|
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
Drug In Focus: Clopidogrel
The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic equivalents of Sanofi and Bristol Myers Squibbs blockbuster platelet aggregation inhibitor Clopidogrel (Plavix® and Isocover®). US sales increased by 8% in 2011, generating US$6.62 billion of the US$7.09 billion gained globally; resulting in a global sale increase of 6%. Sales in non-US markets declined 9%.
General information
Clopidogrel is currently available as a film coated oral tablet in eq. 75 and 300 mg base strengths. Oral fixed-dose combinations with Aspirin are also available (CoPlavix®, DuoPlavix® and DuoCover®). The Clopidogrel molecule was discovered by Sanofi-Aventis and was co-developed and marketed under a strategic alliance with Bristol Myers Squibb.
Figure 1: General Information table for Clopidogrel
INN Constraint Comment
The most constraining patent families generally preventing the launch of generics relate to the API, patents in these families protect both the racemic mixture of Clopidogrel (FR8212599A) and the enantiomerically pure form and its salts (FR8702025A).
Patents in family FR8212599A (Figure 2), which claim the Clopidogrel molecule per se, have already expired. However, due to the later expiring API family FR8702025A, protection has been extended to 17 May 2012 in the US courtesy of a 6 month paediatric extension to the 1375 day s156 patent term extension. The term of the equivalent European patent has also been extended by supplementary protection certificates (SPC) until 16 February 2013. The completion of an agreed Paediatric Investigational Plan (PIP) has resulted in applications for a further 6 month paediatric extension to SPCs in major European countries (Figure 3). However the scope of the patent offering protection in many European countries is restricted to the Clopidogrel bisulfate salt, therefore is only considered to constrain generic equivalents containing this specific salt form. This has allowed proactive generic competitors to submit applications for market authorisation after the expiry of data exclusivity in Europe and consequentially launch generic versions of Clopidogrel using an alternate salt, prior to the expiry of the SPCs (Figure 4), e.g. the besylate salt from around 2008. The presence of generics in Australia and Canada, prior to patent expiration, can be attributed to the revocation of patents AU597784 and CA1336777, both being found invalid. This is due to orders for revocation being filed by Apotex in the respective countries.
Family US19033294A protects one of the approved indicated uses for Clopidogrel so is considered to be constraining for that indicated use only. The family contains only a US member and therefore only considered to be a constraint in the US. This patent has recently been removed from the Orange Book, the reason for which is not clear at the time of writing.
Families FR9008482A, FR9008749A and FR9712441A, identified as a result of granted Japanese patent term extension, claim intermediates or process for the preparation of Clopidogrel and are therefore not considered to be a constraint because the protected technologies are likely to be circumvented.
Family with priority FR9807464A claims polymorphic form II of Clopidogrel bisulfate; the crystalline form contained in Plavix. It is likely that generic competitors will develop alternate crystalline forms, therefore this family is not considered to be a constraint as it is likely to be circumvented.
Litigation overview
Extensive patent infringement litigation has been identified in the US, Bristol Myers Squibbs largest market for Plavix®, relating to the enantiomerically pure Clopidogrel molecule patent family (FR8702025A). Sanofi and Bristol Myers Squibb have file multiple infringement suits against Apotex, Dr Reddys, Cobalt, Teva and Sun Pharmaceuticals after they filed ANDAs with Paragraph IV certifications asserting invalidity or unenforceability of US4847265. For a brief period beginning 8 August 2006 Apotex launched their generic Clopidogrel product at risk, after the expiry of the 30 month stay, trying to benefit from their awarded 180-day market exclusivity for being first to file an ANDA with a Paragraph IV certification. Sanofi and Bristol Myers Squibb moved swiftly in response by filing a preliminary injunction to prevent sales of generic Clopidogrel until the resolution of litigation. The validity and enforceability of the US4847265 patent was upheld, preventing the launch of a generic until 17 May 2012.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Clopidogrel
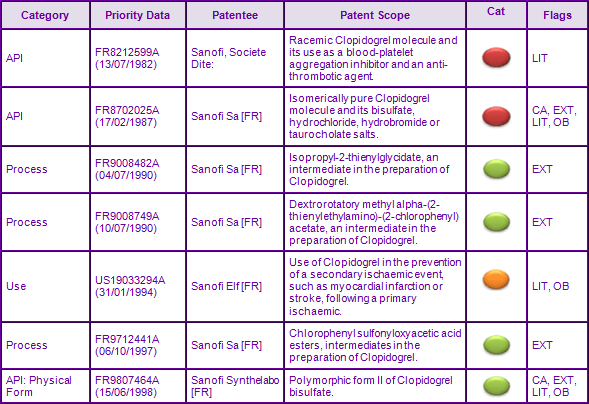
Figure 3: Patent Family View priority application FR8702025A
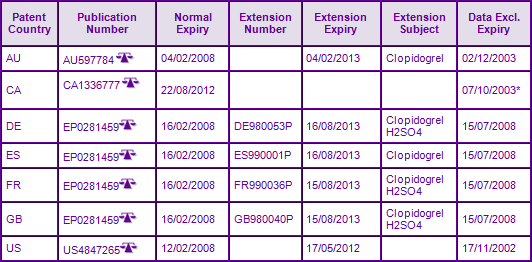
Figure 4: Marketing Authorisations for products containing Clopidogrel in Key Countries
Amongst the US approvals, tentative FDA approvals have been identified for generic Clopidogrel, awarded to Apotex, Dr Reddys, Mutual Pharma, Mylan, Roxane, Sun Pharma and Torrent. Launch is expected upon expiry of patent protection on 17 May 2012. Dr Reddys Laboratories is the only one to have a tentative market authorisation for the 300 mg strength dosage form. Generic Clopidogrel equivalents using the besilate, hydrobromide or hydrochloride are available in the UK, as indicated above. UK Market authorisations have also been granted for generic equivalents using the bisulfate salt, these are expected to be launched upon expiry of the SPC, suggesting that generic companies who identified the opportunity to develop a different salt to launch early in these major European markets were rewarded well for their efforts. Currently there have been no generic authorisations for the 300 mg dosage form in the UK.
Given that patents in the earlier molecule family for Clopidogrel (racemate) expired in 2003 in most countries, one may ask the question why it took so long for a generic version of Clopidogrel to appear on the European markets. The answer is of course that data exclusivity constraints prevented applications for a generic MA until July 2008 in some countries, leaving a potential launch date of late 2009 as a possibility. This underscores the reason why data exclusivity is an important second line of protection to patents, and should always be considered when planning an initial development strategy.
Figure 5: Top Patent Applicants
A representation of patentees who have filed the most patent families for this INN. Note that 21% are by the innovator Sanofi and the prominence of generic filing in the top 5.
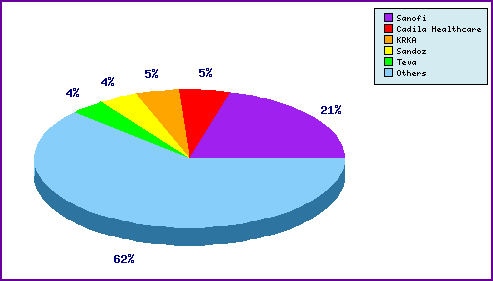
Figure 6: Patent Filing Trend
Represents the timing of the earliest priority filing date for each patent family identified for this molecule as well as the type of claims found in the applications. Note the strong response by generic companies post-launch and in the lead up to the expiry of data exclusivity, particularly in new molecule forms and process patents. New molecular forms were patented heavily by generic competitors to circumvent the bisulfate salt (enantiomer) patent as soon as the product showed promise as a blockbuster.
Figure 7: Patent Category Distribution
The types, number and relative distribution of patents that have been filed for Clopidogrel obtained via a comprehensive patent search (Pipeline Developer report). Note the volume of process patents, the majority of which relate to intermediates and methods of purification. Most of these have been filed by generic companies trying to circumvent innovator patents.
In summary, Sanofi and Bristol Myers Squibb have gone to significant lengths to prevent the launch of generic Clopidogrel products by filing patent families protecting the racemic and enantiomerically pure Clopidogrel. The protection from the enantiomerically pure patent family will prevent the launch of a generic in US until its expiry in May 2012. In Europe, generic competitors have been able to circumvent API patents by using an alternate salt. Sanofi and Bristol Myers Squibb have gone to additional lengths to protect their monopoly for Clopidogrel beyond that of the API families, resulting in patent filings by generic competitors as they try to circumvent patents and protect their own technologies. Bristol Myers Squibb quite rightly anticipates significant decline in sales upon patent expiry in the US.
|