Leighton Howard is a patent information expert with extensive generics experience. He has worked within the professional patent information industry and major generic pharmaceutical firms and now provides consultancy services to the generics industry and government organisations via his company XIP Pty Ltd. He is also the founder of GenericsWeb and remains instrumental in maintaining the high quality and accuracy of data contained in Pipeline Patent Intelligence. Please email any comments or queries. |
|
Drug In Focus: Bicalutamide
Expiry of patents covering the Bicalutamide active ingredient in some major markets in the near future are likely to result in interest from generic competitors wishing to launch equivalent products. Based on information contained in the GenericsWeb Pipeline Selector report for Bicalutamide, this months Drug In Focus analyses the patent landscape surrounding this product with a view to the ease or difficulty with which such generics competitors may have developed generic equivalents.
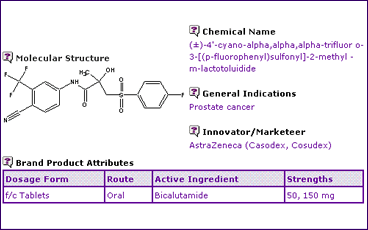
Bicalutamide is the active ingredient in AstraZenecas Casodex(R), indicated for the treatment of prostate cancer. The treatment is a non-steroidal, anti-androgenic product orally administered as a 50 mg or 150mg tablet, as can be seen from the Bicalutamide General Information (Table 1).
Table 1: Bicalutamide General Information
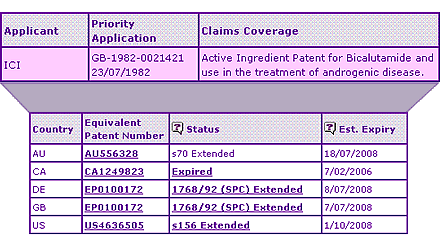
The Key Patent Indicator (Table 2) for Bicalutamide confirms that the extensions on the active ingredient patent expire in Australia, Germany and the UK in July 2008. In the USA, the patent term has been extended to October 2008 and no paediatric exclusivity has been applied.
In Canada, the molecule expired in 2006. This was earlier than in other key markets due to the lack of provisions for extension of term for pharmaceutical patents which exist elsewhere. The listing of NOCs in Canada tells a story of a product that lost significant market share to generic competitors, with six equivalent products being authorised.
Table 2: Biculatamide Key Patent Indicator
Will the same happen in other markets? The absence of any other patent families identified by the Key Patent Indicator that protect the marketed Casodex product, and which may have acted as a hurdle for generic competitors, suggests that this story will be repeated in all countries immediately after molecule patent expiry.
Indeed, the last of the data exclusivity protection in major markets expired in Europe in February 2005, allowed generic competitors ample time to push marketing authorisations through the regulatory authorities before molecule patent expiry. Analysis of the UK Marketing Authorisations shows that six generic versions of Bicalutamide, most in both strengths, have already been approved more than 8 months before the earliest launch date.
Similarly, the filing of 18 US DMFs suggest that many generic players are planning to battle it out for market share of one the few products nowadays that has not been subject to a pargraph IV filing. The lack of 180 days exclusivity as a reward for filing a paragraph IV certification means that the significant first mover advantage in obtaining the lions share of the market is not automatically attributed to one generic company (or two if you count authorised generics), but will probably be divided more evenly between all competitors. This ultimately results in a price war, of which only the health funds are the victors.
So one may ask the question why will this product meet so much generic competition, when the marketers of some other products can stave of generic competition for many years? The answer ultimately comes down to lifecycle management, and those intellectual property-related activities that (legitimately or not) have been undertaken much earlier in the life of other products, for which no evidence is found for Casodex.
Study of the Patent Risk Analysis section, based on comprehensive patent data (details of which are accessible in the corresponding Pipeline Developer subscription) shows a surprising statistic that a generic manufacturer has filed more patent applications in respect of this molecule than the innovator. This is the first indication that the innovator has not taken the maximum opportunity to protect more specific elements of the product, such as crystalline forms, efficient processes for manufacture and more stable formulations. This created an opportunity for the first generic firms to analyse the product in more detail and protect some variations on the current marketed product..
Figure 1: Bicalutamide Top Patent Applicants
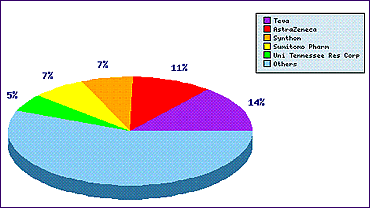
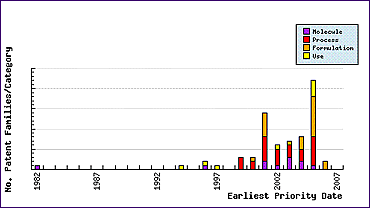
The Patent Filing Trends graph for this molecule (Figure 2) confirms that patent filings prior to the 1995 launch of the product, usually filed by the innovator, were very low in number. In fact this rare case shows that patenting specifically in relation to this product ceased after molecular discovery with the exception of one secondary use application. However, since the launch of the product, several generic companies have filed applications on molecular forms, processes and formulations (the Top Patent Applicants graph shows that there are at least 11 patentees in addition to the top five).
Of course, with generics companies now vigorously defending their patent portfolios, caution is necessary with regard to these later filed patents to ensure that the claims of all relevant patents and applications have been considered from an infringement perspective. One of the biggest problems in determining the risks when launching a generic product protected by so many generic patent filings is that they have a high level of pendency at the date of molecule patent expiry, due to their filing dates being late in the lifecycle of the product. This means that broad claims found in patent applications need thorough evaluation to establish the likelihood of grant and, if granted, the anticipated scope and validity of the final claims. Fortunately, many of these patent applications are not awarded with a significant level of protection. However, even the most obvious of inventions can be granted protection by patent offices creating enormous problems for some generic competitors, and creating huge opportunities for others. Monitoring of patent publications and statuses throughout a generic development and post-launch is therefore essential. Figure 2: Bicalutamide Filing Trends
Now, consider if these patents had instead been filed by the innovator earlier in the lifecycle of the product, preferably before launch. They would stand a much higher chance of being granted with broader claims based on the lower level of prior art in the public domain at the time. This would significantly enhance protection of the product from generic competitors and at the very least result in a diminished number of generic competitors at molecule patent expiry and at best prevent generic competition for an additional number of years.
On the other hand, the fact that the innovator has not undertaken such lifecycle management activities means that:
a) The innovator has focused its valuable R&D resources in discovering truly innovative products instead of defending old ones;
b) Any generic product will be an exact equivalent of the innovator product and not a slightly different crystalline form, salt or formulation thus enhancing safety; and
c) The value in the generic product will be destroyed by the sheer number of generic competitors due to the relative ease of development.
In summary, the market for Bicalutamide has not been well protected by the innovator beyond the life of the active ingredient patents. Later patenting by generic competitors will create some risk and uncertainty for fellow generic competitors but if indicators from off-patent markets are true the later-expiring markets will be flooded with generic versions of Casodex after molecule patent expiry in 2008, resulting in limited returns on the costs of development. Although lifecycle management by the innovator may have bought an extra few years monopoly life, this has been foregone (whether intentionally or unintentionally) resulting in their ability to concentrate on what they do best innovation, rather than fighting generic companies in court.
However, generics companies take note: There are unlikely to be many more molecules left with such a simple patent landscape. Many innovators share the view that lifecycle management activities are truly value adding, thus most future generic developments will be much more complex than Bicalutamide from a patent perspective. This means there will be some big opportunities, accompanied by some big inherent risks.
Comprehensive data for patent families relating to Bicalutamide, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications.
|