SPC News June 2007
LAST NEWS ON SPC in EU countries
Medicinal and Plant Protection SPCs in Europe
About ten thousand nine hundred SPC applications have been filed in Europe from January 1991 to December 2006:
● about 9850 for medicinal products regulated by EC Regulation 1768/92 or National laws
● about 1050 for plant protection products regulated by EC Regulation 1610/96 or National laws.
EU SPC Regulation entered in force in Bulgaria and Romania on 1st January 2007. Until May 2007, 14 SPCs were published in Romania.
COURT AND PATENT OFFICE DECISIONS AFFECTING SPCs
Recent ECJ decision
Yissum brought an action before the national court against the decision of the British Patent Office to refuse its application for an SPC for calcitriol referring to EP0129003 patent (composition for use in topical treatment of skin disorders comprising calcitriol and a suitable carrier) and to Galderma marketing authorisation for Silkis® ointment.
The High Court of Justice (England & Wales), Chancery Division (Patents Court) decided to refer the following question to the Court of Justice for a preliminary ruling:
"In a case in which the basic patent protects a second medical application of a therapeutic agent what is meant by product in Article 1(b) of the Regulation No 1768/92 and in particular does the application of the therapeutic agent play any part in the definition of product for the purpose of the Regulation?"
In its decision of 17 April 2007 in case C-202/05, ECJ ruled that:
Article 1(b) of Council Regulation No 1768/92 is to be interpreted as meaning that in a case where a basic patent protects a second medical use of an active ingredient, that use does not form an integral part of the definition of the product."
So, Yissum SPC for calcitriol should be rejected.
National decisions for atorvastatin containing drugs
● Hemicalcium salt of atorvastatin: EP0409281
In The Netherlands and The United Kingdom the patent was declared invalid for anticipation and obviousness, in September 2006 and October 2005, respectively.
In Germany there is a nullity action.
● Atorvastatin: EP0247633
In The Netherlands and The United Kingdom the patent was declared valid. So, atorvastatin remains protected until November 2011 by SPCs referring to this patent.
In Germany there is a nullity action.
● Other patents
Note that, in May 2007, 4 Norwegian patents covering atorvastatin intermediates or amorphous atorvastatin were declared invalid (appeal from Pfizer).
EPO decision affecting Fosavance® drugs
Merck & Co. EP0998292 patent, relating to the use for inhibiting bone resorption of oral dosage forms containing 8.75 to 140 mg of alendronic acid and its salts, according to a schedule of 3 to 16 days, was revoked by EPO on 30 June 2006.
Note that Merck & Co. filed on 19 May 2004 a patent application relating to combinations of a bisphosphonate and a vitamin D compound, such as combination of alendronate and vitamin D3; it could protect Fosavance® drug until 2024 if a patent is granted.
SPCs for aceclofenac
Almirall-Prodesfarma SPCs for aceclofenac refer to EP0119932 and to 2 different MAs, Portuguese MAs 9737312 and/or 9737304 of 19 March 1990 and British MA 08448/0001 of 24 April 1995:
● SPCs referring to Portuguese MAs expired in March 2005 in The United Kingdom, Italy, The Netherlands and Sweden and
● SPCs referring to British MA will expire in March 2009 in Austria, Belgium, Germany, France and Luxembourg.
MEDICINAL PRODUCT SPCs IN EUROPE
Medicinal Product SPCs in Europe from 1991 to 2006
About 9850 SPC applications for medicinal products regulated by EEC Regulation 1768/92 or National laws have been filed in European countries, from January 1991 to December 2006, and published until May 2007.
In the graph, is shown, for each country, the number of SPCs filed from 1991 to 2006, dispatched according to their status (May 2007).
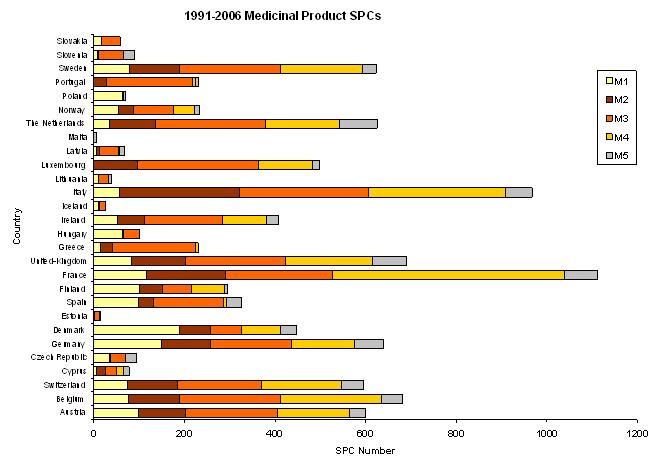
Where:
M1 = SPC applications
M2 = granted SPCs in force
M3 = granted SPCs not yet in force
M4 = expired, lapsed, invalid SPCs
M5 = rejected or withdrawn SPC applications
Medicinal Product SPCs in Europe in 2006
How many SPCs?
632 SPCs filed in 2006 were published at the end of May 2007.
The figures below represent the number of SPCs filed in 2006.
In the first graph, is shown the number of SPCs sorted by country.
In the second graph, is shown the number of SPCs sorted by therapeutic activities.
Medicinal Product SPCs filed in 2006
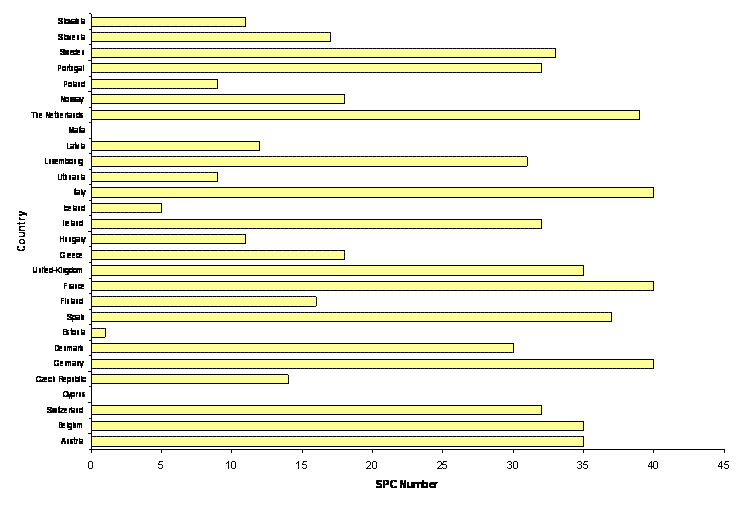
Note that Cyprian Patent Office did not publish any SPC information since the entry in force of EC regulation in Cyprus (May 2004).
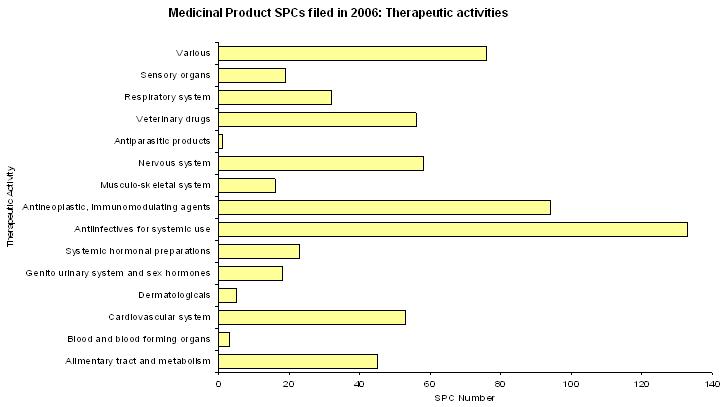
21.0 % of these SPCs relate to antiinfectives for systemic use, 14.9 % to antineoplastic and immunomodulating agents, 9.2 % to drugs for nervous system, 8.4 % to drugs for cardiovascular system and 8.9 % to veterinary drugs.
Which medicinal products?
The 632 SPCs filed in 2006 and published until May 2007 cover about 90 different products (active ingredients, combinations of active ingredients, or formulations).
We noted 30 products, for which were lodged at least 8 SPCs. These products can be roughly sorted according to their therapeutic category:
Alimentary tract and metabolism: rimonabant (24), insulin aspart (10)
Cardiology: ivabradine (14), sitaxentan (10), olmesartan medoxomil + hydrochlorothiazide (10), amlodipine + atorvastatin (8)
Hormonal contraceptives: drospirenone (11)
Calcium haemostasis: parathyroid hormone (19)
Antivirals: tipranavir (17), entecavir (15)
Vaccines: rotavirus vaccines (25 SPCs for 2 vaccines), varicella-zoster vaccine (12)
Antibacterials: tigecycline (19), daptomycin (15)
Antimycotics: posaconazole (20)
Antineoplastics: clofarabine (10), erlotinib (16), sorafenib (13), natalizumab (47)
Bone diseases: alendronate + cholecalciferol (15)
Analgesics: fentanyl (17)
Anti-Parkinson drugs: rotigotine (18)
Antidepressants: atomoxetine (14)
Obstructive airway diseases: omalizumab (29)
Ocular antineovascularisation agent: pegaptanib (18)
Iron chelating agent: deferasirox (12)
Detoxifying agent for antineoplastic treatment: palifermin (45)
Veterinary: antibacterials (cefovecin: 12), antihelmintics (emodepside + praziquantel: 29)
Who filed Medicinal product SPCs and for which products?
497 SPCs out of these 632 SPCs were lodged by 24 holders.
For these 497 SPCs, the figure below represents the number of SPCs sorted by holder.
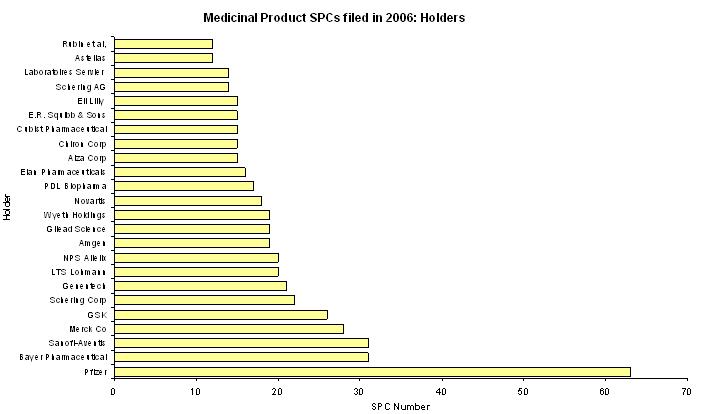
We can note for three of these holders:
PFIZER (63 SPCs)
Tipranavir:
17 SPCs in BE, CH, CZ, DE, ES, FI, GB, GR, IT, LU, LV, NL, NO, PL, SE, SI, SK, referring to 1995 Pharmacia & Upjohn patent and to August 2005 Swiss MA or to October 2005 EU MA, with an extension of 5 years. There were SPCs filed in 2005 in FR, IE, LT, PT.
Erlotinib:
17 SPCs in AT, BE, CZ, DK, ES, FR, GB, GR, IE, IT, NL, NO, PL, PT, SE, SI, SK, referring to June 1995 or March 1996 Pfizer + OSI Pharmaceuticals patent and to March 2005 Swiss MA with an extension of 4 years and about 9 months or to September 2005 EU MA with an extension of 5 years. There were SPCs filed in 2005 in CH, DE, LU.
Cefovecin:
12 SPCs in AT, BE, DE, DK, ES, FR, GB, IE, IT, LU, NL, SE, referring to Pfizer 1991 patent and to June 2006 EU MA with an extension of 5 years.
Amlodipine + atorvastatin:
7 SPCs in AT, CH, ES, IS, LU, LV, PT, SI, referring to 1998 Pfizer patent and to July 2005 French MA with an extension of 1 year and about 11 months and 1 SPC in Switzerland referring to July 2006 Swiss MA with an extension of 2 years and about 11 months.
Maropitant:
5 SPCs in DE, LU, NL, PT, SK, referring to 1992 Pfizer patent and to September 2006 EU MA with an extension of 5 years.
Other products:
Ceftiofur: 1 SPC filed in CH; Eletriptan: 1 SPC filed in HU and tulathromycin: 1 SPC filed in PL (corresponding SPCs formerly filed in other countries) and varenicline: 1 SPC filed in NO.
BAYER (31 SPCs)
Emodepside + praziquantel:
17 SPCs in AT, BE, CZ, DE, DK, ES, FI, FR, GB, IE, IT, NL, NO, PL, PT, SE, SK, referring to December 1994 or January 1995 Bayer patents and to July 2005 EU MA, with an extension of 5 years and 1 SPC in Switzerland referring to September 2005 Swiss MA with an extension of 5 years.
Note 17 Astellas SPCs for emodepside or emodepside + praziquantel, 14 filed in 2006 in AT, BE, CH, ES, FR, GB, IE, IT, NL, PL, PT (refused), SE and 3 filed in 2005 in DE, DK, LU, referring to March 1993 Astellas patents and to the same 1st MA.
Sorafenib:
13 SPCs in AT, BE, DE, FI, FR, GR, HU, IE, LT, LU, LV, NL, PT, SI, SK, referring to January 2000 Bayer patents and to July 2006 EU MA with an extension of 1 year and about 6 months and 1 SPC in Switzerland referring to Mars 2006 Swiss MA with an extension of 1 year and about 3 months.
SANOFI-AVENTIS (31 SPCs)
Rimonabant:
25 SPCs in AT, BE, CZ, DE, DK, EE, ES, FI, FR, GB, GR, HU, IE, IT, LT, LU, LV, NL, NO, PL, PT, SE, SI, SK, referring to Sanofi-Aventis 1993 (SK) or 1994 or 1998 (LV, IS, EE) patent and to June 2006 EU MA with an extension of 5 years for SPCs referring to 1993 or 1994 patents and an extension of 3 years and about 3 months for SPCs referring to 1998 patent.
Other products:
Insulin glulisine: 3 SPCs in CZ, NO, SK and DTPa-IPV-Hib vaccine: 2 SPCs in NL, PT (corresponding SPCs formerly filed in other countries) and fumagillin: 1 SPC in FR.
PLANT PROTECTION PRODUCT SPCs IN EUROPE
About 1050 SPC applications for plant protection products regulated by EC Regulation 1768/96 or National laws have been filed in European countries from February 1997 to December 2006.
The figure below represents the number of SPCs sorted by country. For each country, is shown the number of SPCs dispatched according to their status (May 2007).
1991-2006 Plant Protection Product SPCs
How many SPCs?
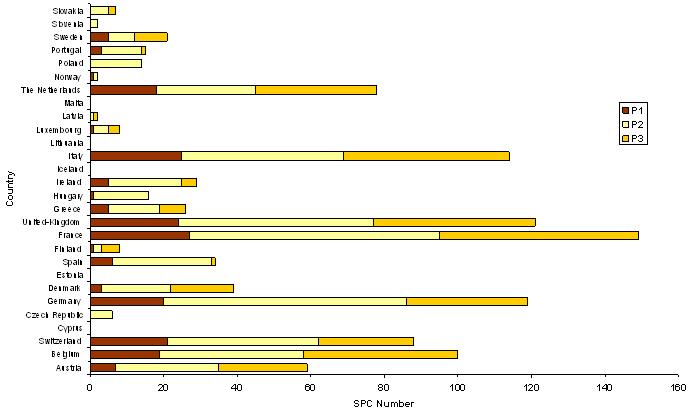
Where
P1 = granted SPCs in force
P2 = SPC applications and granted SPCs not yet in force
P3 = expired, lapsed, invalid SPCs and rejected or withdrawn SPC applications
We can rank countries by number of SPCs, which are an indication of the importance of the market:
● at least 100 SPCs: Belgium, Germany, France, The United-Kingdom and Italy
● 50 to 100 SPCs: Austria, Switzerland and The Netherlands
● more than 20 SPCs: Denmark, Spain, Greece, Ireland and Sweden.
Who filed Plant Protection SPCs?
836 SPCs out of these 1057 SPCs were lodged by 9 holders.
For these 836 SPCs, the figure below represents the number of SPCs sorted by holder.
Plant Protection SPC Holders

