Leighton Howard is a patent information expert with extensive generics experience. He has worked within the professional patent information industry and major generic pharmaceutical firms and now provides consultancy services to the generics industry and government organisations via his company XIP Pty Ltd. He is also the founder of GenericsWeb and remains instrumental in maintaining the high quality and accuracy of data contained in Pipeline Patent Intelligence. Please email any comments or queries. |
|
Drug In Focus: Perindopril
Coversyl (Perindopril Erbumine) is an ACE inhibitor which accounts for 31% of Serviers 2.8 billion global revenues . This large reliance on one active ingredient may well explain why Servier has taken a proactive approach in defending its top selling product from generic competition by asserting its intellectual property portfolio. Februarys Drug in Focus will review the extent of protection for Perindopril products from generic competition based on the corresponding GenericsWeb Pipeline Selector report, with a specific focus on the major geographical markets.
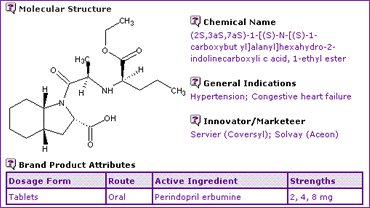
The General Information (Table 1) demonstrates that Coversyl is available in three strengths of oral tablet, containing the erbumine (t-butylamine) salt of Perindopril as the active ingredient. This does not imply that formulating generics equivalents would be problematic, however when considering the five chiral centres shown in the molecular structure, it starts to become apparent that this is no easy molecule to synthesise.
Table 1: Perindopril General Information
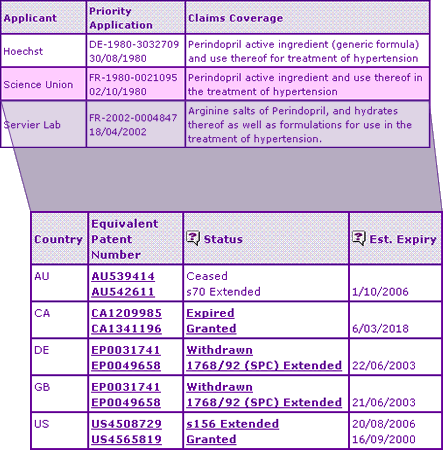
The Key Patent Indicator (Table 2) outlines the most significant patent protection for the Perindopril products and shows that the Supplementary Protection Certificate, often considered to be the ultimate determinant of the date of generic competition in Europe, expired in June 2003, with corresponding extensions in Australia and the US expiring late 2006. But in this case, the Key Patent Indicator is not elaborate enough to fully reveal the extent of Serviers strong patent protection of this product.
Table 2: Perindopril key Patent Indicator
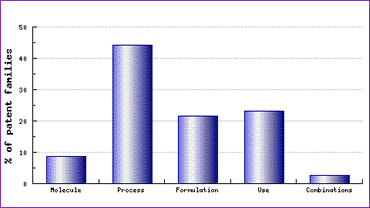
Analysis of the Patent Category Distribution for Perindopril (Figure 1) shows a strong presence of claims in the areas of molecular form and processes. The population of these categories due to innovator patent filings is often amplified by generic competitors attempting to circumvent the patents of concern, themselves filing patents to protect their R&D efforts and potentially minimise the effect of price erosion due to large numbers of other generic manufacturers entering the market. As such, these graphical analyses tell a convincing story about the patent protection of this product and the task of finding a non-infringing route of synthesis is therefore not be taken lightly.
Figure 1: Perindopril Patent Category Distribution
Indeed, previously settled litigation between Servier and Niche Generics had focussed on process patents, whilst recent interlocutory injunctions granted in the UK appear to be based on assertion of molecular form patents relating to the alpha polymorph. Opposed by no less than eight separate companies, including the majority of those with generic authorisations in the UK, European patent EP1296947 was the cause of shockwaves through the generics industry last September when the EPO decided to reject the opposition and grant claims to the alpha polymorph per se, despite opponents claims that a product containing it was in the public domain before the patent was applied for. This decision has been appealed at the EPO and the patent is being challenged nationally in the UK courts, whilst opposition proceedings are underway in Australia. Examination of equivalent patent applications is underway at both the US and Canadian patent offices, no doubt leaving an amount of uncertainty over generics currently under development in those markets.
Last-minute switching of active ingredients in Australia to the protected arginine salt created confusion amongst pharmacists and confusion alike due to the different strength labelling. Both the TGA and the PBS deemed the new salt to be bioequivalent to the erbumine salt, apparently removing any commercial advantage for Servier, and resulting only in a great deal of angst amongst the Australian pharmacist community.
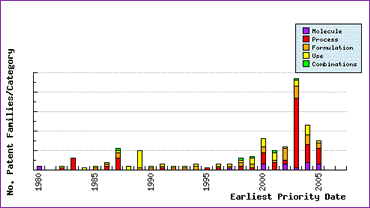
Whilst Serviers protection of certain molecular forms and the manner in which they apply it commercially may be somewhat questionable, study of the Patent Filing Trends (Figure 2) for Perindopril give further insight into the reason for strong protection of the manufacturing processes. Strong process patent activity in the years immediately preceding launch of the product in the first major market in 1988 offer protection until after SPC expiry and will constrain all but the most informed generic competitors from launching without risk of legal action by Servier. Potential entrants should therefore be very cautious in ensuring that all patents protecting intermediates and synthetic schemes have been identified and their scope understood prior to commercialising a generic product. Large amounts of process patent filings by generic applicants from 2000 onwards suggest that there are ways around the Servier process patents, but these may ultimately be monopolised by their inventors who are just as keen to protect their investment in R&D as Servier appear to be.
Figure 2: Perindopril Patent Filing Trends
To summarise, it appears that Servier realised early on in the development of Perindopril that it would be of significant value and has used lifecycle management to extend its monopoly marketing period to beyond the normal 15 years. A complex web of molecular form and process patent applications, combined with seemingly low standards of patent examination has created initial problems for generic competitors, but it should not be long before alternative development strategies and aggressive litigation by proactive generic developers results in non-infringing generics being available in at least some major markets. Aggressive litigation and product switching strategies are signs of an innovator who is desperate to hold on to its monopoly, hence a great deal of caution is required by smaller generic companies who are not prepared to fight for their right to compete in the generic market.
Comprehensive data for patent families relating to Perindopril, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
|