Leighton Howard is a patent information expert with extensive generics experience. He has worked within the professional patent information industry and major generic pharmaceutical firms and now provides consultancy services to the generics industry and government organisations via his company XIP Pty Ltd. He is also the founder of GenericsWeb and remains instrumental in maintaining the high quality and accuracy of data contained in Pipeline Patent Intelligence. Please email any comments or queries. |
|
Drug In Focus: Temozolomide
Expiry of patents covering the anti-cancer active ingredient Temozolomide (Temodar, Temodal) in some major markets last month did not result in the usual entry of generic competitors. Based on information contained in the GenericsWeb Pipeline Selector report for Temozolomide, this months Drug in Focus analyses the patent landscape surrounding this product with a view to launching generic equivalents to determine the likely constraints on generic competitors wanting to launch an equivalent of this costly product.
The Temozolomide General Information (Table 1) indicates that the product is available only as oral capsules, in six strengths, containing the Temozolomide in base form.
Table 1: Temozolomide General Information
An initial glance at the marketing authorisations for Temozolomide in the major territories (Table 2a) indicates at least one constraint to generic competitors entering the market in Europe, with applications for authorisation of generics not being allowed prior to 26/01/2009. Data exclusivity in Canada and Australia appears to have lapsed, whilst the US innovator product will benefit from Orphan Drug Exclusivity until 2012 (Table 2b), at least for the Glioblastoma Multiforme (GBM) indication.
Table 2a: Temozolomide First Marketing Authorisation Dates

Table 2b: Temozolomide US Data Exclusivity
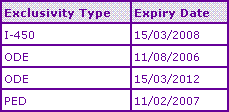
The Key Patent Indicator (Table 3) confirms that extensions on the Temozolomide active ingredient patent expired in Australia, Germany and the UK last month leaving the molecule and any routes of synthesis outlined in the molecule patent in the public domain. This also enables commercialisation of any generic products to potential customers e.g. to lock in supply deals, subject to receiving marketing authorisation for the product. However, in contrast, the US and Canadian patent protection extends out to 2012 and 2014 respectively due to variation in patent filing systems and legislation around the world.
Table 3: Temozolomide Key Patent Indicator
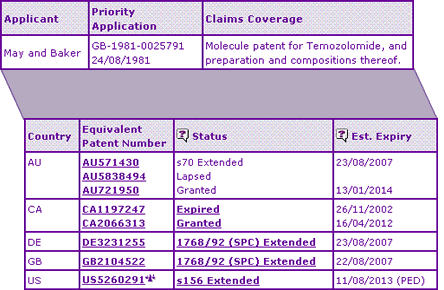
Given the existence of only innovator products listed in the Australian Register of Therapeutic Goods (ARTG), a question that might be raised is Why have no generic competitors entered the market in Australia now that there is no patent or data exclusivity protection? (the later Australian patent expiring in 2014 related to concomitant therapy with an ATPase inhibitor, so is not considered to be constraining). The reasons for this must lie in either the commercial aspects of developing a generic product, or perhaps the existence of later patents that protect other aspects of the innovator product beyond the chemical compound per se, such as process or formulation patents.
Given the grant of Orphan drug status to the two main indications in the US (suggesting a patient size of below 0.1% of the population), the market size in Australia could be considered to be too small to warrant the substantial costs of development, bioequivalence studies and application. However, the situation for European and US markets is of course different, in that one development could be used to generate revenues from a larger population, enabling a viable investment. The commercial constraints are therefore unlikely to prevent generic competition completely, but a significant advantage will be gained by those generic competitors who can enter or secure a market first (a US paragraph IV filing by Barr is already listed), and an even larger financial return will result if the number of generic entrants can be limited to just a few, maintaining relatively high prices.
Therefore the question of other patent constraints becomes relevant, to see how difficult it is likely to be for generic companies to navigate the patent landscape, who these competitors may be and whether they might exclude other generic competitors with their own patent protection.
Aside from the molecule patent family, no other constraining patent families have been identified by the Key Patent Indicator, suggesting that there should be no significant patent obstacles. Hence the next stage is to more comprehensively analyse the patent filings for this product.
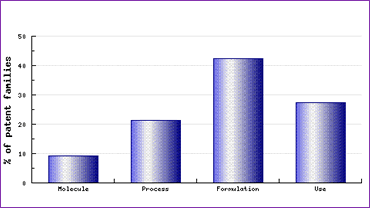
Study of the Patent Risk Analysis section, based on comprehensive patent data , indicates that filing activity relating to this drug is spread across molecule, process, formulation and use patents, with the emphasis of filings being in the formulations section (Figure 1). This is quite a regular distribution for any molecule and therefore caution should be exercised in regards to each aspect of a generic development.
Figure 1: Temozolomide Patent Category Distribution
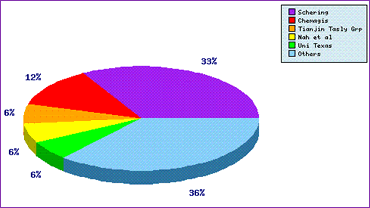
The Top Patent Applicants analysis (Figure 2) suggests that at least one generic competitor is developing a generic product (this being Chemagis, now part of the Perrigo group), and also identifies a number of filings by the innovator, Schering. Patents filed by these applicants relate to many different aspects of a generic product, and may cause problems to lesser-informed generic developers if granted in relevant territories, thus a thorough analysis with reference to the proposed generic product is required prior to committing to such a costly development. A further insight from this graph is that the top five patent applicants represent 64% of the total filings, suggesting that this molecule is either not one that many generic competitors are interested in developing (for one of many reasons) or that the patent situation is simple enough so that any significant R&D effort to circumvent patents by generic developers is unnecessary. The reasons lie in the comprehensive patent detail found in the Pipeline Developer report.
Figure 2: Temozolomide Top Patent Applicants
Based on this analysis and interpretation of the GenericsWeb Pipeline Selector report it can be clearly established that the launch of generic equivalents of Temozolomide is currently constrained by data exclusivity restrictions alone in European markets, a combination of data exclusivity and patents in the USA and Canada, and by commercial constraints in Australia. It is possible that the commercial constraints for smaller markets will be overcome when developing a product for multiple markets whilst the Paragraph IV filing in the US shows a clear intent to launch there on the day of patent expiry. However, the winner of the race in Europe is an uncertainty that will ultimately be decided by the best regulatory submission filed on 26th January 2009 and the number of competitors who finish the race will be decided by the confidence that potential entrants have in their patent information and their regulatory expertise.
Comprehensive data for patent families relating to Temozolomide, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector & Developer reports are available for any active ingredient upon request.
|