Leighton Howard is a patent information expert with extensive generics experience. He has worked within the professional patent information industry and major generic pharmaceutical firms and now provides consultancy services to the generics industry and government organisations via his company XIP Pty Ltd. He is also the founder of GenericsWeb and remains instrumental in maintaining the high quality and accuracy of data contained in Pipeline Patent Intelligence. Please email any comments or queries. |
|
Drug In Focus: Ropinirole
Patent strategies used by innovators and generic companies alike are becoming increasingly sophisticated as companies are recognising the tremendous value in devising long-term patent strategies to maximise the lifetime of a drug. Whilst the legality of such strategies in Europe is currently under question (a sector inquiry was recently launched into the pharmaceutical industry), it is no surprise that patent information has become a key component in the development of a generic. Comprehensive patent information ensures that your generic avoids infringing not only innovator patents, but also patents that have been applied for by generic developers in a bid to develop their own versions of the drug for launch at the earliest possible opportunity. Patent information can also provide valuable insight into the activities of competitors or even potential partners for your drug development.
This months Drug In Focus examines the GlaxoSmithKline dopamine agonist, Ropinirole (Requip, Repreve, Ardetel, Vunexin), using information found in the GenericsWeb Pipeline Selector report.
Table 1: Ropinirole General Information
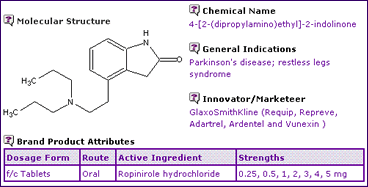
The General Information section (Table 1) reveals that products containing Ropinirole are available only as an oral tablet formulation in a variety of strengths for treatment of two indications: Parkinsons disease and Restless Legs Syndrome. Table 2: Ropinirole Drug Master Files (DMFs) 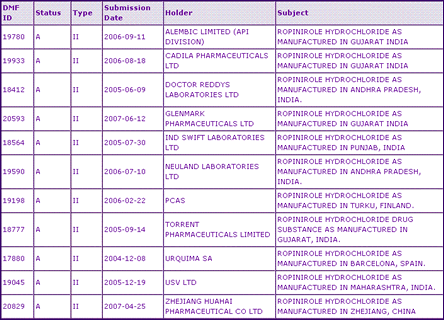
Upon inspection of the Ropinirole regulatory data, we can see from the first marketing authorisation dates that data exclusivity has expired in all major territories, with the exception of US data exclusivity for the Restless Legs Syndrome indication which expires in May 2008. In Europe, the 1996 first marketing date suggests that applications for approval of generic Ropinirole products may be filed from January 2006. However, analysis of GB marketing authorisation holders reveals that only GlaxoSmithKline holds current authorisations, which include 2006/7 approvals for products carrying the secondary indication for Restless Legs Syndrome. If applied for at the earliest point in time, the first generic authorisations are likely to be seen in the near future, allowing for a period of around 18 months from filing. The fact that DMFs have been filed in the US by eleven different international companies suggests that many generic competitors are working on this product and therefore such approvals are highly likely.
Figure 1: Ropinirole Key Patent Indicator
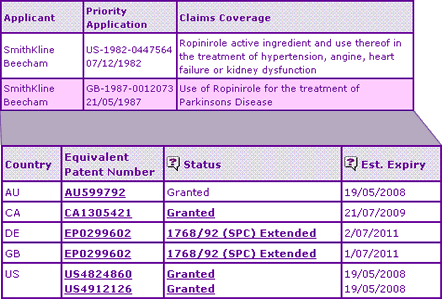
The molecule patent has been extended to expire in November 2008 in the UK, Germany and Australia, although the equivalent US and Canadian patents have already expired. However, we can see from the Key Patent Indicator (Figure 1) that a second patent family is highlighted which protects the use of Ropinirole for the treatment of Parkinsons Disease. This offers protection in the US until May 2008 (15 days after expiry of the Restless Legs Data Exclusivity) but, in a very unusual case, a secondary SPC has been granted for the same molecule on this later patent family in both Germany and the UK, which expires in 2011. It is thought that this was only possible because the application date for both SPCs is identical, as EC regulation 1768/92 states that:
A certificate shall be granted if, in the Member State in which the application referred to in
Article 7 is submitted and at the date of that application:
(c) the product has not already been the subject of a certificate;
Similar extensions were not applied for in Australia or the US, suggesting that the product will experience generic competition much earlier in these territories than in Europe.
It is noteworthy that no additional patent protection is shown in the Key Patent Indicator in respect of the Restless Legs Syndrome secondary use, although analysis of the corresponding Pipeline Developer report, based on comprehensive patent searching, may reveal that attempts have been made. The net result of this situation is that generic Ropinirole products indicated for Restless Legs Syndrome may become available in Europe before their corresponding products carrying the Parkinsons Disease indication.
Figure 2: Ropinirole Top Patent Applicants
The Top Patent Applicants chart (Figure 2) shows that almost a quarter of the patents for this API are held by the innovator. Considering the number of patents involved, this high percentage suggests that the innovator has implemented a long-term strategy to manage the lifecycle of this product. Also worth noting is that the top 5 applicants by volume together comprise only 40% of the applications, indicating that a large number of other companies have invested their research capabilities into the development of Ropinirole. This distribution suggests the likelihood of strong competition once the main patents expire for Ropinirole.
Figure 2: Ropinirole Filing Trends
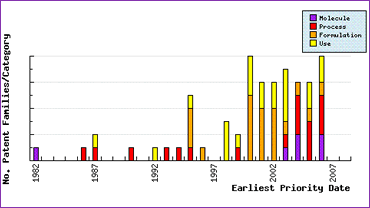
Patent Filing Trends (Figure 3) for this API show that in the 12 years following the filing of the 1982 molecule patent only process and use patents were applied for, most likely filed by the innovator. However, in 1995, the year before launch, we can see a burst of patent activity in the formulation category, as well as applications in the process and use categories. The subsequent response in patent filings post-launch suggests that many generic competitors have developed new formulations and routes of synthesis, and that caution should be exercised in avoiding the scope of these patents when developing a generic product.
In summary, we can conclude that GlaxoSmithKline have done well to extend the monopoly life of their Requip product carrying the primary indication of Parkinsons Disease by way of filing multiple SPCs on the active ingredient in a manner that seemingly falls within the scope of the legislation (although perhaps not its intent). On the other hand, they seem to have missed a similar opportunity in Australia where multiple extensions are more accepted. Furthermore, beyond the expiry of the basic patents there appear to be very few formulation or molecular form patents in the way of generic competitors, and GlaxoSmithKlines failure to obtain patent protection for the Restless Legs Syndrome indication could see their Anti-Parkinsonian market share eroded in Europe by off-label use of equivalent generic products before the SPC expires on the Parkinsons Disease use patent.
Comprehensive data for patent families relating to Ropinirole, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
|