|
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
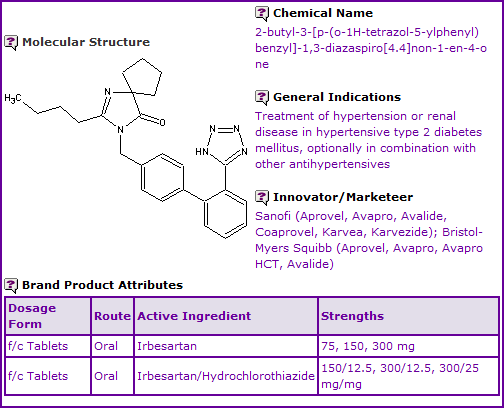
Drug In Focus: Irbesartan
The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic equivalents of Sanofi/Bristol Myers Squibbs antihypertensive drug Irbesartan (Aprovel®/CoAprovel®, Avapro®/Avalide®, Karvea®/Karvezide®). Sanofi reported global Aprovel®/CoAprovel® sales of 1.29 billion in 2011, a decrease of 2.4% from 2010.
General information
Irbesartan is currently available as oral film-coated tablets in 75, 150 and 300 mg strengths and, as a fixed-dose combination with Hydrochlorothiazide (HCTZ), in 150/12.5, 300/12.5, 300/25 mg/mg strengths. The Irbesartan molecule was discovered by Sanofi and is co-marketed under a collaborative arrangement with Bristol Myers Squibb.
Figure 1: General Information table for Irbesartan
INN Constraint Comment
Patent protection for the Irbesartan molecule (FR9003563A) expired in Canada in March 2011 and the US in March 2012 after benefiting from six months of paediatric extension to a 197 day s156 extension (see Figure 3). In Europe, patent protection of the Irbesartan molecule has been extended by Supplementary Protection Certificates (SPC) which expire in August 2012. However, the Spanish portion of EP0454511 only claims processes for the preparation of Irbesartan and as such may be able to be circumvented. Additional SPCs protecting the combination of Irbesartan and Hydrochlorothiazide have been granted and expire in October 2013, however GenericsWebs proprietary SPC analyser had identified the basic patent as being a C2 category, suggesting that the claims do not specify or identify all components of the combination that is the subject of the SPC, and therefore the SPC may be invalid. It is currently uncertain what mechanisms are available to generic competitors who wish to request amendment of the SPC by the individual patent offices who granted them. A s70 patent term extension protects the Irbesartan molecule from generic competition in Australia until February 2013.
The patent family based on priority US47261895A claims formulations comprising Irbesartan and co-formulations with Hydrochlorothiazide (see Figure 2). These patents protect the marketed formulations but are not considered to be a constraint to generic competition as the protected technology is likely to be circumvented by re-formulation.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Irbesartan
Figure 3: Patent Family View priority application FR9003563A
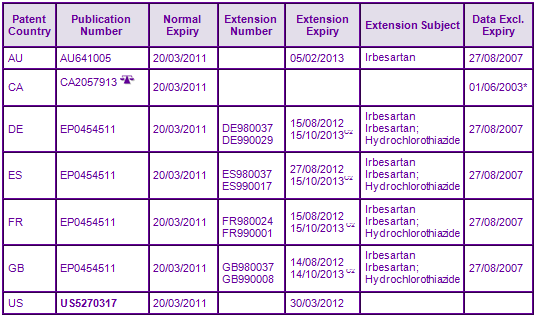
Figure 4: Marketing Authorisations for products containing Irbesartan in Key Countries
Amongst the US approvals, Teva is the only generic competitor to launch upon molecule patent expiry, offering both the single active and combination products. This is due to their 180-day market exclusivity for both Irbesartan products, having filed the first Abbreviated New Drug Applications with paragraph IV certifications, preventing rival generic companies from obtaining final market approval. Increased market pressure is expected when waiting generic competitors gain final market approval after the expiry of the 180-day exclusivity. The large number of generic authorisations awaiting launch in the UK and Australia is indicative of the likely competition the Aprovel® and CoAprovel® products will face upon extension expiry. Only a small number of the UK MA holders have been granted authorisations for the combination product, and so are poised to take advantage of the possibly weak SPC protection on this fixed dosage form and may launch upon expiry of the single-active SPC later this year, instead of waiting until 2013. Time will tell what these companies strategy for the UK launch is.

Figure 5: Top Patent Applicants
A representation of patentees who have filed the most patent families for this INN. Note the prominence of relevant applications filed by manufacturers of competing sartan drugs, Novartis (Valsartan), Takeda (Candesartan and Azilsartan), Merck (Losartan) and SmithKline Beecham (Eprosartan); as well as the diversity of applicants represented by the significant portion outside of the top five, largely representing generic companies.
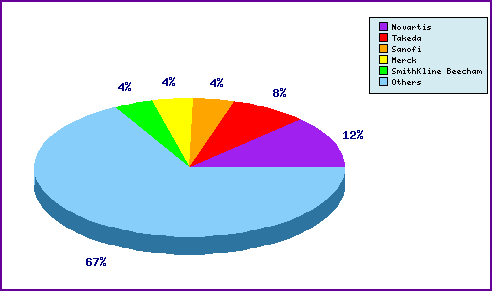
Figure 6: Patent Filing Trend
Represents the timing of the earliest priority filing date for each patent family identified for this molecule as well as the type of claims found in the applications. The majority of patent applications filed prior to the launch of the Irbesartan products are by innovator companies. Note the consistency in filings post launch filing is dominated by generics companies trying to circumvent innovator patents.
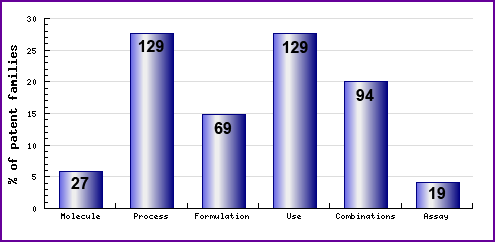
Figure 7: Patent Category Distribution
The types, number and relative distribution of patents that have been filed for Irbesartan obtained via a comprehensive patent search (Pipeline Developer report). Note the existence of equal number of process and use patents filed. Of the process patents over 50% of the patents filed relate to intermediates, suggesting broad patenting due to the similar molecular structures of some of the sartan drugs. The use patents cover an extensive range of indications which often differs to the currently marketed indications.
In summary, protection remains for the Irbesartan molecule in Europe as a result of SPCs and extended patents in Australia and as such represents a barrier to generic competitors. However, in certain European countries the basic molecule patent only protects processes for the preparation of Irbesartan and as such may have been circumvented, allowing generic competitors to launch prior to patent expiry. Strong generic competition is expected in Europe and other markets upon SPC expiry later this year, as evidenced by the volume of generic market authorisation holders in these markets. Further market pressure is expected in the US when other generic companies gain final market approval after the expiry of Tevas 180-day market exclusivity period.
Do you want find out more about how we can assist your generic drug development?
Share this article:
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
|