|
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
Drug In Focus: Capecitabine
The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic Capecitabine equivalents of Hoffman-La Roches blockbuster anti-neoplastic drug Xeloda®. In the first half of 2012, Xeloda® sales generated CHF 763 million globally, a 14% increase in sales from the first half of 2011.
General information
Capecitabine is currently available as film-coated oral tablets in 150 and 500 mg strengths in most major markets. The Capecitabine molecule was discovered and marketed by Hoffmann-La Roche.
Figure 1: General Information table for Capecitabine
INN Constraint Comment
At the time of publication, patents within the API families (Figure 2) are likely to prevent the launch of generics until expiry. The first patent family EP87116926A (17/11/1987) claims the Capecitabine molecule by general formula (Figure 2). Patents in this family expired in Australia, Canada and the US after the expiry of 765 day s156 term extension. European patents have been extended by supplementary protection certificate (SPC) until June 2013 in France and the UK, and November 2013 in Germany and Spain. GenericsWebs proprietary SPC analyser identified the German and Spanish SPCs as having not used the first European MA date in their applications, and therefore the later expiry date of the SPCs in these countries may be invalid.
However, the patent protection from the second family, EP92121538A (18/12/1992), which claims the Capecitabine molecule by specific formula, are considered to further constrain the launch of generics until expiry. European patents in Core countries will expire in December 2013, after the expiry of SPCs on the general molecule patent (Figure 3). In Australia, the patent term has been extended by s70 until April 2014. Patent infringement litigation has been identified for the US and Canada members of this family. In the US, Hoffmann La Roche filed multiple suits against Teva, Mylan, Roxane, Accord and Intas following their filing for ANDAs with paragraph IV certifications to US5472949, which expires in December 2012. However, the proceedings were dismissed due to Hoffmann La-Roche failing to prove they were the owner of the patent. An appeal by Hoffmann La-Roche was voluntarily dismissed in May 2012. In Canada, Hoffmann La-Roche filed a suit against Teva, in relation to their ANDs filing, for an order of prohibition until the expiry of CA2103324 in November 2013.
Data exclusivity is generally not considered to constrain generic entry for this molecule in major territories.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Capecitabine
Figure 3: Patent Family View priority application EP92121538A
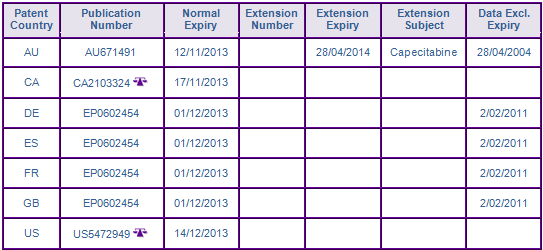
Figure 4: Marketing Authorisations for products containing Capecitabine in Key Countries
Amongst the UK approvals, authorisations have been awarded to Glenmark, AstraZeneca, Actavis and AET for both the 150 and 500 mg dosage strengths. These approvals came through the decentralised procedure where the UK was listed as the reference member state. Other generic companies, Teva, Krka and Accord, have been awarded market authorisation through the centralised procedure. This suggests generic competition is likely to be strong upon patent expiry in Europe. Currently no other generic authorisations have been identified in other regions.
Figure 5: Top Patent Applicants
Note that among the top individual patent filers are generic companies Dr Reddys and Generics UK, their patent filings relating to molecular form and preparation of the API. These are in an attempt to circumvent innovator patents and protect their own technological developments. In total there exist over 120 individual patentees assigned to patent families relating to Capecitabine.
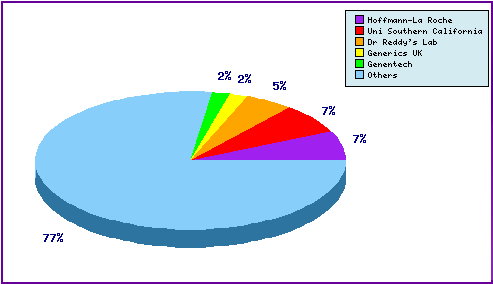
Figure 6: Patent Filing Trend
Note the responsive post approval patenting by generic companies relating to processes for preparing the API and formulations, compared with the low pre-launch patenting by the innovator. This suggests that perhaps more could have been done to better manage the life-cycle of Capecitabine post molecule patent expiry.
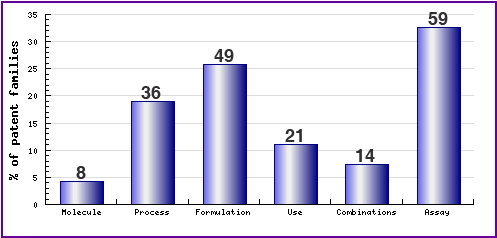
Figure 7: Patent Category Distribution
The types, number and relative distribution of patents that have been filed for Capecitabine obtained via a comprehensive patent search (Pipeline Developer report) are shown in this graphic. Note the amount of assay patents - this is representative of different methods of determining patient suitability for a particular drug regimen. Of the formulation patents, 55% relate to general formulations and methods and 18% relate to oral formulation.
In summary, Hoffmann La-Roche have taken some basic measures to protect the Capecitabine molecule, obtaining patents that protect the general (EP87116926A) and specific (EP92121538A) forms of the API. Although, patents in the general API family were extended in Europe and the US, the protection conferred by the later, specific API family is likely to extend Hoffmann La-Roches exclusivity, preventing the launch of a generic until its expiry. Marketing authorisation holders identified in the UK, ANDAs with Paragraph IV in the US and ANDs filing in Canada are indicative of the high interest shown by generic companies for Capecitabine in these markets. It is therefore likely that a large portion of the market share will go to generic competitors upon patent expiry and as the costs reduce due to multiple competitors launching simultaneously.
Do you want find out more about how we can assist your generic drug development?
Share this article:
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
|