|
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
Drug In Focus: Vildagliptin
The following data is extracted from GenericsWeb Pipeline Patent Intelligence and is intended to give a brief outline of factors affecting the potential launch of generic equivalents of Galvus® Novartis dipeptidyl peptidase-4 (DPP-4) inhibitor containing Vildagliptin. Galvus® is indicated for the treatment of Type II diabetes with reported global sales of USD $391m in 2010.
General information
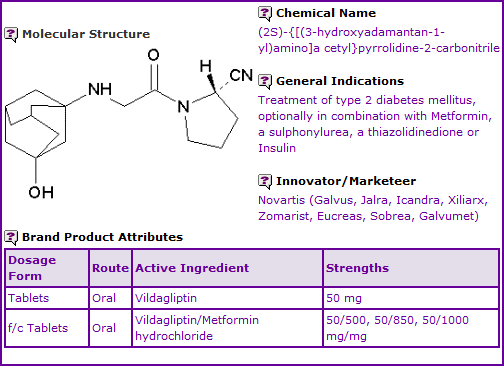
Vildagliptin is currently available as an oral tablet formulation. Oral fixed-dose combinations with Metformin (Eucreas®) are also available. Vildagliptin products are currently authorised in Europe and Australia, however it is not currently approved in Canada or the US.
Figure 1: General Information table for Ezetimibe
INN Constraint Comment
Pipeline Scope identified three key patent families relating to the Vildagliptin products, all of which contain patents that are the basis of SPC applications in Europe or s70 extensions in Australia.
The first key family (priority DE19616486A) relates to the use of DPP-4 inhibitors for the treatment of diseases which are based on glucose concentrations, a group amongst which Vildagliptin is included. However opposition proceedings were brought against the granted European patent within this family, EP0896538, by Boehringer Ingelheim, Bristol-Myers Squibb, Glaxo, Novartis, Novo Nordisk, Takeda, Tanabe Seiyaku and Pfizer, resulting in revocation of the patent in 2008. Divisional European applications claiming similar subject matter are currently still under examination. If granted and extended via SPC or s70, such patents would expire in September 2022. The Australian equivalent has been granted and will be in force until 2017.
The second key patent family identified, having priority US20906898A (see Figures 2 and 3), protects the Vildagliptin molecule specifically. In Europe, patent protection for Vildagliptin has been extended via Supplementary Protection Certificates (SPCs), expiring September 2022. Due to a later launch date of 2010 in Australia, the equivalent patent has been granted a full five year extension and expires more than two years after its European counterparts. Additional SPC applications have also been filed based on the EU marketing authorisation of the fixed dose combination of Vildagliptin with Metformin. These will provide nearly two months additional protection for the combination product if granted, however such SPC applications have been rejected in certain European states. Due to the lack of an extension system in Canada and an FDA approved product on which to base a s156 extension in the USA, the Vildagliptin molecule is able to be commercialised in these countries from 2019. Whilst a generic is not possible without an approved reference product, this opens up the opportunity to use manufacturing capabilities in these countries for launch stocks in Europe and Australia.
A third identified patent family (priority US32574400P) contains patents with normal expiry dates of 2021 and SPC applications potentially extending to November 2022, based on the marketing authorisation for the fixed dose combination of Vildagliptin and Metformin. Patents within this family claim combinations comprising DPP-4 inhibitors, specifically Vildagliptin, and a further anti-diabetic compound for simultaneous, separate or sequential use. Certain patents within the family specifically claim fixed-dose formulations comprising Vildagliptin in combination with Metformin and as such are considered to be constraining for marketed products containing this fixed combination.
Figure 2: Key Patent Indicator; the most significant patents protecting products containing Vildagliptin
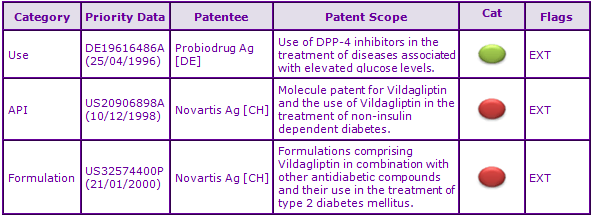
Figure 3: Patent Family View priority application US20906898A
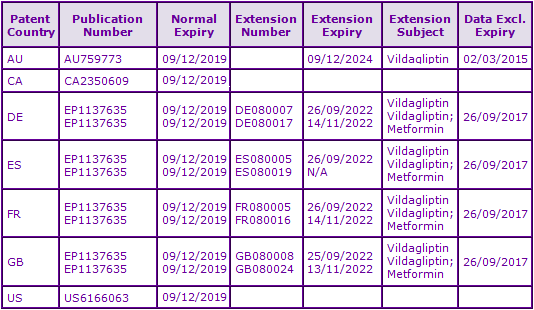
Figure 4:Top Patent Applicants
The graph below is a representation of patentees who have filed the most patent families for this INN. The innovator Novartis has filed nearly a quarter of the patent applications identified as relating to Vildagliptin. Note the diversity of applicants represented by the large portion outside the top 5, and also the prominence of those innovators identified in the aforementioned opposition proceedings including Boehringer Ingelheim (Linagliptin), Bristol-Myers Squibb (Saxagliptin), Takeda (Alogliptin) and GlaxoSmithKline due to cross over of claimed inventions into other, similar products.
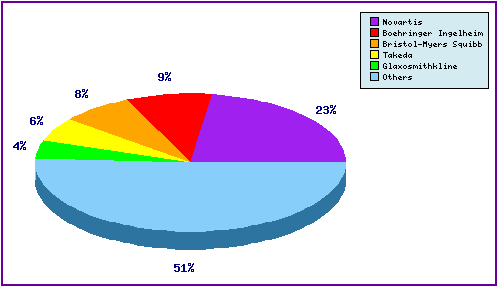
Figure 5: Patent Filing Trend
This graph represents the timing of the earliest priority filing date for each patent family identified for this molecule as well as the type of claims found in the applications. Note the consistent filing of patent applications in the area of combination, use and analytical method (assay) patents.
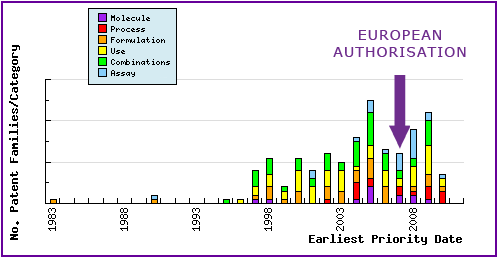
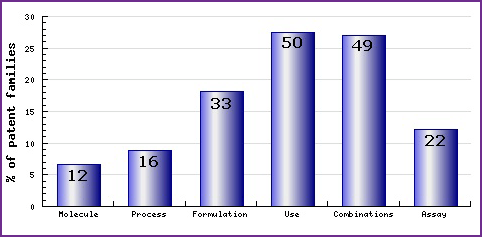
Figure 6: Patent Category Distribution
This figure represents the timing of the earliest priority filing date for each patent family identified for this molecule as well as the type of claims found in the applications. Note the increasing filing of patent applications in the area of process since the initial approval of Ezetimibe in 2002; and the consistent filing of combination patent applications since the later approval of Ezetimibe/Simvastatin combination products.
In summary, protection remains in force for the Vildagliptin molecule as a result of specific molecule patents which represent a barrier to generic competition to Galvus®. Furthermore, generic combinations of Vildagliptin and Metformin are also protected for an additional period of less than two months due to subsequent filing of SPCs. However the protection varies significantly across countries even in the EU, where patent protection is sought to be harmonised. Due to the complex patent landscape across three families, it is likely that the launch date of Vildagliptin generic products will vary significantly in individual countries. Subsequent protection of molecular forms, processes and formulations for this molecule appear to be relatively weak, thus when generic entry is possible, it is likely that it will be heavily competed.
The prominence and continuing growth of the global diabetes pharmaceutical industry means that even at this relatively early stage generic companies will closely watch the patent landscape surrounding Vildagliptin and any future developments in its lifecycle to ensure they overcome any obstacles and create opportunities by protecting technical work arounds with their own patents.
Do you want find out more about how we can assist your generic drug development?
Share this article:
To register for GenericsWeb's free monthly newsletter 'INNsight', click here
|