cation is multivalent), AstraZeneca have used a duplicate filing strategy in most European countries to increase their chances of obtaining protection. Duplicate filing involves filing a European patent designating a country and filing an equivalent national patent in that country at the same time. This has resulted in the grant of some national patents in major European territories and the US, which contain claims that have been blocked by the European Patent Office, thus highlighting the disparity of patent examination quality around the world. Nevertheless, a significant amount of formulation development has been undertaken by generic manufacturers as highlighted in the Patent Filing Trends (figure 1) thus it is likely that this patent family, even if granted with broad claims, will not constrain generic competition completely.
Finally, it is worth mentioning that several Legal Battles are underway in the US as a result of Paragraph IV filings by a number of generic competitors including Apotex, Aurobindo, Cobalt, Mylan, Par, Sandoz and Sun, as shown in the corresponding PIV Filings and Litigation Alerts section of the Pipeline Selector report.
In summary, it appears that AstraZeneca has done a considerable amount of lifecycle management in regards to Rosuvastatin and has laid down a number of constraints for generic competitors to overcome should they seek to market a generic product on the day of expiry of the active ingredient patent. However, as generic competitors identify potential candidates for development earlier and become more sophisticated at circumventing or challenging patents, such innovator patents become less troublesome and of little use in preventing generic competition. Of course none of this would be possible without first comprehensively identifying, from the 50 million+ published patent documents worldwide, the relevant patents required for the generic development and keeping this information up to date. This is not an area where leading generic companies take chances so they use experts such as GenericsWeb to assist in this area.
Comprehensive data for patent families relating to Rosuvastatin, based on professional patent searching, may be accessed by subscribing to the GenericsWeb Pipeline Developer report which includes twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
|