Drug In Focus August 06: Eplerenone
After a short deviation from the regular Drug in Focus feature for the past few months, this months article reviews patent protection of the congestive heart failure drug Inspra (Eplerenone), based on information contained in the GenericsWeb Pipeline Selector report.
This product is an unusual case in that marketing authorisations in major territories were first granted very late in the life of the Eplerenone molecule patent protection, resulting in what appears at first glance to be a heavy reliance on data exclusivity to prevent generic competition. However, a review of the patent protection shows that Pfizer have an armoury of patent protection that will keep generic developers (or at least their lawyers) busy.
The Eplerenone General Information (Table 1) indicates that the product is available as 25 or 50mg film-coated tablet for treatment of congestive heart failure, suggesting that the product would be relatively simple to develop from a generic perspective.
Table 1: Eplerenone General Information
The Key Patent Indicator (Table 2) confirms that the extensions on the Eplerenone active ingredient patent expire in the US in April 2007 and in Germany in April 2009, benefiting from a full, five-year SPC. However the corresponding SPC application in the UK was refused, resulting in an expiry date of April 2004. Similarly a 2004 expiry date applies to the Australian equivalent patent due to the post-expiry authorisation date for the product, resulting in ineligibility for a s70 extension.
Table 2: Eplerenone Key Patent Indicator
The new Market Authorisations feature in the Pipeline Selector report shows that there are no generic equivalents authorised in Australia or the UK despite lack of molecule patent protection. Analysis of the first marketing dates offers an explanation for this, with European data exclusivity provisions preventing the filing of generic applications until 2010 in 6 year countries and 2014 in 10 year countries or for centralised applications. This implies that first European generic authorisations could be expected in late 2011 and 2015 respectively. Countries with shorter data exclusivity periods such as the US might see generic activity somewhat earlier.
In addition to the molecule patent family, two other patent families have been identified in the Key Patent Indicator, one relating to combinations or Eplerenone with an ACE inhibitor and the other, perhaps of greater concern, relating to compositions comprising micronised Eplerenone. The latter patent family contains granted equivalents in most major territories with expiry dates of 2019 and beyond. A granted s70 extension on the Australian equivalent patent strongly implies that the commercial product contains such micronised active ingredient, suggesting that achieving bioequivalence in a generic product may be difficult for any or the territories where patents in this family are still in force.
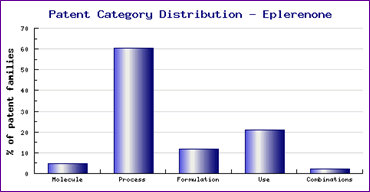
Study of the Patent Risk Analysis section, based on comprehensive patent data (details of which are accessible in the corresponding Pipeline Developer subscription) indicates that patenting activity relating to this drug is mostly concentrated in the area of process patents (Figure 1).
Figure 1:
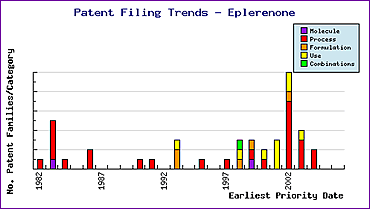
The Eplerenone Patent Filing Trends (Figure 2) shows that, whilst the later of these patents will likely act as stumbling blocks for lesser informed generic developers, a significant portion of these will expire prior to the data exclusivity, providing opportunities to develop non-infringing active ingredients. However, caution should be exercised in all areas of generic development because Pfizer are responsible for a number of patent filings, including molecule process, formulation and use patents, shortly before market authorisation. These are intended to protect the extensive effort Pfizer has invested in innovations to gain approval for this seemingly difficult product, and are likely to be vigorously defended, based on recent litigation activities by this company. Such patenting is likely to meet with responsive patent filings by generic competitors, creating even more complexity for those companies starting this development at a later time.
Figure 2:
In summary, the market for Eplerenone appears to be protected beyond molecule patent expiry, mainly by virtue of data exclusivity provisions. Furthermore, some proactive life-cycle management by Pfizer later in the product authorisation stage should act to ensure that generic competitors will not flood the market as soon as data exclusivity allows.
Comprehensive data for patent families relating to Eplerenone, based on professional patent searching, may be accessed by subscribing to GenericsWeb Pipeline Developer reports which include twelve monthly updates to keep you abreast of recently published patents and applications, status changes and litigation. GenericsWeb Pipeline Selector reports are available for any active ingredient upon request.
Leighton Howard
Managing Director
GenericsWeb
August 2006
l.howard@genericsweb.com
To register for GenericsWeb's free monthly newsletter 'INNsight', click here