Medicinal Product SPCs in Europe from 1991 to 2005
About 8950 SPC applications for medicinal products regulated by EEC Regulation 1768/92 or National laws have been filed in European countries from January 1991 to December 2005, and published until August 2006.
The situation differs from one country to another due to the importance of the markets and/or to the applicable laws and/or to the date of entry in force of the EU Regulation.
524 SPCs have been filed in the 10 new European countries (number of SPCs from 6 for Malta to 90 for Hungary).
The figures below represent the number of SPCs filed from 1991 to 2005. In the first graph, is shown, for each country, the number of SPCs dispatched according to their field of use: human, veterinary or human/veterinary. In the second graph, is shown, the number of SPCs sorted by therapeutic activities.
1991-2005 SPCs: Field of use
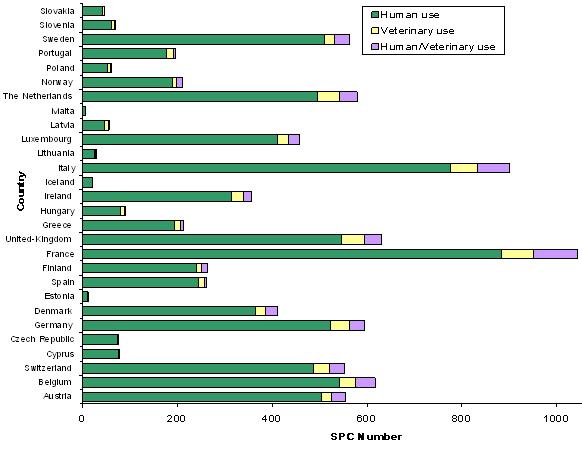
88% of SPCs relate to drugs for human use, 6% to drugs for veterinary use and 6% to drugs for human/veterinary use.
1991-2005 SPCs: Therapeutic activities
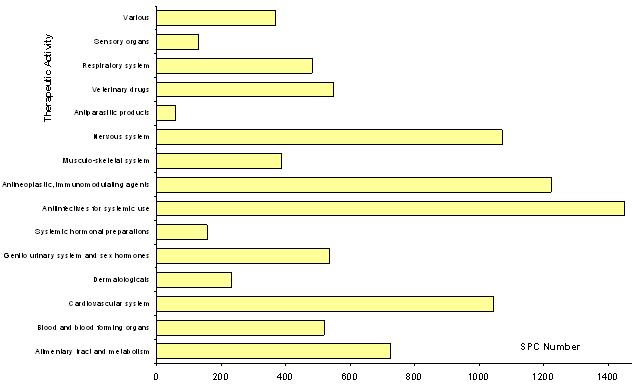
16.2% of SPCs relate to antiinfectives for systemic use, 13.7 % to antineoplastic and immunomodulating agents, 11.9% to drugs for nervous system and 11.7% to drugs for cardiovascular system.
Medicinal Product SPCs in Europe in 2005
How many SPCs?
715 SPCs filed in 2005 were published at the end of August 2006.
The figures below represent the number of SPCs filed in 2005. In the first graph, is shown, for each country, the number of SPCs dispatched according to their field of use: human, veterinary or human/veterinary. In the second graph, is shown, the number of SPCs sorted by therapeutic activities.
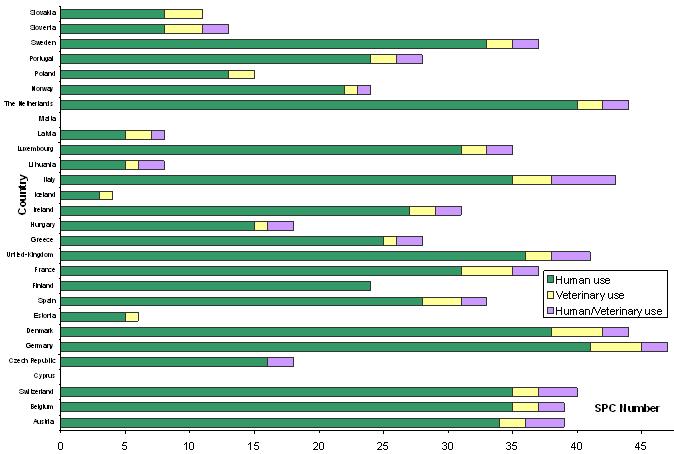
Medicinal Product SPCs filed in 2005: Field of use
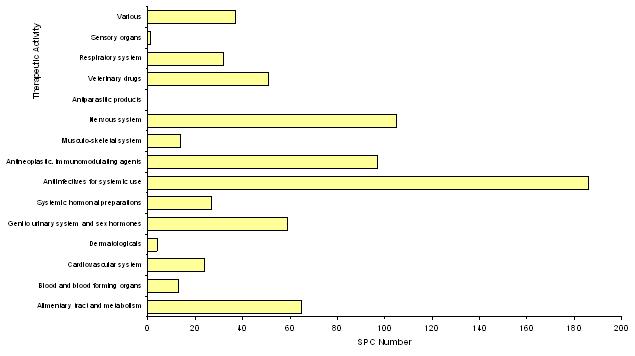
Medicinal Product SPCs filed in 2005: Therapeutic activities
Which medicinal products?
The 715 filed in 2005 and published SPCs cover about 110 different products (active ingredients, combinations of active ingredients, or formulations).
We noted 31 products, for which were lodged at least 7 SPCs. These products can be roughly sorted according to their therapeutic category:
Alimentary tract and metabolism: palonosetron (18), nitisinone (15), insulin glulisin (15), insulin aspart (9)
Cardiology: ezetimibe + simvastatin (14)
Urology: darifenacin (37), solifenacin (8)
Hormones: cinacalcet (16), pegvisomant (10)
Antivirals: enfuvirtide (24), emtricitabine + tenofovir (24), abacavir + lamivudine (19), atazanavir (18)
Vaccines: vaccines containing combined bacterial and viral antigens against diphtheria, tetanus, pertussis, and hepatitis B and/or poliomyelitis and/or Haemophilus influenzae type b (75 SPCs for 8 vaccines),
Cancer: bevacizumab (40), pemetrexed (21)
Bone diseases: strontium ranelate (9)
Analgesics: ziconotide (14)
Anti-parkinson drugs: rasagiline (28)
Antidepressants: duloxetine (41)
Obstructive airway diseases: omalizumab (15), ciclesonide (14)
Magnetic resonance imaging contrast media: gadoxetic acid (18)
Veterinary: anti-infectives (ceftiofur crystalline: 16), antiinflammatory (firocoxib: 22)
Who filed Medicinal product SPCs and for which products?
541 SPCs out of these 715 SPCs were lodged by 20 holders.
For these 541 SPCs, the figure below represents the number of SPCs sorted by holder.
Medicinal Product SPCs Main Holders
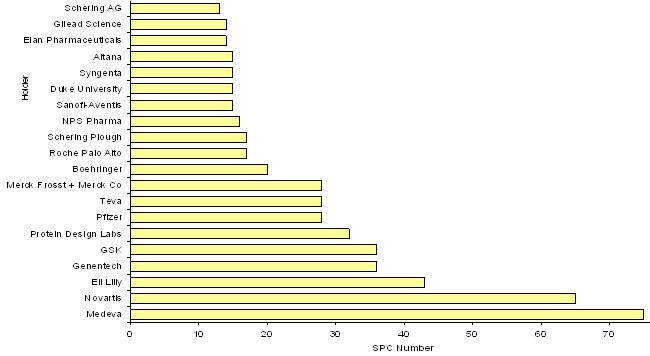
We can note for four of these holders:
NOVARTIS (65 SPCs)
Atazanavir:
17 SPCs in AT, BE, DE, DK, ES, FI, FR, GB, GR, IE, IT, LU, NL, NO, PT, SE, SI referring to 1997 Novartis patent and to March 2004 EU MA, with an extension of about 1 year 11 months and 1 SPC in Switzerland referring to May 2004 Swiss MA with an extension of about 2 years 1 month. Until now there are no published SPCs in the 10 new EU countries (except Slovenia).
Darifenacin:
37 SPCs in AT, BE, CZ, DE, DK, ES, FI, FR, GB, GR, HU, IE, IT, LU, NL, NO, PL, PT, SE, SK, referring to 1990 and/or 1996 Novartis patents and to March 2004 EU MA with an extension of 5 years for SPCs referring to 1990 patent and 3 years 2 months for SPCs referring to 1996 patent.
Everolimus:
4 SPCs in CZ, HU, IT, PL, referring to Novartis 1996 patent and to July 2003 Swedish MA with an extension of about 4 years 10 months and 1 SPC in Switzerland referring to April 2005 Swiss MA with an extension of about 5 years.
There were SPCs filed in 2004 in AT, BE, DE, DK, ES, FI, FR, GR, HU, IE, IT, LU, NL, NO, PL, PT, and filed in 2003 in Sweden. There is no SPC in UK.
Other products:
Pimecrolimus: 2 SPCs filed in IT, SK, and mycophenolate: 3 SPCs filed in BE, IT, GR (corresponding SPCs formerly filed in other countries).
MEDEVA (75 SPCs)
SPCs referring to Medeva 1990 patent for 8 vaccines:
DTPa-HB vaccine (Infanrix HepB):
10 SPCs in AT, BE, DE, DK, ES, GB, IT, LU, NL, SE, referring to July 1997 EU MA with an extension of 2 years and about 3 months and 1 SPC in Switzerland referring to October 2000 Swiss MA with an extension of 5 years.
DTPa-HB-IPV vaccine (Infanrix Penta):
11 SPCs in AT, BE, CH, DE, DK, ES, GB, IT, LU, NL, SE, referring to October 2000 Swiss MA with an extension of 5 years.
DTPa-HB-IPV-Hib vaccine (Infanrix Hexa):
11 SPCs in AT, BE, CH, DE, DK, ES, GB, IT, LU, NL, SE, referring to October 2000 Swiss MA with an extension of 5 years.
DTPa-Hib vaccine (Infanrix Hib):
10 SPCs in AT, BE, DE, DK, ES, GB, IT, LU, NL, SE, referring to October 1996 German MA with an extension of 1 year and about 6 months and 1 SPC in Switzerland referring to February 1997 Swiss MA with an extension of 1 year and about 10 months.
DTPa-IPV vaccine (Infanrix IPV or Infanrix Tetra):
10 SPCs in AT, BE, DE, DK, ES, GB, IT, LU, NL, SE, referring to August 1996 French MA with an extension of 1 year and about 4 months and 1 SPC in Switzerland referring to August 1999 Swiss MA with an extension of 4 years and about 4 months.
DTPa-IPV vaccine (Repevax):
7 SPCs in AT, BE, DE, DK, GB, LU, NL, referring to November 2001 German MA with an extension of 5 years.
DTPa-IPV-Hib vaccine (Infanrix IPV/Hib or Infanrix Quinta):
11 SPCs in AT, BE, DE, DK, ES, GB (2), IT, LU, NL, SE, referring to June 1997 French MA with an extension of 2 years and about 2 months and 1 SPC in Switzerland referring to September 1998 Swiss MA with an extension of 3 years and about 5 months.
DTPa vaccine:
1 SPC in CH, referring to September 1995 Swiss MA with an extension of 4 months and about 10 days.
ELI LILLY (43 SPCs)
Duloxetine:
21 SPCs in AT, BE, CZ, DE, DK, ES, FR, GB, GR, HU, IE, IT, LT, LU, LV, NL, NO, PL, PT, SE, SI, referring to Eli Lilly 1987 or 1995 patents and to August 2004 EU MA with an extension of 5 years for SPCs referring to 1987 patent and an extension of 2 years and about 1 month for SPCs referring to 1995 patent and 1 SPC in Switzerland referring to December 2004 Swiss MA with an extension of 5 years.
Pemetrexed:
17 SPCs in AT, BE, DE, DK, FI, FR, GB, GR, HU, IT, LT, LU, LV, NL, NO, SE, SI, referring to Eli Lilly 1990 or 1998 patents and to September 2004 EU MA with an extension of 5 years for SPCs referring to 1990 patent (1 year for 1998 patent) and 1 SPC in Switzerland referring to January 2005 Swiss MA with an extension of 5 years.
Atomoxetine:
3 SPCs in DE, NL, NO, referring to Eli Lilly 1996 patent and to August 2004 British MA with an extension of 3 years 3 months and about 15 days (1 SPC filed in UK in 2004).
TEVA (28 SPCs)
Rasagiline:
28 SPCs in AT, BE, CZ, DE, DK, EE, FR, GB, GR, HU, IE, IT, LT, LU, LV, NL, PT, SE, SK, referring to Teva Pharmaceutical and Technion Research & Development Foundation 1991 or 1994 or 1996 patents and to February 2005 EU MA with an extension of 5 years for SPCs referring to 1991 or 1994 patents (3 years and about 9 months for SPCs referring to 1996 patents).
Alice de Pastors
September 2006

