|
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
Drug In Focus: Montelukast
The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic equivalents of Mercks blockbuster asthma treatment Montelukast (Singulair®). Montelukast products had global sales of USD $5bn in 2010.
General information
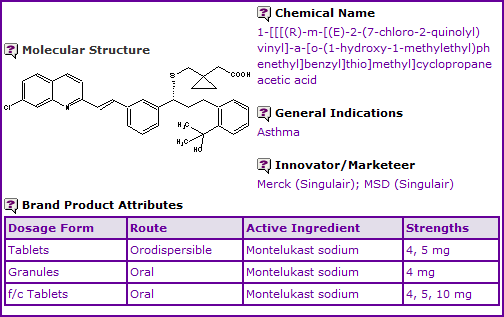
Montelukast is currently available in film coated tablet and orodispersible tablet formulations for once-daily administration, and also available as an oral granule formulation which is specifically designed for administration to paediatric patients.
Figure 1: General Information table for Montelukast
INN Constraint Comment
Whilst patent protection for the Montelukast molecule expired in Canada in October 2011, protection extends to August 2012 in the US (see Figure 3). In Europe, many SPCs protecting the Montelukast molecule were granted with expiries in August 2012. However, Merck successfully complied with the agreed paediatric investigation plan (PIP) and successfully applied for 6 month extensions to the terms of several SPCs, giving a potential expiry of February 2013 in some of Western Europes largest markets, namely France, Germany and the UK, as well as in many of the regions smaller markets. Due to later authorisation of the product in Australia, the s70 extension on the molecule patent in this jurisdiction extends to April 2013.
Patent family US17493193A claims crystalline Montelukast sodium and processes for its preparation (see figure 2). Patents within this family are not considered to be a constraint to generic competition because the protected technology may possibly be circumvented by the synthesis and use of different molecular forms and/or salts. Patent family US33954901P relates to the specific marketed oral granule formulation of Montelukast. Patents within this family are also not considered to be a constraint to generic competition because the protected technology is likely to be circumvented by generic re-formulation. Opposition against the European patent within this later family was filed by Hexal on the grounds of lack of novelty and inventive step which subsequently led to the decision to revoke the European patent, however this decision is currently under appeal.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Montelukast
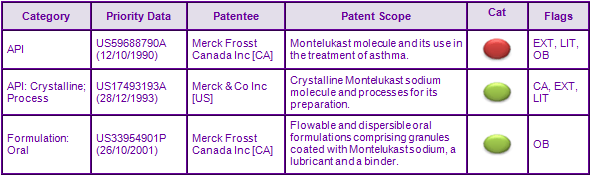
Figure 3: Patent Family View priority application US59688790A
Figure 4: Marketing Authorisations for products containing Montelukast in Key Countries
Amongst the US approvals, tentative FDA approvals have been identified for generic Montelukast sodium, awarded to Endo, Glenmark, Mylan, Roxane, Sandoz, Teva and Torrent. The large number of generic authorisations awaiting launch in the UK is indicative of the likely competition the Singulair product will face across Europe upon SPC expiry.
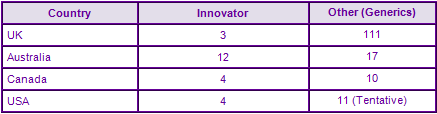
Figure 5: Top Patent Applicants
A representation of patentees who have filed the most patent families for this INN. Note the prominence of generic players Chemagis, Dr Reddys, Synthon and Teva in the top five, focusing primarily on molecular form and process patent applications. In total, over 90 individual applicants have been identified as filing patent applications relating to Montelukast.
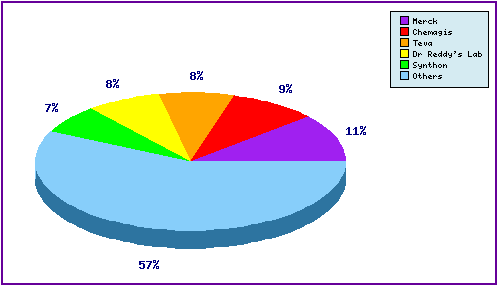
Figure 6: Patent Filing Trend
Represents the timing of the earliest priority filing date for each patent family identified for this molecule as well as the type of claims found in the applications. Note the consistent filing of molecular form and process patent applications since the initial approval of Montelukast in 1998, confirming the preferred strategy of generic manufacturers is to circumvent the crystalline form patent.
Figure 7: Patent Category Distribution
The types, number and relative distribution of patents that have been filed for Montelukast obtained via a comprehensive patent search (Pipeline Developer report). Note the dominance of process patent families which cover intermediates, the complete synthesis of Montelukast and the purification of Montelukast. Of the molecule patent families identified, the majority relate to various polymorphic forms of Montelukast and a range of Montelukast salts. In addition to oral formulations, several other administration-specific formulations have also been identified.
In summary, the API patent remains in force in most major territories and means there still exists a significant barrier to generic competition. The number of generics available in Canada, which were authorised just one day after Canadian molecule patent expiry, the large number of marketing authorisation holders identified in Australia and the UK and the number of Paragraph IV filings observed in the US is indicative of the critical interest shown by generic companies for Montelukast generics; indeed Merck predicts that it will lose substantially all US sales of its best-selling medication Singulair® in the first two years following expiry of the US patent, a prediction that will likely be mirrored in Europe following SPC expiry.
|