This month's 'Drug In Focus' is written by Trevor Giacomelli, a patent information expert who currently maintains the high quality and accuracy of data contained in GenericsWeb's Pipeline Patent Intelligence. He has extensive chemistry knowledge through his studies under scholarship at the University of Sydney. He is currently studying a Masters of Legal Studies at the University of Technology Sydney. Please email any comments or queries. |
|
Drug In Focus: Dasatinib
Dasatinib (Sprycel) was recently launched by Bristol-Myers Squibb as an oral anti-cancer agent that is used to treat chronic myeloid leukemia and certain types of acute lymphoblastic leukemia (Figure 1). Dasatinib targets proteins in specific cancer cells and stops those cells from growing. This months Drug in Focus will analyse patent protection surrounding Dasatinib by utilising GenericsWebs Pipeline Patent Intelligence with a view to launching a non-infringing generic.
Figure 1: Dasatinib General Information
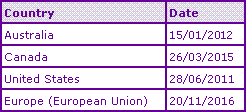
The first hurdle that will be encountered in the development process is that of data exclusivity provisions existing across the globe (Table 1). With the launch being so recent, there are varying dates in which regulatory bodies may begin accepting applications for approval of generics that rely on the data contained in relation to the new chemical entity marketing authorisation. It can be found that the following exclusivity dates apply in the four territories listed: Australia 15/01/2012; Canada 26/03/2013, which would be the earliest date for filing a generic application and 26/03/2015 being the earliest possible authorisation date of a generic; United States 28/06/2011 (or 28/06/2010 if the application contains a certification of patent invalidity or non-infringement, i.e. a Paragraph IV filing); and European Union 20/11/2016. The range of dates here differs significantly and so careful planning must be undertaken to ensure that data exclusivity provisions do not interfere with launch strategies, and that opportunities for earliest filing/approval are not missed.
Table 1: Dasatinib Data Exclusivity
At this early stage only the molecule patent has been identified in the Key Patent Indicator (KPI) (Table 2). Currently, the three patents in this family which have been granted in the U.S. are due to expire in 2020, whilst it appears that an extension under USC s156 could be granted taking the expiry date to the June 28th 2020, plus any paediatric extension that may apply. The Australian granted patent is due to expire in early 2022 due to a granted extension provision based on a later marketing authorisation date. At this stage, no other patents in major territories have been granted, but their acceptance would be expected once they have been examined by the various patent offices. Knowing that there exists an extension in Australia, one could assume that similar extensions will appear in other regions allowing this special term based on similar provisions.
Table 2: Dasatinib Key Patent Indicator
The reason only one patent family has been recognised in the KPI could be due to a number of reasons. Firstly, there may be key patents that have been filed and are still awaiting grant and are therefore yet to appear in databases such as the Orange Book. Secondly, patents may not have even been published yet, due to the patent black hole, or more specifically, the normal lag of about 18 months from the date that provisional specification is lodged until it is gazetted.
There is a very strong possibility that more patents will be added to the KPI in the future. Innovator companies are becoming much more diligent in protecting their monopoly by life cycle management activities. Such patent-based strategies are very successful in extending market share much further than simply expiry of the molecule patent, unless generic competitors review the situation early and block ways of circumventing these key patents to secure their own place in the market by themselves patenting. The key point is having comprehensive patent data that is updated regularly so that you are well informed to make confident business decisions.
The full Pipeline Developer comprehensive patent search report does indeed indicate some life cycle management being performed by Bristol-Myers Squibb. There are several patent families filed by the innovator in a range of categories which could pose potential problems for companies relying on basic patent information. Analysis of the Patent Risk Analysis section of the Pipeline Selector report provides further insight into the likely obstacles that will be encountered.
The Top Patent Applicants graph (Figure 2) shows that Bristol-Myers Squibb owns a high proportion of the patents on this API. Other top applicants shown in the figure are likely due to cross-patenting; for example Novartis presence is possibly owing to their patenting activity on their Imatinib (Gleevec) product, in a way that may give their own generic divisions access to their competitors markets. As proactive generics companies assess the patent landscape around this time, and patent viable ways of circumventing the strong innovator patents that already exist, the proportion of the others section will increase dramatically.
Figure 2: Dasatinib Top Patent Applicants
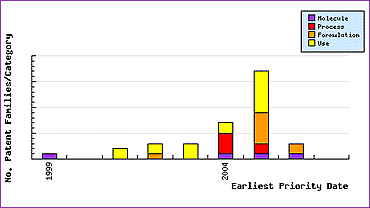
The Patent Filing Trends graph (Figure 3) shows a noticeable rise in patenting activity just prior to the drugs launch. Most of these can be assumed to be innovator patents, which would again indicate life cycle management. The relatively even spread of molecule, process, formulation and use patents filed in this period shows that the innovator is attempting to gain the maximum protection across as many facets of the marketed product as possible. These patents are likely to contain broad claims due to the lack of prior art, so may provide strong protection and, if granted, will expire long after the patent on the active ingredient thus possibly extending the monopoly on this molecule.
Figure 3: Dasatinib Patent Filing Trends
This brings us to the question: why do I need this information now if the expiry of the molecule is not for another 13 years or more? Selecting a drug that fits in with your pipeline should be done in a manner that takes into consideration all of the potential risks. To make the most of research and development activities, a generics company should have their product on the market first, preferably with as few competitors as possible following. Assessing the patent landscape immediately after launch provides a proactive generics company with the best opportunity to begin their R & D, and start to file their own patents to fill in the holes left by the innovator. Patent strategies are more important than ever in todays generics market, so developers are well advised to get reliable, insightful patent information, and to get it early.
Comprehensive data for patent families relating Dasatinib, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector & Developer reports are available for any active ingredient upon request.
|