|
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
Drug In Focus: Ziprasidone
The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic Ziprasidone equivalents of Pfizers blockbuster atypical antipsychotic drug (Geodon® and Zeldox®). Global Geodon®/Zeldox® sales generated US$1.02 billion in 2011.
General information
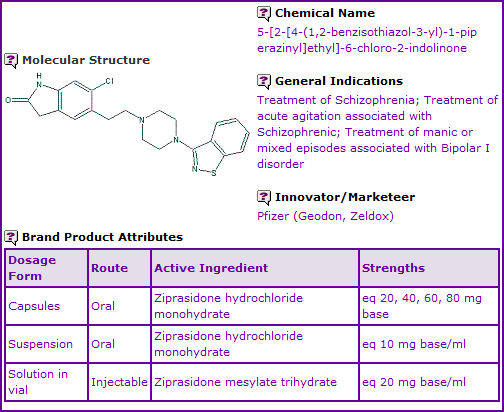
Ziprasidone is currently available as oral capsules in eq. 20, 40, 60 and 80 mg base strengths and an eq. 10 mg base/ml oral suspension, used in the treatment of schizophrenia and manic or mixed episodes associated with bipolar I disorder. It is also available as 20 mg/ml intramuscular (IM) injection for use in the treatment of acute agitation associated with schizophrenia. The Ziprasidone molecule was discovered by Pfizer.
Figure 1: General Information table for Ziprasidone
INN Constraint Comment
The most constraining patent families generally preventing the launch of a generic relate to the API US8700423W and US14688688A; both claiming the Ziprasidone molecule per se (Figure 2). Patents in US8700423W have been extended by supplementary patent certificate (SPC) in certain European countries expiring in February 2013 and in Australia by an s70 patent term extension expiring in March 2013 (Figure 3). However the Spanish portion of EP0281309 in this family only claims processes for the preparation of Ziprasidone, so may be circumvented whilst remaining in force. Genericswebs proprietary SPC analyser identified and flagged a discrepancy with the market authorisation date used for the application of the German SPC not citing the first market authorisation date in Europe, and therefore the expiry date may be invalid. The patent term was not extended by SPC in France or the UK as Ziprasidone is currently not market authorised in these countries.
API patent protection expired in the US in March 2012, after benefiting from a full 5 years of patent term extension (Figure 4).
Patent family US6806997P claims the use of Ziprasidone in the treatment of a psychiatric condition, including bipolar I disorder and dementia. The US and Canadian members, if granted with current claims, are considered to be a constraint for the authorised use of Ziprasidone in the treatment of Bipolar I disorder until expiry. The claims of the granted European member limit its use to the treatment of dementia, so do not protect any authorised use of Ziprasidone. The Australian member has ceased, therefore not considered to be a constraint for this indication.
Patent families US32390189A, US1920496P and US13626899A relate to current market authorised dosage forms of Ziprasidone. Whilst not considered as such a considerable constraint as the API patent families, they are still worthy of consideration. Patent families US32390189A claims Ziprasidone mesylate trihydrate, the salt form used in the IM injectable form. Patent family US1920496P claims injectable formulations containing an inclusion complex of Ziprasidone and a cyclodextrin, also protecting the currently marketed IM dosage form. Patent family US13626899A protects the currently marketed oral suspension Ziprasidone formulation. The patents claim specific inactive ingredients, a polysorbate and colloidal silicon dioxide, and/or quantities thereof, and may be circumvented by generic re-formulation. However, formulating aqueous suspension could be problematic due to Ziprasidone free base and its acid addition salts having low solubility and are difficult to wet. The apparent lack of generics for these dosage forms in off-patent territories, further suggests these patents are a constraining factor.
Patent families US32390189A, US93617992A and US8922998P are not considered to be a constraint to generic competition as the protected technologies are likely to be circumvented.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Ziprasidone
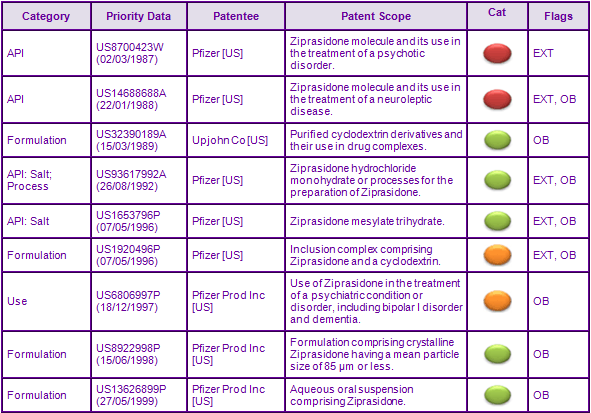
Figure 3: Patent Family View priority application US8700423W
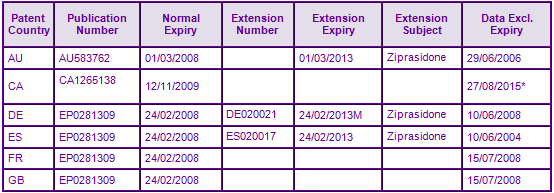
Figure 4: Patent Family View priority application US14688688A

Figure 5: Marketing Authorisations for products containing Ziprasidone in Key Countries
In the US Lupin Pharms, Apotex and Dr Reddys Lab gained market authorisation upon expiry of the molecule patent and Sandoz in June; all for the capsule form in all dosage strengths. Ziprasidone is not an authorised medicine in the UK, however several generic companies have been identified in Europe holding market authorisation for Ziprasidone capsules. Launch is expected upon expiry of the molecule patent in Europe. No generics have been identified in Canada due to a later MA and unexpired data exclusivity preventing an applicant from filing an application for a generic MA until August 2013 and launching until August 2015. No generics have been identified in Australia due to molecule patent protection preventing the launch of generics. There is currently no evidence of any market authorisation obtained by generic companies relating to the oral suspension or the IM injectable dosage forms.

Figure 6: Top Patent Applicants
A representation of patentees who have filed the most patent families for this INN. Note that almost one third of patent applications were filed by the innovator Pfizer and the prominence of generic filing in the top 5.
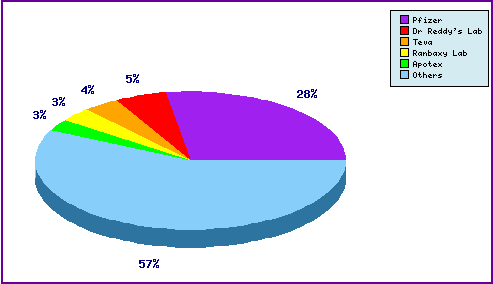
Figure 7: Patent Filing Trend
A representation of patentees who have filed the most patent families for this INN. Note that almost one third of patent applications were filed by the innovator Pfizer and the prominence of generic filing in the top 5.
Figure 8: Patent Category Distribution
The types, number and relative distribution of patents that have been filed for Ziprasidone obtained via a comprehensive patent search (Pipeline Developer report). A large majority of the patents relate to process and formulation. Amongst the formulation, there has been patenting by generic companies in an effort to circumvent innovator patents, much of this being formulation patents in the circumvent innovator patents for the capsule form. There is a lack of patenting activity by generic companies relating to oral suspension or the injectable formulations, further suggesting the difficulty in developing aqueous Ziprasidone formulations.
In summary, the API patent remains in force in Europe and Australia posing a significant barrier to the launch of generics. Loss of API patent exclusivity in the US has seen the entry of generic competitors and this is likely to be the case in Europe and other markets. Pfizer have gone to significant lengths to maintain their monopoly for Ziprasidone, by developing and patenting different formulations. Pfizer still have exclusivity for IM injectable and oral suspension dosage forms. There is currently no evidence of any market authorisation obtained by generic companies for these dosage forms, suggesting the difficulty in developing aqueous Ziprasidone formulations and circumventing innovator patents.
Do you want find out more about how we can assist your generic drug development?
Share this article:
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
|