Leighton Howard is a patent information expert with extensive generics experience. He has worked within the professional patent information industry and major generic pharmaceutical firms and now provides consultancy services to the generics industry and government organisations via his company XIP Pty Ltd. He is also the founder of GenericsWeb and remains instrumental in maintaining the high quality and accuracy of data contained in Pipeline Patent Intelligence. Please email any comments or queries. |
|
Drug In Focus: Alendronic Acid
Much attention was recently drawn to Mercks Fosamax (Alendronic Acid) bone resorption inhibitor medicine due to the purported reinstatement of a patent by the European Patent Office (EPO) relating to a once-weekly dosing regimen.
This months drug in focus will analyse the patent protection in respect of the Fosamax product line with a view to launching generic equivalents, based on the information contained in a GenericsWeb Pipeline Selector Report.
The General Information (Table 1) for Alendronic Acid shows that the active ingredient is present in Fosamax as the trihydrate form of the sodium salt, in both oral tablet and oral solution dosage forms. Of critical importance is the 70mg strength, which is the equivalent weekly dose indicated for treatment of post-menopausal osteoporosis, accounting for the vast majority of Fosamax prescriptions.
Table 1: Alendronic Acid General Information
Inspection of the Regulatory Information demonstrates that no data exclusivity is in force for this product in major markets, most of which have published marketing approvals for generic equivalents with the notable exception of the US.
Analysis of the Key Patent Indicator (Figure 1) for this molecule shows that the patent family protecting the chemical compound was not broadly patented by the original applicant, Gentili, and so lacked patent protection in some key markets, for example Australia and Canada. Revocation of the UK equivalent patent also left the door open for generic competition in a major EU market whilst the US market is protected from generic entry until February 2008. However, four further patent families are identified as protecting a molecular form, formulations and dosing regimens as approved for the innovator product.
Table 2: Alendronic Acid Key Patent Indicator
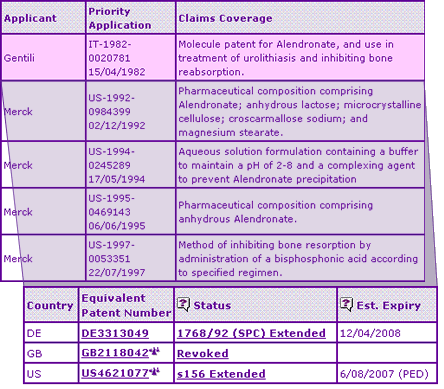
The Orange Book entry of a patent relating to the anhydrous sodium salt is confusing, based on the current labelling of the product in the USA which clearly states that the trihydrate form is in the article of commerce. The patent families relating to the commercial formulation do not appear to have constrained generic developers significantly, as indicated by the multitude of formulation patents filed in relation to this active ingredient shown in Figure 2, based on the comprehensive patent searching that forms the basis of the Pipeline Developer report.
Figure 1: Alendronic Acid Patent Category Distribution
The patent that media articles would have readers believe has taken some generic manufacturers by surprise is that relating to the 70mg dosage regimen. Whilst the original European patent in the family was revoked in early 2006, four divisional applications were filed prior to this which gave rise to four further pending European patents, all of which seek to protect the 70mg formulation. The grant of one of these applications in March 2007 was the action that was often referred to as the reinstatement of a revoked patent, but it by no means took generic competitors by surprise, with two filing opposition proceedings on the day of publication of the granted patent, and two more within the following week, despite having a further 9 months to lodge such an opposition. The results of the opposition, as well as the final outcome of the other three pending European equivalents in the family will not be known for a long time. However the withdrawal of Great Britain as a designated state from the granted patent by Merck, the applicant, would probably not have been expected and offers some certainty for a major EU market at least in the shorter term.
One further patent family of key concern to generic developers in certain markets due to international variation in patent legislation is that protecting the trihydrate salt per se. Whilst in most jurisdictions the claims of the patents relating to the salt per se were not granted or have been revoked, the patent still constrains generic competition in Australia, where the only generic equivalent is in the form of the monohydrate sodium salt. This demonstrates the need for consideration of patent protection in individual markets prior to embarking on a global development strategy.
Finally, the Patent Filing Trends graph for Alendronic Acid (Figure 3) tells a familiar story for Merck, a company who is very experienced in managing the life-cycle of their blockbuster products with many patent filings prior to the mid-90s launch of the Fosamax product. The response in patent filings by generic competitors post-launch demonstrates the additional effort required to circumvent the intricate web of patents laid down by the innovator, causing many developments to be abandoned or put on hold until such time as problem patents have expired.
Figure 2: Alendronic Acid Patent Filing Trends
In summary, Fosamax is an example of a product that has been well protected by the innovator, and remains an uncertainty in terms of the risks of generic launch of certain strengths. This demonstrates the need for comprehensive patent research and ongoing updating of information to ensure that the right decisions are made in terms of generics development and launch.
Comprehensive data for patent families relating to Alendronic Acid, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
|