Leighton Howard is a patent information expert with extensive generics experience. He has worked within the professional patent information industry and major generic pharmaceutical firms and now provides consultancy services to the generics industry and government organisations via his company XIP Pty Ltd. He is also the founder of GenericsWeb and remains instrumental in maintaining the high quality and accuracy of data contained in Pipeline Patent Intelligence. Please email any comments or queries. |
|
Drug In Focus: Fluvastatin
Whilst a significant amount of attention is being paid to blockbuster product Lipitor (Atorvastatin) by generic developers, this months Drug in Focus will examine the patent landscape for a similar product, Lescol (Fluvastatin), based on information provided in the corresponding GenericsWeb Pipeline Selector report.
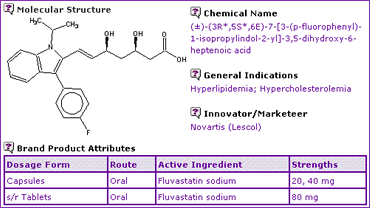
The General Information for Fluvastatin (Table 1) denotes that the active ingredient is available in the form of the sodium salt as instant-release capsule and sustained release tablet forms, indicated for treatment of hyperlipedemia and hypercholesterolemia.
Table 1: Fluvastatin General Information
The Key Patent Indicator lists three patents including the active ingredient patent and two formulation patents, one of which only applies to the sustained release product. Unusually for drug products of this era, no additional molecular form or use patents have been listed in the Orange book, however this has often been shown to be an unreliable indicator of worldwide patent protection of the innovator product.
Table 2: Fluvastatin key Patent Indicator
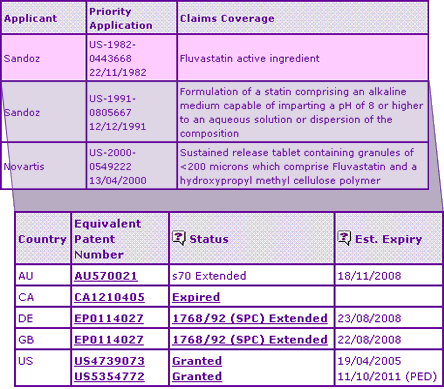
The active ingredient patent family view confirms that the SPCs granted in major European territories will expire in August 2008, whilst the corresponding extension in Australia expires after a full five year term in November 2008, based on the later authorisation date. Further disparity in patent systems around the world are seen with the Canadian patent equivalent having already expired, and the US patent having a Paediatric extension until April 2012, delaying generic competition for over 3 years beyond Europe.
The key formulation patent that is common to both the instant-release and the sustained release products relates to pH stabilization of the active ingredient. This patent is also listed in the Orange Book patents for Atorvastatin (perhaps under license to Pfizer) and is likely to create similar formulation issues when developing non-infringing generics for Fluvastatin.
The Patent Category Distribution graph (Figure 1) demonstrates this, wherein formulation patents feature prominently in patent filings as a response by generic developers. However, the biggest problem in developing non-infringing generics appears to lie in the patents that are not listed in patent registers such as the Orange Book: process patents. This implies that sourcing a non-infringing active ingredient without intricate knowledge of the patent filings in this area is likely to be very problematic.
Figure 1: Fluvastatin Patent Category Distribution
Although one sustained release formulation patent is found in the Orange Book, analysis of the Pipeline Developer report for Fluvastatin uncovers a further granted (but opposed) European patent to the innovator, which broadly protects sustained release formulations. No US equivalent is granted hence this patent family cannot be identified by Orange Book analysis methods, hence it is crucial to have undertaken extensive patent analysis to uncover and enable monitoring of patents such as this to ensure that a generic development does not meet with unexpected obstacles prior to launch, wasting valuable time and resources. The outcome of this opposition, as well as the prosecution of other patent applications in this family should be monitored carefully by those companies intending to launch a sustained release generic version of Lescol.
The Patent Filing Trends graph (Figure 2) shows that, prior to launch, the majority of innovator filings focused on protecting processes of Fluvastatin, hence the large post-launch response by generic competitors to circumvent these and protect any identified holes in the innovator protection by filing their own patents. However, heavy generic filings in other areas such as molecular form and formulations indicates that API synthesis is not the only area where development issues are being faced by generic competitors. The DMF filing relating to the Amorphous active ingredient by at least one generic competitor signifies that polymorph issues may be among them.
Figure 2: Fluvastatin Patent Filing Trends
The Fluvastatin patent filing trends thus shows an in depth graphical analysis of areas which could prove to be a challenge for generic competition and allows developers of generics to see, at a glance, potential areas which would need to be addressed for the development of a successful generic.
In summary, Fluvastatin is a heavily patented drug, both by the innovator and subsequent generic filings. Similar to Atorvastatin, lifecycle management appears to have created significant obstacles for generic competitors beyond Active Ingredient patent expiry, far beyond those found by basic patent searching. Monitoring the prosecution of certain key patents, and gaining an awareness of the full patent landscape is therefore critical in ensuring launch of a successful generic product.
Comprehensive data for patent families relating to Fluvastatin, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
|