The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic equivalents of Mercks migraine treatment Rizatriptan (Maxalt®). Rizatriptan products had global sales of USD $550m in 2010.
General information
<
Figure 1: General Information table for Rizatriptan
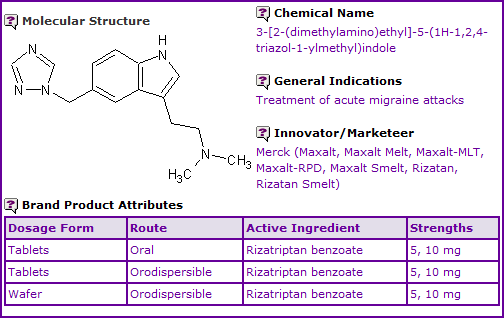
INN Constraint Comment
Whilst patent protection for the Rizatriptan molecule expired in Canada in January 2012, protection extends to December 2012 in the US (see Figure 3); this date includes an additional 6 months paediatric extension on top of an original s156 extension. Noteworthy is that Orange book listed patent US5602162 appears to specifically disclaim the Rizatriptan molecule claimed in related issued patent US5298520, yet a paediatric extension extends expiry of the 162 patent to 2014. Reliance on face value information contained in the Orange Book would therefore mislead the uninformed observer. Due to later authorisation of the product in Australia, the s70 extension on the molecule patent in this jurisdiction extends to June 2014. In Europe, many SPCs protecting the Rizatriptan molecule were granted with expiries in February 2013. Within the last few months however, Merck successfully complied with the agreed paediatric investigation plan (PIP) and has begun the process of applying for 6 month extensions to the terms of several SPCs, giving a potential expiry of August 2013.
Patent family US10448693A, the only additional key patent family identified for this INN, claims a freeze-dried unit bearing an identifying mark prepared in a container. Patents within this family are not considered to be a constraint to generic competition because the protected technology is likely to be circumvented.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Rizatriptan
 Figure 3: Patent Family View priority application GB9102222A
Figure 3: Patent Family View priority application GB9102222A
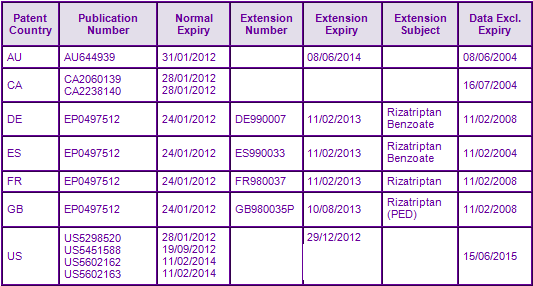 Figure 4: Marketing Authorisations for products containing Rizatriptan in Key Countries
Figure 4: Marketing Authorisations for products containing Rizatriptan in Key Countries
Amongst the US approvals, tentative FDA approvals have been identified for generic Rizatriptan Sodium, awarded to Cipla, Mylan, Sandoz AND Teva. In the UK, Generic authorisations awarded to Actavis, Aspire, Generics UK, Glenmark, Jenson, Sandoz, Teva and Winthrop is indicative of the likely competition Rizatriptan will face across Europe upon SPC expiry.
Figure 5: Top Patent Applicants
This chart shows a representation of patentees who have filed the most patent families for this INN. Note the prominence of relevant applications filed by manufacturers of competing triptans, Pfizer (Eletriptan) and Glaxo (Naratriptan, Sumatriptan) whose scope overlaps with Rizatriptan; as well as the diversity of applicants represented by the significant portion outside the top five.
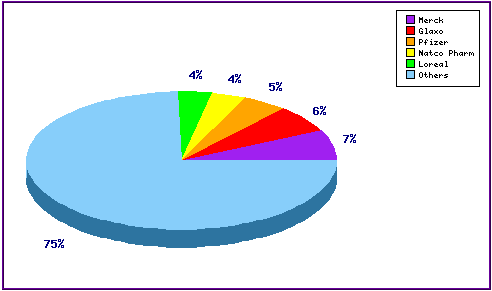
Figure 6: Patent Filing Trend
This chart represents the timing of the earliest priority filing date for each patent family identified for this molecule as well as the type of claims found in the applications. Note the strong responsive patenting by generic companies post-launch, particularly in the area of formulations.
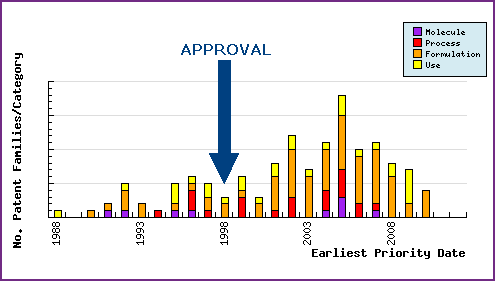
Figure 7: Patent Category Distribution
The types, number and relative distribution of patents that have been filed for Rizatriptan obtained via a comprehensive patent search (Pipeline Developer report). Note the dominance of formulation and use patent families. In addition to a wide range of oral formulations, several other administration-specific formulations have also been identified, including transdermal, nasal, buccal and inhalable formulations. The use patent families identified relate to a wide range of indications in addition to the currently approved migraine indication.