Leighton Howard is a patent information expert with extensive generics experience. He has worked within the professional patent information industry and major generic pharmaceutical firms and now provides consultancy services to the generics industry and government organisations via his company XIP Pty Ltd. He is also the founder of GenericsWeb and remains instrumental in maintaining the high quality and accuracy of data contained in Pipeline Patent Intelligence. Please email any comments or queries. |
|
Drug In Focus: Famciclovir
Expiry of patents covering the antiviral active ingredient Famciclovir (Famvir) in most major European markets at the end of 2008 will likely result in an increase of activity from generic manufacturers seeking to steal a share of the significant revenues for this product. Based on information contained in the GenericsWeb Pipeline Selector report for Famciclovir, this months Drug In Focus analyses the patent landscape surrounding this product with a view to launching generic equivalents, and the difficulties faced in doing so.
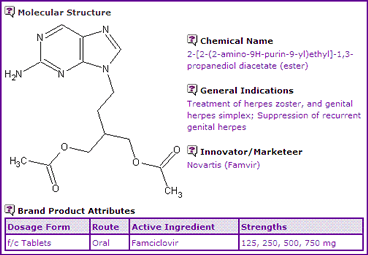
The Famciclovir General Information (Table 1) indicates that the active ingredient is made available as the basic compound in only oral tablet form. It is worthwhile noting that the strengths of the active ingredient are relatively high, and therefore the cost of the active ingredient manufacture may play a more significant part in the commercial viability of development of generic versions of this product. Most significantly, this product carries three indications, the latter approved being Suppression of recurrent genital herpes, suggesting that patent protection may be possible in respect of at least this indication.
Table 1: Famciclovir General Information
The Regulatory Data demonstrates that data exclusivity has expired in all major territories. The market authorisation data for Canada shows a number of companies holding licences for generic versions of this product. Interestingly, it can also be seen from the data that Teva submitted a paragraph IV certification and have since launched a generic version of the Famvir product. It is also clear from the DMF filings that at least three other generic API manufacturers are interested in supplying API for generic products to be launched in the US market.
Table 2: Famciclovir Data Exclusivity
Analysis of the Key Patent Indicator (Table 2) for Famciclovir confirms that the patent protection on the Famciclovir active ingredient doesnt expire in the US, and Australia until 2010, much later than the corresponding protection in Europe. It can further be seen that the basic patent expired in 2006 in Canada, explaining the existence of generic competitors currently (the later-expiring Canadian patent in this family does not appear to protect Famciclovir per se but is nevertheless important to identify and assess). The disparity in patent expiry dates is due to the differences in calculation of standard patent terms and/or rules applying to term and allowability of extensions in these countries. However, based on this information, it appears that Teva have launched a generic product whilst the innovator product is still under patent protection.
Table 3: Famciclovir Key Patent Indicator
The Litigation Alert in respect of the molecule patent in the US confirms that Teva has launched at risk in the US pending the results of its paragraph IV litigation and is currently enjoying 180 days exclusivity due to being the first to file a Paragraph IV certification. The benefit of this exclusivity is seemingly quite low if no other manufacturers have filed Paragraph IV certifications and are awaiting the 2010 molecule expiry date.
In addition to the molecule patent family, two other constraining patent families have been identified by the Key Patent Indicator. Both of these relate to uses of Famciclovir which appear to encompass the current indications and specifically the later approved indication Suppression of recurrent genital herpes. These patent families each comprise granted patents in major territories which expire in 2014 and 2015 respectively, thus any company intending to launch a generic version of Famciclovir should seek legal advice to ascertain the risk of being found to infringe these patents upon doing so, and to investigate any options for launching a product with partial indications.
Study of the Patent Risk Analysis section, based on comprehensive patent data (details of which is accessible in the corresponding Pipeline Developer subscription) indicates that patenting activity relating to this drug is heavily focused on methods of API synthesis, with nearly half of the total patent families falling into this category (Figure 1). This indicates that a significant amount of attention should be paid to identifying and evaluating such process patents to ensure that any generic product does not contain an active ingredient manufactured by a protected process at the time of commercialisation.
Figure 1: Famciclovir Patent Category Distribution
Furthermore, analysis of patent filing trends for this product shows that most of the process patents were applied for in the five years prior to first EU approval of the Famvir product in 1993, meaning that any process patents granted are likely to be in force beyond the expiry of the Supplementary Protection Certificate on the molecule patent. Thus a considerable amount of effort would be required to develop a non-infringing API for launch in a product at the end of 2008.
Figure 2: Famciclovir Filing Trends
A further insight that may be drawn from the patent filing trends is that patenting in relation to this molecule post launch (generally attributable to generic competition) seems to be lower than pre-launch patenting activity (generally innovator activity). This is a rare case in todays generic market and is likely to be the result of generic companies deciding not to develop this product on the basis that any non-infringing API would be too difficult or too expensive to produce commercially, which in turn is a result of the myriad of patents laid down by the innovator. It is also noteworthy that a number of molecular form patents have been applied for more recently, and may provide some uncertainty with regard to the protection of certain forms of the active ingredient in generic products, or the impurities they may contain.
In summary, the market for Famciclovir has been very well protected by the innovators beyond the life of the active ingredient patents. Later patenting of uses of Famciclovir and processes for manufacture thereof, compounded with proactive lifecycle management to introduce patented indications into the portfolio of protection of the innovator product may limit the amount of generic competition by eliminating the commercial viability of supply and/or by restricting substitution in respect of certain indications. This is highlighted by the relative lack of interest by generic competitors filing secondary patents. However, the entry of generic competition in most major markets upon molecule patent expiry is inevitable where such competitors have undertaken expert research and evaluation of the patent issues surrounding this product many years ago, and produced a sound intellectual property strategy.
Comprehensive data for patent families relating to Famciclovir, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
|