The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic Sildenafil equivalents of Pfizers blockbuster phosphodiesterase-5 inhibitor (Viagra® and Revatio®). In the first nine months of 2012, global sales of Viagra® and Revatio® increased by 3% and 5%, generating US$1.5 billion and US$414 million, respectively, compared to the same period in 2011.
General information
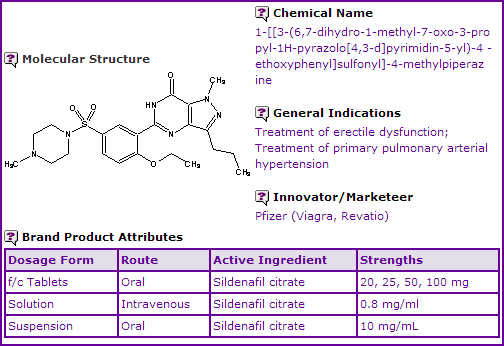
Sildenafil is currently available as film-coated tablets in eq. 25, 50 and 100 mg base strengths for use in the treatment of erectile dysfunction. It is also available in three dosage forms used in the treatment of pulmonary arterial hypertension. All dosage forms contain the active ingredient in the form of the citrate salt.
Figure 1: General Information table for Sildenafil
INN Constraint Comment
Ordinarily, the most significant family preventing the launch of a generic is the family protecting the API (see Figure 2). The patent members of GB9013750A expired in Canada in June 2011 and in the US in September 2012 following the expiry of a 6 month paediatric extension and a 283 day s156 extension. In Europe, the equivalent patent has been granted, with an SPC extending it until June 2013 (see Figure 3). The Spanish translation of the European patent originally protected only processes for the preparation of Sildenafil and therefore would have been assumed to allow generic entry whilst remaining in force. However, a revision to the Spanish translation (ES2071919) of the European patent to allow the product claim for the Sildenafil molecule
per se, was upheld in November 2010 in accordance with Article 12 of Royal Decree 2424/1986 provided by Article 27.1 and 70 of TRIPS. The decree allowed a procedural avenue for product claims in Spain. Prior to the decision several companies launched or applied for a market authorisation for generic Sildenafil in Spain, triggering Pfizer to file a motion for preliminary injunction which was granted in February 2012, preventing sale or launch of generic Sildenafil until patent/SPC expiry. However, the Spanish SPC is showing a validity flag which is a proprietary indicator applied by GenericsWeb to suggest that the SPC expiry date is based on an incorrect first EU market authorisation date. In this case, the Spanish SPC expiry date, listed in the OEPM registry and not amended at the time of publication, was based on the EU centralised approval for Sildenafil on 14/09/1998, not the earlier Swiss approval date of 22/06/1998, which was recognised in the EU state of Liechtenstein.
Families with priorities GB9311920A and GB9925970A (see Figure 2) seek to protect both of the authorised indicated uses of Sildenafil, so must be considered when determining rights which constrain generic entry. Patents in the family with priority GB9311920A claim the use of Sildenafil in the treatment of erectile dysfunction. The European member (EP0702555) has been revoked following opposition proceedings at the EPO. A recent decision, on 8 November 2012, by the Supreme Court of Canada found the Canadian member (CA2163446) invalid and void. Subsequently Pfizer filed a motion to amend the Courts decision. The validity and enforceability of US6469012 was upheld after a challenge from Teva. The patent has been granted a 6 month paediatric extension, so will constrain the launch of a generic Sildenafil product until expiry in May 2020 in the USA unless other challenges to the patents validity are successful. Sales of Viagra® in the US contributed approximately 85% (US$1.27 billion) of its sales revenue in the first nine months of 2012. Protection conferred by this use patent is key to maintaining Pfizers exclusivity its largest market.
Patents in the GB9925970A family claim the use of Sildenafil for the treatment of pulmonary hypertension. However, the European and Canadian members of this family have been invalidated. Although this indication is no longer protected by patent in Europe, 10 years of data exclusivity is available for orphan indications (Article 8.1 of Regulation (EC) No. 141/2000) and therefore may prevent submission of applications for generic market authorisation of Sildenafil products carrying this indicated use until October 2015. At the time of publication, there was no issued US equivalent, the Orange book thus suggesting that generic entry for products carrying the pulmonary hypertension indication would be allowed following the expiry of the API patent (see Figure 4). However, Pipeline Patent Intelligence identifies two US applications in this family with potential expiry dates in 2029 if either patent is issued.
Patent families IB700003W, US7558098P, GB9822238A and US11033598P are not considered to be a constraint to generic competition as the protected technologies are likely to be circumvented.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Sildenafil
Figure 3: Patent Family View priority application GB9013750A
Figure 4: Marketing Authorisations for products containing Sildenafil in Key Countries