Leighton Howard is a patent information expert with extensive generics experience. He has worked within the professional patent information industry and major generic pharmaceutical firms and now provides consultancy services to the generics industry and government organisations via his company XIP Pty Ltd. He is also the founder of GenericsWeb and remains instrumental in maintaining the high quality and accuracy of data contained in Pipeline Patent Intelligence. Please email any comments or queries. |
|
Drug In Focus: Atorvastatin
Lipitor is a brand that every generics company is aware of, being a global blockbuster having sales of 13 billion US Dollars in 2006. However, the various players in the industry have mixed views as to whether or not Atorvastatin is a molecule they want to add to their generics pipeline. Some argue that it is too difficult to develop or will be too heavily contested upon API patent expiry, whilst some companies are investing heavily in research & development and/or litigation. Others simply cannot decide whether they want this product or not.
The GenericsWeb Pipeline Selector was developed in response to customer demands for a cost-effective, single source of patent, regulatory and other data to assist the reader in making the decision of whether to add a particular generic product to their generic pipeline. This article, similar to all other INNsight Drug in Focus articles, is based solely on interpretation of data provided in the corresponding GenericsWeb Pipeline Selector report and demonstrates, with particular reference to Atorvastatin, how a decision about a very complex generic product can be vastly simplified and expedited for the cost of AUD500/USD420/EUR310/GBP210.
An initial review of the Atorvastatin general information shows that the active ingredient is an enantiomer and contains a combination of functional groups that may cyclise to form a lactone impurity unless stabilised. It further shows that the product is available as an oral instant-release dosage form in four strengths, as well as a combination product with Amlodipine (Caduet), both products containing Atorvastatin in the crystalline form of the calcium salt trihydrate, designated form I.
Table 1: Atorvastatin General Information
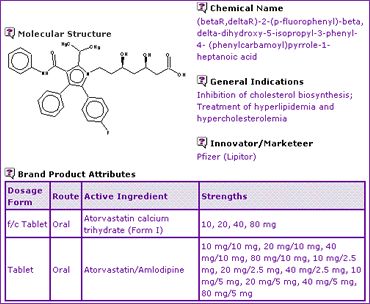
This information is not only useful for understanding what form the generic product must take, but is also very useful as a reference when reviewing patent protection in relation to the product[1].
Moving on to regulatory data, we can see that NCE data exclusivity for Atorvastatin in major territories has expired, particularly in the markets with the longest period of 10 years, but that data protection is still in force in the US in respect of certain indications. The take-home message here is that patents are likely to be Lipitors only form of protection from generic competition and that regulatory filing is no longer prevented in these markets.
Further regulatory data shows US DMF filings, which can be useful both as an indicator of which API suppliers actually have product available and also of whether others have prepared alternative salts or polymorphic forms to the form used in the innovator product. This information is relevant regardless of the intended generic market. In this case it is clear to see that some companies have developed amorphous Atorvastatin as well as different salts and polymorphic forms, indicating some kind of patent-related problem but, more importantly, also indicating that developing such an API is a viable alternative.
Table 2: Atorvastatin DMF filings
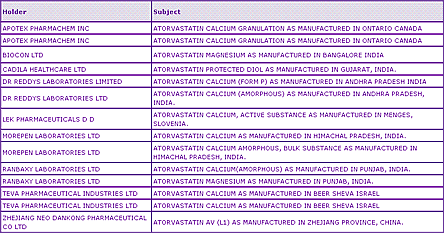
For those focused on the US market, the presence of Paragraph IV filings is an important indicator of the opportunities that are available for this product, given the significant effect of the resulting 180 day exclusivity on market share post-expiry.
The marketing authorisation data identified in key countries show that no generics have been approved as yet. Such information can not only be used to monitor such generic approvals but can also be used to track new product line extensions that have been launched by the innovator.
The Key Patent Indicator (Table 3) for Atorvastatin confirms that patent families have been filed that protect both the racemic mixture of Atorvastatin and the single enantiomeric form. Drilling down to the expiry (including SPC) details of the latter family and further to the claims shows the period of protection is longer in some countries, but that the scope is restricted to the calcium salt. At this point a reason for the appearance of alternative salt forms (Magnesium) in the US DMF filings becomes clear. Given that the enantiomer/salt patent may offer some months extra protection over the basic molecule (racemate) patent, circumventing the former could offer a proactive generic competitor a valuable head-start over competitors who wait to use the calcium salt. The abundance of litigation alerts demonstrates the importance of these patents as they are subject to litigation in every major territory, details of which can be analysed by clicking through to a judgment summary.
Table 3: Atorvastatin Key Patent Indicator
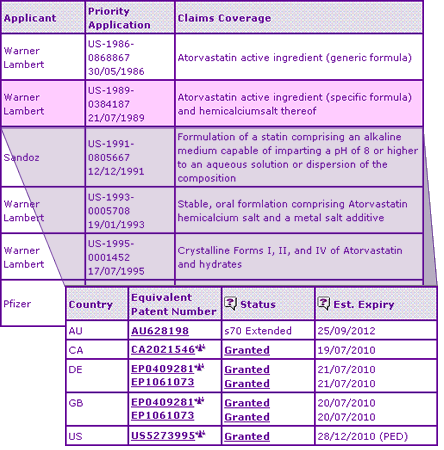
In addition to the above active ingredient patent families, four later-expiring patent families are identified by the Key Patent Indicator, two relating to formulations, one relating to the polymorphic form I, and one relating to a combined Atorvastatin/Amlodipine dosage form or kit. The polymorphic form patent further explains the presence of numerous polymorphic forms, and indeed amorphous forms in the US DMF filings, as generic competitors try to avoid awaiting expiry of this later key patent. The two key formulation patents (one in the name of Novartis, thought to originally have been developed for Fluvastatin) combined with the inherent physicochemical differences accompanying the use of a new molecular form would be expected to result in a large amount of required effort in formulating generics. This effect can be clearly seen in the Patent Category Distribution graph (Figure 1), based on comprehensive patent data[2].
Figure 1: Atorvastatin Patent Category Distribution
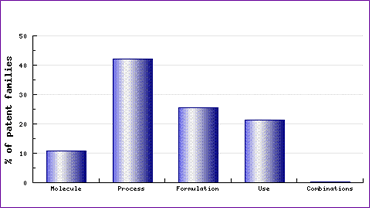
This graph also shows that process patents form a significant part of the patent protection surrounding the Atorvastatin product. This is partly due to the effort made in attempting to prepare and purify alternative molecular forms, which also represent a disproportionate amount of patent filings compared to other molecules analysed.
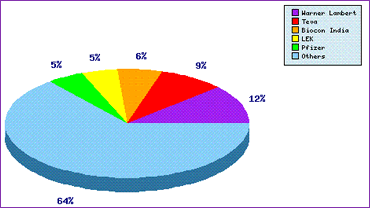
The question of who these patent filings belong to can be answered specifically by the Top Patent Applicants graph (Figure 2), which also shows what proportion of the patent filings are to applicants with lower research and development activity than these top five i.e. how many other applicants are proactively developing this molecule.
Figure 2: Atorvastatin Top Patent Applicants
The patent filing trends graph shows more generally who the filings are likely to have originated from (innovator or generic) by drawing a vertical line around the launch date of 1996. To the left of this Launch Line patents in the name of the innovator are predominantly found. The number, type and timing of these patent filings represent the lifecycle management activities undertaken by the innovator. The effectiveness of this innovator activity can often be judged by the response in patent filings to the right of the Launch Line, which are predominantly in the name of generic competitors.
Figure 3: Atorvastatin Patent Filing Trends
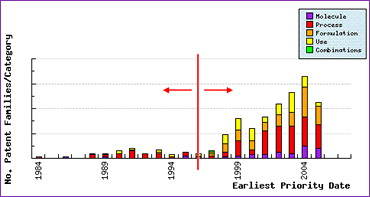
The large response in patent filings post-launch demonstrates the complex array of patents laid down by the innovator in relation to the Atorvastatin molecule. In particular, the size of the response in certain patent categories is often an amplification of the strength of the same category of patents filed by the innovator pre-launch. For example, the presence of a single innovator polymorphic form patent pre-launch has resulted in a raft of molecular form filings post-launch, suggesting that this patent alone has created a significant development hurdle for those generic companies turning early attention to Atorvastatin, and perhaps indicating the first question to ask a potential API supplier. A further question that may be answered from this graph is whether it is too late for an in-house API or finished-dose development. Often it is useful to understand at what stage others are at in terms of developing the product and where their effort has been focussed, before deciding whether it is too late to take on the project and therefore to start looking at licensing-in options.
In summary, the market for Atorvastatin has been well protected by the innovators beyond the life of the active ingredient patents, resulting in even broader protection being laid down by generic competitors attempting to secure a monopoly in relation to their own R&D activities. Despite all of this patenting activity on both sides of the innovator/generics divide it appears that more than a few generic products will likely arrive on the market as soon as the basic molecule patents expire. The answer to the question asked by many generics companies as to whether to add this molecule to their generics pipeline is therefore dependant on whether or not they started development many years ago and, if not, whether they are prepared to pay a premium to licence in product from one of the companies who did.
Comprehensive data for patent families relating to Atorvastatin, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
[1] This data also forms the basis of the patent search strategy and inclusion policy applied to the corresponding Pipeline Developer report outlining patents relating to development of generic equivalent of this product.
[2] Full details of patents represented in Patent Landscape Analysis graphs are accessible with a corresponding Pipeline Developer subscription.
|